- Current
- Browse
- Collections
-
For contributors
- For Authors
- Instructions to authors
- Article processing charge
- e-submission
- For Reviewers
- Instructions for reviewers
- How to become a reviewer
- Best reviewers
- For Readers
- Readership
- Subscription
- Permission guidelines
- About
- Editorial policy
Articles
- Page Path
- HOME > Diabetes Metab J > Volume 42(4); 2018 > Article
-
Original ArticleClinical Diabetes and Therapeutics Cardio-Ankle Vascular Index as a Surrogate Marker of Early Atherosclerotic Cardiovascular Disease in Koreans with Type 2 Diabetes Mellitus
-
So Young Park1, Sang Ook Chin2, Sang Youl Rhee2, Seungjoon Oh2, Jeong-Taek Woo2, Sung Woon Kim2, Suk Chon2

-
Diabetes & Metabolism Journal 2018;42(4):285-295.
DOI: https://doi.org/10.4093/dmj.2017.0080
Published online: July 27, 2018
1Department of Medicine, Graduate School of Medicine, Kyung Hee University, Seoul, Korea.
2Department of Endocrinology and Metabolism, Kyung Hee University School of Medicine, Seoul, Korea.
- Corresponding author: Suk Chon. Department of Endocrinology and Metabolism, Kyung Hee University Hospital, Kyung Hee University School of Medicine, 23 Kyungheedae-ro, Dongdaemun-gu, Seoul 02447, Korea. imdrjs@khu.ac.kr
Copyright © 2018 Korean Diabetes Association
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/4.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
ABSTRACT
-
Background
- Carotid artery intima medial thickness (IMT), brachial-ankle pulse wave velocity (baPWV), and ankle-brachial index (ABI) are commonly used surrogate markers of subclinical atherosclerosis in patients with type 2 diabetes mellitus (T2DM). The cardio-ankle vascular index (CAVI) is a complement to the baPWV, which is affected by blood pressure. However, it is unclear which marker is the most sensitive predictor of atherosclerotic cardiovascular disease (ASCVD).
-
Methods
- This was a retrospective non-interventional study that enrolled 219 patients with T2DM. The correlations among IMT, ABI, and CAVI as well as the relationship of these tests to the 10-year ASCVD risk were also analyzed.
-
Results
- Among the 219 patients, 39 (17.8%) had ASCVD. In the non-ASCVD group, CAVI correlated significantly with IMT after adjusting for confounding variables, but ABI was not associated with CAVI or IMT. The analyses after dividing the non-ASCVD group into three subgroups according to the CAVI score (<8, ≥8 and <9, and ≥9) demonstrated the significant increase in the mean IMT, 10-year ASCVD risk and number of metabolic syndrome risk factors, and decrease in the mean ABI in the high-CAVI group. A high CAVI was an independent risk factor in the non-ASCVD group for both a high 10-year ASCVD risk (≥7.5%; odds ratio [OR], 2.42; P<0.001) and atherosclerosis (mean IMT ≥1 mm; OR, 1.53; P=0.007).
-
Conclusion
- In Korean patients with T2DM without ASCVD, CAVI was the most sensitive of several surrogate markers for the detection of subclinical atherosclerosis.
- Chronic complications of type 2 diabetes mellitus (T2DM) can be divided into macrovascular complications, including cardiovascular, cerebrovascular, and peripheral arterial disease, and microvascular complications, most commonly, retinopathy, nephropathy, and neuropathy. The increasing incidence of these chronic complications has been accompanied by an increase in the mortality rate among diabetic patients [12]. In Korea, the prevalence of myocardial infarction, cerebrovascular disease, cataracts, and glaucoma is higher in diabetic patients than in the normal population according to the Korean National Diabetes Program [3]. Annual screening for the microvascular and macrovascular complications of diabetes has thus been recommended to allow early detection and treatment.
- Atherosclerosis is the most prominent manifestation of cardiovascular disease, and atherosclerotic vascular changes lead to diabetic microvascular complications [45]. As demonstrated in several studies, the development of several surrogate markers of subclinical atherosclerosis, including the carotid artery intima medial thickness (IMT), ankle-brachial index (ABI), and pulse wave velocity (PWV), have contributed to the prevention of chronic diabetic complications [678]. IMT, which is measured by ultrasonography, reflects early atherosclerosis and vascular remodeling status. ABI is calculated by measuring upper- and lower-extremity blood pressure, and PWV is calculated by non-invasive evaluations of the velocity of a pulse wave traveling between two defined points in a blood vessel. Because PWV is significantly affected by blood pressure, an arterial stiffness index, the cardio-ankle vascular index (CAVI), is used to compensate for the weakness of PWV, and its utility has been well demonstrated [910]. However, the various tests conducted to evaluate T2DM-related complications are often economically burdensome for patients. Studies of the correlation of these surrogate markers and determination of a single marker that best detects subclinical atherosclerosis would therefore be of value. In this study we analyzed the correlation between surrogate markers of subclinical atherosclerosis and their optimal use in diabetic patients. The same markers were also examined for their relationship to 10-year atherosclerotic cardiovascular disease (ASCVD) risk.
INTRODUCTION
- Study population
- This study was conducted with 312 patients with T2DM who visited the Department of Endocrinology and Metabolism, Kyung Hee University Hospital, between July 2010 and December 2011. Patients 18 years of age or older diagnosed with T2DM who underwent IMT, ABI, and CAVI tests were included in the study. Patients in whom at least one of the tests was not performed were excluded, as were patients with severely compromised renal function (creatinine levels ≥1.6 mg/dL, overt proteinuria ≥1 g/day) or a history of cancer or depression. Thus, the final study population consisted of 219 patients.
