
- Current
- Browse
- Collections
-
For contributors
- For Authors
- Instructions to authors
- Article processing charge
- e-submission
- For Reviewers
- Instructions for reviewers
- How to become a reviewer
- Best reviewers
- For Readers
- Readership
- Subscription
- Permission guidelines
- About
- Editorial policy
Search
- Page Path
- HOME > Search
- COVID-19
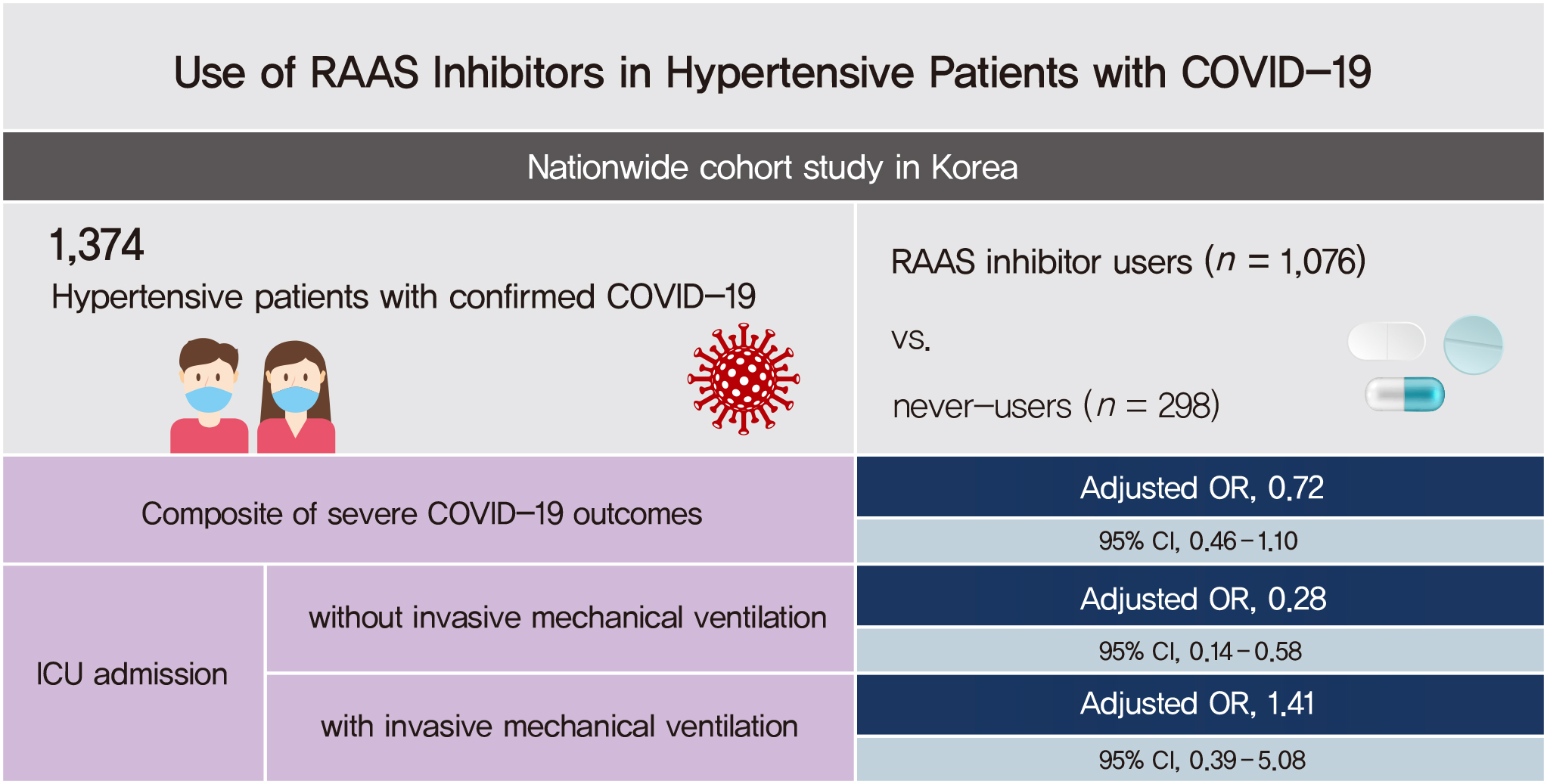
- Use of Renin-Angiotensin-Aldosterone System Inhibitors and Severe COVID-19 Outcomes in Patients with Hypertension: A Nationwide Cohort Study
- Jae Hyun Bae, Sun Kyu Choi, Nam Hoon Kim, Juneyoung Lee, Sin Gon Kim
- Diabetes Metab J. 2021;45(3):430-438. Published online February 22, 2021
- DOI: https://doi.org/10.4093/dmj.2020.0279
- 7,950 View
- 296 Download
- 3 Crossref
-
 Graphical Abstract
Graphical Abstract
 Abstract
Abstract
 PDF
PDF Supplementary Material
Supplementary Material PubReader
PubReader  ePub
ePub 
- Background
Angiotensin-converting enzyme 2 facilitates the entry of severe acute respiratory syndrome coronavirus 2 into the human body. We investigated the association of renin-angiotensin-aldosterone system (RAAS) inhibitor use with severe coronavirus disease 2019 (COVID-19) outcomes in hypertensive patients.
Methods
We identified hypertensive patients with confirmed COVID-19 from the Korean Health Insurance Review and Assessment Service from inception to May 15, 2020. The primary outcome was the composite of intensive care unit (ICU) admission, invasive mechanical ventilation (IMV), continuous renal replacement therapy (CRRT), extracorporeal membrane oxygenation (ECMO), and death from COVID-19. The individual components were evaluated as secondary outcomes.
Results
Of 1,374 hypertensive patients with COVID-19, 1,076 (78.3%) and 298 (21.7%) were users and never-users of RAAS inhibitors, respectively. The RAAS inhibitor users were not associated with the risk of the primary outcome (adjusted odds ratio [aOR], 0.72; 95% confidence interval [CI], 0.46 to 1.10). The risk of ICU admission was significantly lower in the users than the never-users (aOR, 0.44; 95% CI, 0.24 to 0.84). The RAAS inhibitors were beneficial only in ICU admissions that did not require IMV (aOR, 0.28; 95% CI, 0.14 to 0.58). The risk of death from COVID-19 was comparable between the groups (aOR, 1.09; 95% CI, 0.64 to 1.85). We could not evaluate the risks of CRRT and ECMO owing to the small number of events.
