
- Current
- Browse
- Collections
-
For contributors
- For Authors
- Instructions to authors
- Article processing charge
- e-submission
- For Reviewers
- Instructions for reviewers
- How to become a reviewer
- Best reviewers
- For Readers
- Readership
- Subscription
- Permission guidelines
- About
- Editorial policy
Search
- Page Path
- HOME > Search
Review
- Pathophysiology
- Immune-Checkpoint Inhibitors-Induced Type 1 Diabetes Mellitus: From Its Molecular Mechanisms to Clinical Practice
- Yun Kyung Cho, Chang Hee Jung
- Diabetes Metab J. 2023;47(6):757-766. Published online July 24, 2023
- DOI: https://doi.org/10.4093/dmj.2023.0072
- 2,970 View
- 341 Download
- 2 Crossref
-
 Abstract
Abstract
 PDF
PDF PubReader
PubReader  ePub
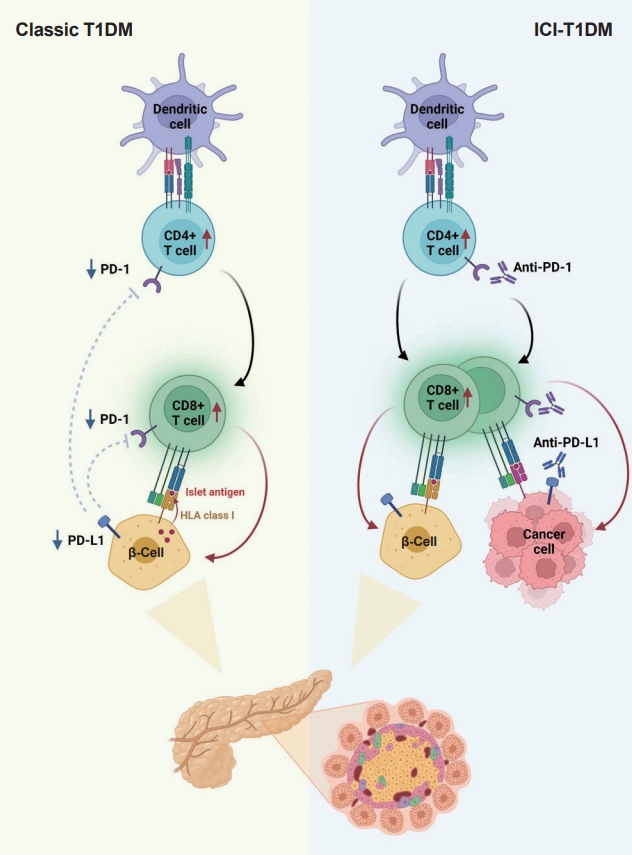
ePub - With the increasing use of immune-checkpoint inhibitors (ICIs), such as anti-cytotoxic T lymphocyte-associated antigen 4 (CTLA-4) and anti-programmed cell death-1 (PD-1), for the treatment of malignancies, cases of ICI-induced type 1 diabetes mellitus (ICI-T1DM) have been reported globally. This review focuses on the features and pathogenesis of this disease. T1DM is an immune-related adverse event that occurs following the administration of anti-PD-1 or anti-programmed death ligand-1 (PDL1) alone or in combination with anti-CTLA-4. More than half of the reported cases presented as abrupt-onset diabetic ketoacidosis. The primary mechanism of ICI-T1DM is T-cell stimulation, which results from the loss of interaction between PD-1 and PD-L1 in pancreatic islet. The similarities and differences between ICI-T1DM and classical T1DM may provide insights into this disease entity. ICI-T1DM is a rare but often life-threatening medical emergency that healthcare professionals and patients need to be aware of. Early detection of and screening for this disease is imperative. At present, the only known treatment for ICI-T1DM is insulin injection. Further research into the mechanisms and risk factors associated with ICI-T1DM development may contribute to a better understanding of this disease entity and the identification of possible preventive strategies.
-
Citations
Citations to this article as recorded by- Research Advances of Immune Checkpoint Inhibitors Related Endocrine Adverse Events
晶晶 王
Advances in Clinical Medicine.2024; 14(02): 2706. CrossRef - Immune checkpoint inhibitor‑associated diabetes mellitus in patients with HCC: Report of three cases and literature review
Gaocheng Wang, Jingjing Wang, Shuilin Dong, Zhanguo Zhang, Wanguang Zhang, Jianping Zhao
Experimental and Therapeutic Medicine.2024;[Epub] CrossRef
- Research Advances of Immune Checkpoint Inhibitors Related Endocrine Adverse Events

 KDA
KDA
 First
First Prev
Prev