- Study design
- This retrospective non-interventional study was conducted based on medical records; specifically, we reviewed patients' medical history and examination records. Baseline clinical characteristics included age, sex, physical examination results, smoking and alcohol consumption habits, exercise, and comorbidities. Biochemical examinations included complete blood counts; tests for liver enzyme, blood urea nitrogen, glycosylated hemoglobin, and creatinine levels; and lipid profiles. Tests for diabetes mellitus (DM) complications included the urinary albumin creatinine ratio, which is used to screen for microvascular complications, and the IMT, ABI, and CAVI tests, which are used to screen for macrovascular complications. We analyzed both the correlation among the latter three surrogate markers and the association between each one and the ASCVD risk or the presence of factors indicative of metabolic syndrome. The ASCVD was defined as coronary death or nonfatal myocardial infarction or as fatal or nonfatal stroke. Metabolic syndrome was defined according to the criteria proposed by the National Cholesterol Education Program's Adult Treatment Panel III (NCEP ATP III) [11]. The risk factors for metabolic syndrome include waist circumference ≥90 cm in men and ≥80 cm in women, fasting blood glucose ≥110 mg/dL, high density lipoprotein cholesterol (HDL-C) <50 mg/dL in women and <40 mg/dL in men, triglyceride ≥150 mg/dL, and blood pressure (systolic blood pressure ≥130 mm Hg, diastolic blood pressure ≥85 mm Hg).
- Measurement of carotid artery IMT
- A single skilled sonographer measured IMT using a high-resolution real-time B mode ultrasonography (HD11XE; Philips, Bothell, WA, USA) fitted with a linear 5- to 12-MHz probe. After the patient was stabilized in the supine position for approximately 10 minutes, the test was performed with the patient's head turned in the opposite direction from the examiner. IMT was measured at the far wall of the common carotid artery, 10 mm proximal to the bulb, and was defined as the distance between the lumen-intima interface and the media-adventitia interface. An absolute IMT value ≥1 mm was defined as atherosclerosis [1213]. A plaque was defined as a focal structure encroaching into the arterial lumen by 50% of the surrounding IMT value or demonstrating a thickness >1.2 mm. IMT and plaque features were analyzed using IntimaScope version 1.13E (Media Cross Co. Ltd., Tokyo, Japan). Mean and maximal IMT were measured on the left and right sides, and the larger set of values was included in the analysis.
- Measurement of ABI and CAVI
- The ABI and CAVI were measured using the VaSera VS-1000 (Fukuda Denshi Co. Ltd., Tokyo, Japan). After the patient had been stabilized in the supine position for 5 minutes, blood pressure at the posterior tibial and brachial arteries was measured. Dividing the ankle systolic blood pressure by the arm systolic blood pressure yielded the ABI. A value <0.9 was interpreted as lower-limb arterial occlusion; that between 0.9 and 1.3 was interpreted as normal; and that >1.3 was interpreted as lower-extremity arterial calcification and stiffness. The right and left sides were measured, and the value closer to 0.9 was used for the analysis. Four blood pressure cuffs were wrapped on the four extremities to measure the CAVI. Electrocardiography electrodes were attached to both arms, and a microphone was placed on the sternum in the second intercostal space. After the patient had been stabilized in the supine position for 10 minutes, electrocardiography and phonocardiography were monitored. CAVI was calculated using Bramwell-Hill's equation [14]: CAVI=a[(2ρ/ΔP)×ln(Ps/Pd)PWV2]+b, where ‘Ps’ is the systolic blood pressure; ‘Pd’ is the diastolic blood pressure; ‘ΔP’ is Ps–Pd; ‘PWV’ is the cardio-ankle pulse wave velocity; ‘ρ’ is the blood viscosity; and ‘a’ and ‘b’ are the constants for converting the CAVI value to a value obtained using the Hasegawa method. A CAVI value <8 was interpreted as a mild risk of atherosclerosis; that between 8 and 9 was interpreted as a moderate risk of atherosclerosis; and that >9 was interpreted as a high risk of atherosclerosis. The higher of the measured right and left CAVI values was used for the analysis.
- ASCVD risk assessment
- The patients were divided into ASCVD and non-ASCVD groups according to their history of ASCVD. The medical history documented in the records was used to assign patients to the groups. In the non-ASCVD group, the cardiovascular morbidity and mortality risks were estimated according to the 10-year ASCVD risk protocol suggested by the 2013 American College of Cardiology/American Heart Association (ACC/AHA) guideline and based on the Pooled Cohort Equations as well as the work of Lloyd-Jones. This algorithm was published in the 2013 ACC/AHA guideline on the assessment of cardiovascular risk [15]. The estimate of 10-year ASCVD risk was based on sex, age, race (two groups: African-Americans and Caucasians or others), total cholesterol, HDL-C, systolic blood pressure, treatment for hypertension, history of DM, and smoking history.