Conclusion
RAAS inhibitor use was not associated with the composite of severe outcomes in the hypertensive patients with COVID-19 but significantly lowered the risk of ICU admission, particularly in patients who did not require IMV. -
Citations
Citations to this article as recorded by- Systematic review and meta-analysis of the clinical outcomes of ACEI/ARB in East-Asian patients with COVID-19
Nancy Xurui Huang, Qi Yuan, Fang Fang, Bryan P. Yan, John E. Sanderson, Masaki Mogi
PLOS ONE.2023; 18(1): e0280280. CrossRef - Renin‐Angiotensin Aldosterone System Inhibitors and COVID‐19: A Systematic Review and Meta‐Analysis Revealing Critical Bias Across a Body of Observational Research
Jordan Loader, Frances C. Taylor, Erik Lampa, Johan Sundström
Journal of the American Heart Association.2022;[Epub] CrossRef - Renin-angiotensin-aldosterone system blockers in Bulgarian COVID-19 patients with or without chronic kidney disease
Rumen Filev, Lionel Rostaing, Mila Lyubomirova, Boris Bogov, Krassimir Kalinov, Dobrin Svinarov
Medicine.2022; 101(48): e31988. CrossRef
- Systematic review and meta-analysis of the clinical outcomes of ACEI/ARB in East-Asian patients with COVID-19
- Clinical Diabetes & Therapeutics
-
- Asian Subpopulations May Exhibit Greater Cardiovascular Benefit from Long-Acting Glucagon-Like Peptide 1 Receptor Agonists: A Meta-Analysis of Cardiovascular Outcome Trials
- Yu Mi Kang, Yun Kyung Cho, Jiwoo Lee, Seung Eun Lee, Woo Je Lee, Joong-Yeol Park, Ye-Jee Kim, Chang Hee Jung, Michael A. Nauck
- Diabetes Metab J. 2019;43(4):410-421. Published online December 27, 2018
- DOI: https://doi.org/10.4093/dmj.2018.0070
- 6,350 View
- 137 Download
- 18 Web of Science
- 18 Crossref
-
 Abstract
Abstract
 PDF
PDF Supplementary Material
Supplementary Material PubReader
PubReader Background Based on reported results of three large cardiovascular outcome trials (CVOTs) of glucagon-like peptide 1 receptor agonists (GLP-1 RAs), we aimed to investigate the overall effect of GLP-1 RAs on major adverse cardiovascular events (MACEs) and to identify subpopulations exhibiting the greatest cardiovascular (CV) benefit.
Methods Three CVOTs reporting effects of long-acting GLP-1 RAs were included: LEADER (liraglutide), SUSTAIN-6 (semaglutide), and EXSCEL (exenatide once weekly). In all studies, the primary endpoint was three-point MACE, comprising CV death, non-fatal myocardial infarction, and non-fatal stroke. Overall effect estimates were calculated as hazard ratios and 95% confidence intervals (CIs) using the random-effects model; subgroup analyses reported in the original studies were similarly analyzed.
Results Overall, statistically significant risk reductions in MACE and CV death were observed. Subgroup analysis indicated a significant racial difference with respect to CV benefit (
P for interaction <0.001), and more substantial risk reductions were observed in subjects of African origin (relative risk [RR], 0.78; 95% CI, 0.60 to 0.99) and in Asians (RR, 0.35; 95% CI, 0.09 to 1.32). However,post hoc analysis (Bonferroni method) revealed that only Asians exhibited a significantly greater CV benefit from treatment, compared with white subjects (P <0.0001).Conclusion Long-acting GLP-1 RAs reduced risks of MACE and CV deaths in high-risk patients with type 2 diabetes mellitus. Our findings of a particularly effective reduction in CV events with GLP-1 RA in Asian populations merits further exploration and dedicated trials in specific populations.
-
Citations
Citations to this article as recorded by- Sex, racial, ethnic, and geographical disparities in major adverse cardiovascular outcome of glucagon-like peptide-1 receptor agonists among patients with and without diabetes mellitus: A meta-analysis of placebo-controlled randomized controlled trials,
Frederick Berro Rivera, Nathan Ross B. Bantayan, John Paul Aparece, Linnaeus Louisse A. Cruz, John Vincent Magallong, Polyn Luz Pine, Anne Mira Nicca Idian-Javier, Grace Nooriza O. Lumbang, Edgar V. Lerma, Kyla M. Lara-Breitinger, Martha Gulati, Krishnasw
Journal of Clinical Lipidology.2024;[Epub] CrossRef - Coronary Artery Disease in South Asian Patients: Cardiovascular Risk Factors, Pathogenesis and Treatments
Vincenzo Sucato, Giuseppe Coppola, Girolamo Manno, Giuseppe Vadalà, Giuseppina Novo, Egle Corrado, Alfredo Ruggero Galassi
Current Problems in Cardiology.2023; 48(8): 101228. CrossRef - Retrospective Analysis of the Effectiveness of Oral Semaglutide in Type 2 Diabetes Mellitus and Its Effect on Cardiometabolic Parameters in Japanese Clinical Settings
Hodaka Yamada, Masashi Yoshida, Shunsuke Funazaki, Jun Morimoto, Shiori Tonezawa, Asuka Takahashi, Shuichi Nagashima, Kimura Masahiko, Otsuka Kiyoshi, Kazuo Hara
Journal of Cardiovascular Development and Disease.2023; 10(4): 176. CrossRef - Efficacy of treatment with glucagon-like peptide receptor agonists-1 in Asian patients with type 2 diabetes mellitus
L. Yu. Khamnueva, L. S. Andreeva
Problems of Endocrinology.2023; 69(2): 38. CrossRef - Role of diabetes in stroke: Recent advances in pathophysiology and clinical management