- Statistical analysis
- All data are expressed as numbers with proportions for categorical variables or as mean±standard deviation for numerical variables. Differences in the means of the different groups were analyzed using the chi-square test for categorical variables and Student t-test or analysis of variance (ANOVA) for continuous variables. Tukey and Dunnett T3 methods were used for post hoc analysis. Spearman correlation analysis was used to analyze the relationship among ABI, IMT, and CAVI. A multivariate logistic regression analysis was performed to evaluate the utility of CAVI as a surrogate marker for atherosclerosis (mean IMT ≥1 mm) and a high (≥7.5%) 10-year ASCVD risk in the non-ASCVD group. The sensitivity and specificity of a high 10-year ASCVD risk (≥7.5%) in the non-ASCVD group were analyzed using a conventional receiver operating characteristic (ROC) curve. The statistical analyses were carried out using PASW version 20.0 software (IBM Co., Armonk, NY, USA). A P<0.05 was considered to indicate statistical significance.
- Ethics statement
- Approval for this study was obtained from the Institutional Review Board of Kyung Hee University Hospital (KMC IRB 1129-02). An informed consent was waived due to a retrospective study.
METHODS
- Baseline clinical characteristics
- Table 1 shows the baseline clinical characteristics of the patients. The mean age was 58.8±9.7 years, and the mean duration of DM was 9.0±6.7 years. Patients in the ASCVD group (62.0±9.9 years; n=39, 17.8%) were significantly older than those in the non-ASCVD group (58.1±9.5 years; n=180, 82.2%; P=0.022) and had a significantly higher BMI and waist circumference than the non-ASCVD group (26.4±2.4 kg/m2 vs. 25.3±3.3 kg/m2, P=0.049; 95.3±11.9 cm vs. 89.5±8.5 cm, P<0.05, respectively). The differences in the triglyceride and HDL-C levels were not significant, but the low density lipoprotein cholesterol (LDL-C) and apolipoprotein B levels were lower in the ASCVD than in the non-ASCVD group (85.9±29.5 mg/dL vs. 99.4±32.3 mg/dL and 87.7±19.3 mg/dL vs. 96.5±24.0 mg/dL, respectively; P<0.05). The use of lipid-lowering agents was more common in the ASCVD than in the non-ASCVD group (P<0.001), and the mean CAVI (8.8±1.4 vs. 8.1±1.3, P=0.004), mean IMT (1.12±0.28 mm vs. 0.99±0.21 mm, P=0.009), and maximal IMT (1.40±0.35 mm vs. 1.26±0.29 mm, P=0.006) were higher in the ASCVD than in the non-ASCVD group.
- Clinical characteristics according to CAVI in the non-ASCVD group
- The non-ASCVD group was divided into three subgroups according to the CAVI: low (CAVI <8, n=92), moderate (8≤CAVI<9, n=50), and high (CAVI ≥9, n=38). The clinical characteristics of the groups were compared using an ANOVA and a chi-square test (Table 2). Age, blood pressure, history of hypertension, and use of anti-hypertensive agents were elevated in the high-CAVI group (P<0.05). Mean IMT (0.94±0.18 mm vs. 1.00±0.20 mm vs. 1.09±0.27 mm, P=0.001) and maximal IMT (1.21±0.27 mm vs. 1.26±0.28 mm vs. 1.37±0.31 mm, P=0.012) increased with increasing CAVI, as did the presence of atherosclerotic plaques (51.1% vs. 80.0% vs. 86.8% in the low, moderate, and high subgroups, respectively). There were no differences in the LDL-C and triglyceride levels among the three groups, but the HDL-C levels were lowest in the high CAVI group (50.3±12.1 mg/dL vs. 56.3±14.8 mg/dL vs. 48.4±11.7 mg/dL, P=0.008). ABI was also lowest in the high CAVI group (1.12±0.07 vs. 1.11±0.07 vs. 1.08±0.08, P=0.013). The 10-year ASCVD risk (10.1%±8.4% vs. 14.0%±9.2% vs. 26.9%±16.3%, P<0.001) and prevalence of metabolic syndrome (63.0% vs. 68.0% vs. 86.5%, P=0.032) increased in proportion to the CAVI score.
- Correlations among CAVI, IMT, and ABI and association with the 10-year ASCVD risk
- Table 3 shows the correlations among the CAVI, IMT, and ABI. CAVI was correlated with both the IMT and the ABI in all diabetic patients (P<0.05), whereas there was no correlation between ABI and IMT. In the non-ASCVD group, only the correlation between CAVI and IMT was significant. An additional analysis was conducted in the non-ASCVD group to determine the correlation between each surrogate marker of subclinical atherosclerosis and the 10-year ASCVD risk score or the number of metabolic syndrome risk factors. The results showed that ABI was not associated with either the 10-year ASCVD risk score or the number of metabolic syndrome risk factors, whereas CAVI and IMT were correlated with the 10-year ASCVD risk score. The highest correlation coefficient was that for CAVI. IMT was correlated with the number of metabolic syndrome risk factors.
- The value of CAVI for predicting ASCVD
- After an adjustment for confounding factors, CAVI was identified as an independent risk factor for ASCVD and atherosclerosis (Table 4). Therefore, we analyzed the value of CAVI for predicting both the prevalence of ASCVD in all diabetic patients and the risk of ASCVD and atherosclerosis in diabetic patients without a history of ASCVD. The odds ratio (OR) of CAVI for ASCVD prevalence in all patients was 1.38 (95% confidence interval [CI], 1.06 to 1.80; P=0.017). In the non-ASCVD group, the OR of CAVI for the high 10-year ASCVD risk group (≥7.5%) was 2.42 (95% CI, 1.64 to 3.58; P<0.001). Based on a definition of atherosclerosis as a mean IMT ≥1 mm, the OR of CAVI for atherosclerosis in the non-ASCVD group was 1.53 (95% CI, 1.12 to 2.08; P=0.007).
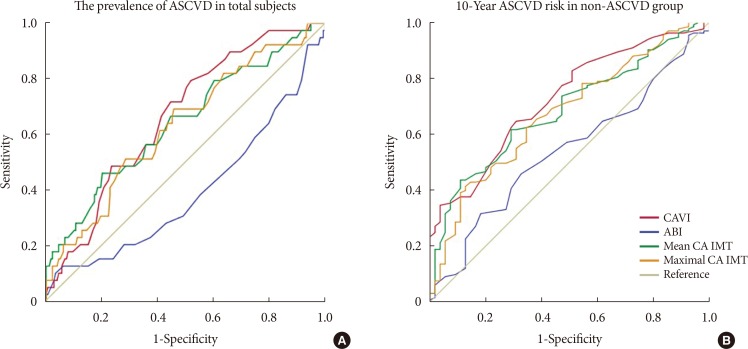
- To detect the prevalence of ASCVD in all patients, the cut-off values and areas under the curve (AUCs) were determined for CAVI, ABI, and IMT. For CAVI, the cut-off was 8.05 with an AUC of 0.66; for ABI, the values were 1.08 and 0.39, respectively; for mean IMT, they were 0.995 and 0.64, respectively; and for maximal IMT, they were 1.225 and 0.63, respectively (Fig. 1A). To detect a high risk of 10-year ASCVD in the non-ASCVD group, the cut-off value was 7.65 with an AUC of 0.74 for CAVI, 1.11 with an AUC of 0.53 for ABI, 0.925 with an AUC of 0.70 for mean IMT, and 1.165 with an AUC of 0.68 for maximal IMT (Fig. 1B).
RESULTS
- This study analyzed the correlations among three surrogate markers of subclinical atherosclerosis and demonstrated the superiority of CAVI. Diabetic patients with a history of ASCVD were more likely to be older, more obese, and to have a high CAVI and IMT. Among diabetic patients without a history of ASCVD, CAVI was correlated with IMT, whereas ABI was not associated with either CAVI or IMT. Thus, whereas CAVI and IMT reflect systemic atherosclerosis, ABI is an indicator of peripheral artery disease of the lower extremities and therefore of local significance. In the ROC curve analysis, among the three surrogate markers, CAVI was of the greatest utility for evaluating the prevalence of ASCVD in both all patients and in the potential high ASCVD risk group. In the non-ASCVD group, the 10-year ASCVD risk was ≥7.5%. Additionally, age, IMT, prevalence of metabolic syndrome, number of metabolic syndrome risk factors, and 10-year ASCVD risk were all significantly higher in the high CAVI group (CAVI ≥9). After adjusting for confounding factors, a high CAVI was a risk factor for ASCVD in all patients as well as a risk factor for atherosclerosis in the non-ASCVD group based on the mean IMT and a high (≥7.5%) 10-year ASCVD risk. According to this result, CAVI is the most sensitive surrogate marker of subclinical atherosclerosis.
- The utility of CAVI has been extensively studied in Japan. In the study performed by Izuhara et al. [16], a high CAVI indicated the progression of carotid and coronary artery arteriosclerosis, and CAVI was more closely linked with arteriosclerosis than was brachial-ankle PWV. Takenaka et al. [17] demonstrated the utility of CAVI for predicting cardiovascular disease in patients undergoing hemodialysis, and Mineoka et al. [18] reported a positive correlation between CAVI and the coronary artery calcification score determined by multislice computed tomography in patients with T2DM. Similar studies have been conducted in Korean and Caucasian patients [192021]. Our results showed a strong correlation between CAVI and IMT and thus the utility of the CAVI as a surrogate marker of ASCVD. However, unlike other studies, we found a weak correlation between CAVI and ABI. This was probably due to the larger number of patients with an ABI <0.9 (at least 1.5% of all subjects) in previous studies than in our study, in which only three of the 219 patients had an ABI <0.9. Nonetheless, our results showed that, among the three surrogate markers of subclinical atherosclerosis, CAVI had the highest sensitivity and specificity. Additionally, we identified an association between a high CAVI and both a higher 10-year ASCVD risk score and a larger number of metabolic syndrome risk factors. Although there may be limitations in directly comparing the 10-year ASCVD risk calculated according to the 2013 ACC/AHA guidelines with the CAVI, ABI, and IMT values, because they may be affected by genetic background and complication progress, the three markers could be used complementarily to assess cardiovascular risk.
- Limitations in the utility of the traditional surrogate markers of subclinical atherosclerosis have been suggested in several studies. The sensitivity and specificity of ABI decreases in diabetic patients due to higher rates of medial arterial calcification [22]. Despite several studies in which IMT was used as a surrogate marker of cardiovascular disease and atherosclerosis [81323], limitations in this method have been suggested because the value of this variable may increase for reasons not related to atherosclerosis, such as smooth muscle cell hyperplasia or fibrocellular hypertrophy [24]. Moreover, it is difficult to evaluate the vulnerability of a carotid artery plaque by ultrasonography. For this reason, the 2013 ACC/AHA guideline did not support routine carotid artery IMT as a risk assessment for an ASCVD event, regardless of initial risk stratification by traditional methods. IMT was therefore downgraded from a class IIa (reasonable) to a class III (none) benefit [25]. Given the limitations of these surrogate markers and the high medical costs incurred by diabetic patients, our demonstration of a strong correlation between CAVI and traditional surrogate markers of subclinical atherosclerosis as well as the superiority and utility of CAVI is meaningful. Our results are also supported by studies reporting the value of CAVI as a clinical marker for carotid and coronary arteriosclerosis screening and as a parameter for the prediction of disease severity and early stage left ventricular diastolic stiffness [2627]. In our study, CAVI, which reflects the stiffness of the thoracic, abdominal, common iliac, and distal arteries and their involvement in arteriosclerosis, was more representative of systemic arteriosclerosis than was IMT, which reflects carotid artery status, and ABI, an indicator of reflects lower-extremity status. This finding explains the superiority of CAVI as a marker. CAVI is also more valuable than PWV because it is not affected by changes in blood pressure in response to the confounding environmental and psychological factors associated with the examination itself; rather, it reflects autonomic nervous system function, including vasoconstriction [10]. The superior clinical value of CAVI vs. PWV was also reported previously [16].