Sian A. Bradley, Kevin J. Spring, Roy G. Beran, Dimitrios Chatzis, Murray C. Killingsworth, Sonu M. M. Bhaskar
Diabetes/Metabolism Research and Reviews.2022;[Epub] CrossRef - Obesity Pillars Roundtable: Obesity and East Asians
Harold Edward Bays, Jennifer Ng, Jeffrey Sicat, Michelle Look
Obesity Pillars.2022; 2: 100011. CrossRef - Pathophysiology, phenotypes and management of type 2 diabetes mellitus in Indian and Chinese populations
Calvin Ke, K. M. Venkat Narayan, Juliana C. N. Chan, Prabhat Jha, Baiju R. Shah
Nature Reviews Endocrinology.2022; 18(7): 413. CrossRef - Effect of race on cardiometabolic responses to once-weekly exenatide: insights from the Exenatide Study of Cardiovascular Event Lowering (EXSCEL)
Timothy M. E. Davis, Anna Giczewska, Yuliya Lokhnygina, Robert J. Mentz, Naveed Sattar, Rury R. Holman
Cardiovascular Diabetology.2022;[Epub] CrossRef - Generalizability of the Results of Cardiovascular Outcome Trials of Glucagon-Like Peptide 1 Receptor Agonists in Chinese Patients with Type 2 Diabetes Mellitus
Xiaoling Cai, Linong Ji
Diabetes Therapy.2021; 12(7): 1861. CrossRef - Current trends in epidemiology of cardiovascular disease and cardiovascular risk management in type 2 diabetes
Jae-Seung Yun, Seung-Hyun Ko
Metabolism.2021; 123: 154838. CrossRef - Sex and ethnic differences in the cardiovascular complications of type 2 diabetes
Jian L. Yeo, Emer M. Brady, Gerry P. McCann, Gaurav S. Gulsin
Therapeutic Advances in Endocrinology and Metabolism.2021; 12: 204201882110342. CrossRef - Efficacy and Safety of GLP-1 Receptor Agonists in Patients With Type 2 Diabetes Mellitus and Non-Alcoholic Fatty Liver Disease: A Systematic Review and Meta-Analysis
Yuan Zhu, Jiao Xu, Dong Zhang, Xingyu Mu, Yi Shi, Shangtao Chen, Zengxiang Wu, Shuangqing Li
Frontiers in Endocrinology.2021;[Epub] CrossRef - Efficacy and safety of dulaglutide in type 2 diabetes patients in endocrinology clinics of Islamabad, Pakistan
Matiullah Kamin, SajjadAli Khan, UmarYousaf Raja, Osama Ishtiaq, Asmara Malik, Tejhmal Rehman, MuhammadUmar Wahab
Indian Journal of Endocrinology and Metabolism.2021; 25(5): 456. CrossRef - Type 2 Diabetes and Atherosclerotic Cardiovascular Disease in South Asians: a Unique Population with a Growing Challenge
Afreen I. Shariff, Nitya Kumar, William S. Yancy, Leonor Corsino
Current Diabetes Reports.2020;[Epub] CrossRef - Antihypertensive and Renal Mechanisms of SGLT2 (Sodium-Glucose Linked Transporter 2) Inhibitors
Christopher S. Wilcox
Hypertension.2020; 75(4): 894. CrossRef - Subpopulation Differences in the Cardiovascular Efficacy of Long-Acting Glucagon-Like Peptide 1 Receptor Agonists in Type 2 Diabetes Mellitus: A Systematic Review and Meta-analysis
Liyun He, Na Yang, Lingling Xu, Fan Ping, Wei Li, Yuxiu Li, Huabing Zhang
Diabetes Therapy.2020; 11(9): 2121. CrossRef - 2020 Consensus of Taiwan Society of Cardiology on the pharmacological management of patients with type 2 diabetes and cardiovascular diseases