- However, one limitation of CAVI is that, although the formula to calculate it is based on the assumption that vascular diameter, blood pressure, and the elasticity from the aortic valve to the ankle are constant, this is not always the case. Other limitations of our study included its inclusion of patients from a single tertiary medical institution, which could limit the generalizability of the results. Multicenter, large-scale studies are therefore needed. Additionally, a 10-year ASCVD risk assessment model (pooled cohort equation) based on the 2013 ACC/AHA guideline and developed for non-Hispanic Caucasian and African-American populations suggested that the risk factors differ for Asians and are thus not more broadly applicable. Moreover, only patients 40 to 75 years of age were included in our model. Risk factors are commonly measured in this age group but not in older or younger patients. Also, there are currently very few ASCVD-risk equations suitable for Asians. The Korean Risk Prediction Model for ASCVD published by Jung et al. [28] has several limitations regarding its general use. Unlike the latter, although CAVI compensates for the weakness of the PWV, its use has not been validated for the Korean population. Our study thus provides important evidence of the utility of the CAVI as a new screening option.
- In conclusion, CAVI was positively correlated with both the 10-year ASCVD risk and IMT and was the most sensitive of the tested surrogate markers for the detection of subclinical atherosclerosis in Korean diabetic patients without ASCVD.
DISCUSSION
-
Acknowledgements
- This research was supported by grants from the Korea Health Technology R&D Project through the Korea Health Industry Development Institute (KHIDI), funded by the Ministry of Health and Welfare, Republic of Korea (grant number: HI14C 2700 020014, HC15C3364).
ACKNOWLEDGMENTS
-
CONFLICTS OF INTEREST: No potential conflict of interest relevant to this article was reported.
NOTES
- 1. Yoon KH, Lee JH, Kim JW, Cho JH, Choi YH, Ko SH, Zimmet P, Son HY. Epidemic obesity and type 2 diabetes in Asia. Lancet 2006;368:1681-1688. ArticlePubMed
- 2. Chan JC, Malik V, Jia W, Kadowaki T, Yajnik CS, Yoon KH, Hu FB. Diabetes in Asia: epidemiology, risk factors, and pathophysiology. JAMA 2009;301:2129-2140. ArticlePubMed
- 3. Rhee SY, Chon S, Kwon MK, Park IeB, Ahn KJ, Kim IJ, Kim SH, Lee HW, Koh KS, Kim DM, Baik SH, Lee KW, Nam MS, Park YS, Woo JT, Kim YS. Prevalence of chronic complications in korean patients with type 2 diabetes mellitus based on the Korean National Diabetes Program. Diabetes Metab J 2011;35:504-512. ArticlePubMedPMC
- 4. Ogawa O, Hayashi C, Nakaniwa T, Tanaka Y, Kawamori R. Arterial stiffness is associated with diabetic retinopathy in type 2 diabetes. Diabetes Res Clin Pract 2005;68:162-166. ArticlePubMed
- 5. Meyer C, Milat F, McGrath BP, Cameron J, Kotsopoulos D, Teede HJ. Vascular dysfunction and autonomic neuropathy in type 2 diabetes. Diabet Med 2004;21:746-751. ArticlePubMedPDF
- 6. Yamashina A, Tomiyama H, Arai T, Hirose K, Koji Y, Hirayama Y, Yamamoto Y, Hori S. Brachial-ankle pulse wave velocity as a marker of atherosclerotic vascular damage and cardiovascular risk. Hypertens Res 2003;26:615-622. ArticlePubMed
- 7. Kojima I, Ninomiya T, Hata J, Fukuhara M, Hirakawa Y, Mukai N, Yoshida D, Kitazono T, Kiyohara Y. A low ankle brachial index is associated with an increased risk of cardiovascular disease: the Hisayama study. J Atheroscler Thromb 2014;21:966-973. ArticlePubMed
- 8. Nichols WW, Pepine CJ, O'Rourke MF. Carotid-artery intima and media thickness as a risk factor for myocardial infarction and stroke. N Engl J Med 1999;340:1762-1763.Article
- 9. Shirai K, Hiruta N, Song M, Kurosu T, Suzuki J, Tomaru T, Miyashita Y, Saiki A, Takahashi M, Suzuki K, Takata M. Cardio-ankle vascular index (CAVI) as a novel indicator of arterial stiffness: theory, evidence and perspectives. J Atheroscler Thromb 2011;18:924-938. ArticlePubMed
- 10. Kim B, Takada K, Oka S, Misaki T. Influence of blood pressure on cardio-ankle vascular index (CAVI) examined based on percentage change during general anesthesia. Hypertens Res 2011;34:779-783. ArticlePubMedPDF
- 11. Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults. Executive summary of the third report of the National Cholesterol Education Program (NCEP) expert panel on detection, evaluation, and treatment of high blood cholesterol in adults (Adult Treatment Panel III). JAMA 2001;285:2486-2497. ArticlePubMed
- 12. Price JF, Tzoulaki I, Lee AJ, Fowkes FG. Ankle brachial index and intima media thickness predict cardiovascular events similarly and increased prediction when combined. J Clin Epidemiol 2007;60:1067-1075. ArticlePubMed
- 13. Chambless LE, Heiss G, Folsom AR, Rosamond W, Szklo M, Sharrett AR, Clegg LX. Association of coronary heart disease incidence with carotid arterial wall thickness and major risk factors: the Atherosclerosis Risk in Communities (ARIC) Study, 1987–1993. Am J Epidemiol 1997;146:483-494. ArticlePubMed
- 14. Shirai K, Utino J, Otsuka K, Takata M. A novel blood pressure-independent arterial wall stiffness parameter: cardio-ankle vascular index (CAVI). J Atheroscler Thromb 2006;13:101-107. ArticlePubMed
- 15. Andrus B, Lacaille D. 2013 ACC/AHA guideline on the assessment of cardiovascular risk. J Am Coll Cardiol 2014;63:2886.Article
- 16. Izuhara M, Shioji K, Kadota S, Baba O, Takeuchi Y, Uegaito T, Mutsuo S, Matsuda M. Relationship of cardio-ankle vascular index (CAVI) to carotid and coronary arteriosclerosis. Circ J 2008;72:1762-1767. ArticlePubMed
- 17. Takenaka T, Hoshi H, Kato N, Kobayashi K, Takane H, Shoda J, Suzuki H. Cardio-ankle vascular index to screen cardiovascular diseases in patients with end-stage renal diseases. J Atheroscler Thromb 2008;15:339-344. ArticlePubMed
- 18. Mineoka Y, Fukui M, Tanaka M, Tomiyasu K, Akabame S, Nakano K, Yamazaki M, Hasegawa G, Oda Y, Nakamura N. Relationship between cardio-ankle vascular index (CAVI) and coronary artery calcification (CAC) in patients with type 2 diabetes mellitus. Heart Vessels 2012;27:160-165. ArticlePubMedPDF
- 19. Gomez-Sanchez L, Garcia-Ortiz L, Patino-Alonso MC, Recio-Rodriguez JI, Frontera G, Ramos R, Marti R, Agudo-Conde C, Rodriguez-Sanchez E, Maderuelo-Fernandez JA, Gomez-Marcos MA. MARK Group. The association between the cardio-ankle vascular index and other parameters of vascular structure and function in Caucasian adults: MARK Study. J Atheroscler Thromb 2015;22:901-911. ArticlePubMed
- 20. Gomez-Marcos MA, Recio-Rodriguez JI, Patino-Alonso MC, Agudo-Conde C, Gomez-Sanchez L, Gomez-Sanchez M, Rodriguez-Sanchez E, Maderuelo-Fernandez JA, Garcia-Ortiz L. LOD-DIABETES Group. Cardio-ankle vascular index is associated with cardiovascular target organ damage and vascular structure and function in patients with diabetes or metabolic syndrome, LOD-DIABETES study: a case series report. Cardiovasc Diabetol 2015;14:7. PubMedPMC
- 21. Park HE, Choi SY, Kim MK, Oh BH. Cardio-ankle vascular index reflects coronary atherosclerosis in patients with abnormal glucose metabolism: assessment with 256 slice multi-detector computed tomography. J Cardiol 2012;60:372-376. ArticlePubMed
- 22. Williams DT, Harding KG, Price P. An evaluation of the efficacy of methods used in screening for lower-limb arterial disease in diabetes. Diabetes Care 2005;28:2206-2210. ArticlePubMedPDF
- 23. Bots ML, Hoes AW, Koudstaal PJ, Hofman A, Grobbee DE. Common carotid intima-media thickness and risk of stroke and myocardial infarction: the Rotterdam Study. Circulation 1997;96:1432-1437. ArticlePubMed
- 24. Huibers A, de Borst GJ, Wan S, Kennedy F, Giannopoulos A, Moll FL, Richards T. Non-invasive carotid artery imaging to identify the vulnerable plaque: current status and future goals. Eur J Vasc Endovasc Surg 2015;50:563-572. ArticlePubMed
- 25. Goff DC Jr, Lloyd-Jones DM, Bennett G, Coady S, D'Agostino RB Sr, Gibbons R, Greenland P, Lackland DT, Levy D, O'Donnell CJ, Robinson JG, Schwartz JS, Shero ST, Smith SC Jr, Sorlie P, Stone NJ, Wilson PW. American College of Cardiology/American Heart Association Task Force on Practice Guidelines. 2013 ACC/AHA guideline on the assessment of cardiovascular risk: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. J Am Coll Cardiol 2014;63:2935-2959. PubMed
- 26. Cho KH, Kim KP, Woo BC, Kim YJ, Park JY, Cho SY, Park SU, Jung WS, Park JM, Moon SK. Relationship between blood stasis syndrome score and cardioankle vascular index in stroke patients. Evid Based Complement Alternat Med 2012;2012:696983. ArticlePubMedPMCPDF
- 27. Mizuguchi Y, Oishi Y, Tanaka H, Miyoshi H, Ishimoto T, Nagase N, Oki T. Arterial stiffness is associated with left ventricular diastolic function in patients with cardiovascular risk factors: early detection with the use of cardio-ankle vascular index and ultrasonic strain imaging. J Card Fail 2007;13:744-751. ArticlePubMed
- 28. Jung KJ, Jang Y, Oh DJ, Oh BH, Lee SH, Park SW, Seung KB, Kim HK, Yun YD, Choi SH, Sung J, Lee TY, Kim SH, Koh SB, Kim MC, Chang Kim H, Kimm H, Nam C, Park S, Jee SH. The ACC/AHA 2013 pooled cohort equations compared to a Korean risk prediction model for atherosclerotic cardiovascular disease. Atherosclerosis 2015;242:367-375. ArticlePubMed
REFERENCES
The utility of cardio-ankle vascular index (CAVI), ankle-brachial index (ABI), mean intima medial thickness (IMT), and maximal IMT for predicting the prevalence of atherosclerotic cardiovascular disease (ASCVD) in all subjects (A), and 10-year ASCVD risk scores in subjects without ASCVD (B). CA, carotid artery.

Baseline clinical characteristics
Values are presented as mean±standard deviation or number (%). For P<0.05 with student t-test and chi-square test.
ASCVD, atherosclerotic cardiovascular disease; WBC, white blood cell; GOT, glutamate oxaloacetate transaminase; GPT, glutamate pyruvate transaminase; GGT, γ-glutamyltransferase; HbA1c, glycosylated hemoglobin; HDL-C, high density lipoprotein cholesterol; LDL-C, low density lipoprotein cholesterol; BUN, blood urea nitrogen; eGFR, estimated glomerular filtration rate; hsCRP, high-sensitivity C-reactive protein; CAVI, cardio-ankle vascular index; ABI, ankle-brachial index; IMT, intima media thickness.
The comparison of clinical characteristics according to the CAVI in the non-ASCVD group
Values are presented as mean±standard deviation or number (%). For P<0.05 with chi-square test. For post hoc tests, Tukey method in case of equal variance satisfied, and otherwise, Dunnett T3 method.
CAVI, cardio-ankle vascular index; ASCVD, atherosclerotic cardiovascular disease; WBC, white blood cell; GOT, glutamate oxaloacetate transaminase; GPT, glutamate pyruvate transaminase; GGT, γ-glutamyltransferase; HbA1c, glycosylated hemoglobin; HDL-C, high density lipoprotein cholesterol; LDL-C, low density lipoprotein cholesterol; BUN, blood urea nitrogen; hsCRP, high-sensitivity C-reactive protein; ABI, ankle-brachial index; IMT, intima media thickness.
aP<0.05 between mild and moderate, bP<0.05 between mild and high, cP<0.05 between moderate and high.
Correlations among CAVI, IMT, and ABI, and the association of 10-year ASCVD risk
The value of CAVI to predict the ASCVD
Values are presented as β (95% confidence interval). Multivariable logistic regression analysis, P<0.05.
CAVI, cardio-ankle vascular index; ASCVD, atherosclerotic cardiovascular disease; IMT, intima media thickness.
aModel 1: adjusted by age, gender, diabetes mellitus (DM) duration, body mass index (BMI), smoking, lipid lowering agents, and antihypertensive agents, bModel 2: adjusted by age, gender, DM duration, BMI, smoking, lipid lowering agents, and antihypertensive agents, cModel 3: adjusted by DM duration, BMI, and lipid lowering agents.
Figure & Data
References
Citations

- The effects of severe periodontitis on arterial stiffness using cardio‐ankle vascular index in patients with type 2 diabetes
Gizem Torumtay Cin, Semin Melahat Fenkci, Ismail Doğu Kiliç, Halil Serdar Aslan, Cihan İlyas Sevgican, Hande Şenol
Journal of Periodontal Research.2024; 59(1): 74. CrossRef - Prediction of cardiovascular disease using deep learning algorithms to prevent COVID 19
Malathi S, Arockia Raj Y, Abhishek Kumar, V D Ashok Kumar, Ankit Kumar, Elangovan D, V D Ambeth Kumar, Chitra B, a Abirami
Journal of Experimental & Theoretical Artificial Intelligence.2023; 35(6): 791. CrossRef - Association of cardio-ankle vascular index and future major adverse cardiovascular events in older adults living with HIV
Amaraporn Rerkasem, Arunrat Tangmunkongvorakul, Linda Aurpibul, Patumrat Sripan, Wason Parklak, Sothida Nantakool, Kriengkrai Srithanaviboonchai, Kittipan Rerkasem
AIDS Care.2023; 35(4): 591. CrossRef - Impact of Fasting Blood Glucose Levels on Blood Pressure Parameters among Older Adults with Prediabetes
Thapanee Roengrit, Ruchada Sri-Amad, Nawiya Huipao, Suphawadee Phababpha, Piyapong Prasertsri, Francesco Giallauria
The Scientific World Journal.2023; 2023: 1. CrossRef - A framework of biomarkers for vascular aging: a consensus statement by the Aging Biomarker Consortium
Le Zhang, Jun Guo, Yuehong Liu, Shimin Sun, Baohua Liu, Qi Yang, Jun Tao, Xiao-Li Tian, Jun Pu, Huashan Hong, Miao Wang, Hou-Zao Chen, Jie Ren, Xiaoming Wang, Zhen Liang, Yuan Wang, Kai Huang, Weiqi Zhang, Jing Qu, Zhenyu Ju, Guang-Hui Liu, Gang Pei, Jian
Life Medicine.2023;[Epub] CrossRef - Diastolic Pressure and ACR Are Modifiable Risk Factors of Arterial Stiffness in T2DM Without Cardiovascular Disease
Gateano Leto, Lida Tartaglione, Silverio Rotondi, Marzia Pasquali, Ernesto Maddaloni, Carmen Mignogna, Luca D’Onofrio, Simona Zampetti, Angela Carlone, Maria Luisa Muci, Daniela Mastroluca, Valeria Fassino, Raffaella Buzzetti, Sandro Mazzaferro
The Journal of Clinical Endocrinology & Metabolism.2022; 107(9): e3857. CrossRef - Risk assessment indicators and brachial-ankle pulse wave velocity to predict atherosclerotic cardiovascular disease
Hung-Ju Ko, Chuan-Chuan Liu, Po-Jui Hsu, Kuang-Chun Hu, Chung-Lieh Hung, Lo-Yip Yu, Yun-Chieh Huang, Shou-Chuan Shih
Medicine.2022; 101(32): e29609. CrossRef - Prediction of Cardiovascular Disease Using Machine Learning Technique—A Modern Approach
Jung-Hwa Kim, Jin-Woo Jeong
Computers, Materials & Continua.2022; 71(1): 855. CrossRef - Cardio-ankle vascular index represents the best surrogate for 10-year ASCVD risk estimation in patients with primary hypertension
Mustafa Tarik Agac, Süret Ağaç, Muhammed Necati Murat Aksoy, Mehmet Bülent Vatan
Clinical and Experimental Hypertension.2021; 43(4): 349. CrossRef - Relation between fragmented QRS complex and cardio-ankle vascular index in asymptomatic subjects
Ali Rıza Akyüz, Sinan Şahin, Ömer Faruk Çırakoğlu, Selim Kul, Turhan Turan, Hakan Erkan
Clinical and Experimental Hypertension.2021; 43(4): 368. CrossRef - Progress of clinical evaluation for vascular aging in humans
Yumin Qiu, Yuanya Liu, Jun Tao
Journal of Translational Internal Medicine.2021; 9(1): 17. CrossRef - Effects of long-term air pollution exposure on ankle-brachial index and cardio-ankle vascular index: A longitudinal cohort study using data from the Electricity Generating Authority of Thailand study
Kanawat Paoin, Kayo Ueda, Prin Vathesatogkit, Thammasin Ingviya, Suhaimee Buya, Arthit Phosri, Xerxes Tesoro Seposo, Nisakron Thongmung, Teerapat Yingchoncharoen, Akiko Honda, Hirohisa Takano, Piyamitr Sritara
International Journal of Hygiene and Environmental Health.2021; 236: 113790. CrossRef The Relationship Between Glycemic Control and Concomitant Hypertension on Arterial Stiffness in Type II Diabetes
Teonchit Nuamchit, Duangduan Siriwittayawan, Piyanuch Thitiwuthikiat
Vascular Health and Risk Management.2020; Volume 16: 343. CrossRef- Relationship between cardio-ankle vascular index and obstructive coronary artery disease
Divya Birudaraju, Lavanya Cherukuri, April Kinninger, Bhanu T. Chaganti, Pishoy Haroun, Sivakrishna Pidikiti, Suvasini Lakshmanan, Sajad Hamal, Ferdinand Flores, Christopher Dailing, Kashif Shaikh, Sion K. Roy, Matthew J. Budoff
Coronary Artery Disease.2020; 31(6): 550. CrossRef - Association of Kidney Function Tests with a Cardio-Ankle Vascular Index in Community-Dwelling Individuals with a Normal or Mildly Decreased Estimated Glomerular Filtration Rate
Javad Alizargar, Chyi-Huey Bai, Nan-Chen Hsieh, Shu-Fang Vivienne Wu, Shih-Yen Weng, Jia-Ping Wu
Medicina.2019; 55(10): 657. CrossRef - Cardiovascular remodeling in patients with diabetic сardiomyopathy
A. S. Veklich, N. A. Koziolova, P. G. Karavaev
Russian Journal of Cardiology.2019; (11): 42. CrossRef - Short‑term impact of aged garlic extract on endothelial function in diabetes: A randomized, double‑blind, placebo‑controlled trial
Sajad Hamal, Lavanya Cherukuri, Divya Birudaraju, Suguru Matsumoto, April Kinninger, Bhanu Chaganti, Ferdinand Flores, Kashif Shaikh, Sion Roy, Matthew Budoff
Experimental and Therapeutic Medicine.2019;[Epub] CrossRef - Association between Breakfast Frequency and Atherosclerotic Cardiovascular Disease Risk: A Cross-Sectional Study of KNHANES Data, 2014–2016
Hyeon Ji Lee, Jieun Jang, Sang Ah Lee, Dong-Woo Choi, Eun-Cheol Park
International Journal of Environmental Research and Public Health.2019; 16(10): 1853. CrossRef - Response: Cardio-Ankle Vascular Index as a Surrogate Marker of Early Atherosclerotic Cardiovascular Disease in Koreans with Type 2 Diabetes Mellitus (Diabetes Metab J 2018;42:285-95)
So Young Park, Suk Chon
Diabetes & Metabolism Journal.2018; 42(5): 449. CrossRef - Letter: Cardio-Ankle Vascular Index as a Surrogate Marker of Early Atherosclerotic Cardiovascular Disease in Koreans with Type 2 Diabetes Mellitus (Diabetes Metab J 2018;42:285-95)
Dongwon Yi
Diabetes & Metabolism Journal.2018; 42(5): 447. CrossRef

 KDA
KDA PubReader
PubReader Cite
Cite