Chern-En Chiang, Kwo-Chang Ueng, Ting-Hsing Chao, Tsung-Hsien Lin, Yih-Jer Wu, Kang-Ling Wang, Shih-Hsien Sung, Hung-I Yeh, Yi-Heng Li, Ping-Yen Liu, Kuan-Cheng Chang, Kou-Gi Shyu, Jin-Long Huang, Cheng-Dao Tsai, Huei-Fong Hung, Ming-En Liu, Tze-Fan Chao,
Journal of the Chinese Medical Association.2020; 83(7): 587. CrossRef - Beneficial effect of anti-diabetic drugs for nonalcoholic fatty liver disease
Kyung-Soo Kim, Byung-Wan Lee
Clinical and Molecular Hepatology.2020; 26(4): 430. CrossRef
- Sex, racial, ethnic, and geographical disparities in major adverse cardiovascular outcome of glucagon-like peptide-1 receptor agonists among patients with and without diabetes mellitus: A meta-analysis of placebo-controlled randomized controlled trials,
- Clinical Diabetes & Therapeutics
- Glucagon-Like Peptide-1 Receptor Agonists for the Treatment of Type 2 Diabetes Mellitus: A Position Statement of the Korean Diabetes Association
- Hyun Jin Kim, Seok O Park, Seung-Hyun Ko, Sang Youl Rhee, Kyu-Yeon Hur, Nan-Hee Kim, Min Kyong Moon, Byung-Wan Lee, Jin Hwa Kim, Kyung Mook Choi
- Diabetes Metab J. 2017;41(6):423-429. Published online December 19, 2017
- DOI: https://doi.org/10.4093/dmj.2017.41.6.423

- 5,813 View
- 71 Download
- 5 Web of Science
- 6 Crossref
-
 Abstract
Abstract
 PDF
PDF PubReader
PubReader The glucagon-like peptide-1 receptor agonists (GLP-1RAs) were recommended as a monotherapy or combination therapy with oral hypoglycemic agents or basal insulin in the position statement of the Korean Diabetes Association 2017 for pharmacological therapy. Many randomized clinical trials and systematic reviews report that GLP-1RAs have considerable glucose-lowering effect and lead to weight reduction and low risk of hypoglycemia when used as a monotherapy or combination therapy. The cardiovascular safety of GLP-1RAs has been assessed in several randomized clinical trials and systematic reviews. The results of cardiovascular outcome trials of long-acting GLP-1RAs (liraglutide, semaglutide) demonstrated cardiovascular benefits in subjects with type 2 diabetes mellitus and a high risk of cardiovascular disease. The GLP-1RA may be a choice of therapy when weight control and avoidance of hypoglycemia are important, and patients with high risk of cardiovascular disease might also favor choosing GLP-1RA.
-
Citations
Citations to this article as recorded by- Anti-inflammatory effect of glucagon-like Peptide-1 receptor agonist on the neurosensory retina in an acute optic nerve injury rat model
Yeon Woong Chung, Ji Young Lee, Hyun Hee Ju, Jin A. Choi
European Journal of Pharmacology.2022; 933: 175269. CrossRef - Diabetes Risk Data Mining Method Based on Electronic Medical Record Analysis
Yang Liu, Zhaoxiang Yu, Yunlong Yang, Zhihan Lv
Journal of Healthcare Engineering.2021; 2021: 1. CrossRef - Paradigm Shift for the Treatment of Type 2 Diabetes Mellitus in Patients with Cardiovascular Disease: Cardiologist's Perspective
Doo Soo Jeon
Cardiovascular Prevention and Pharmacotherapy.2020; 2(1): 11. CrossRef - The Role of Glucagon-Like Peptide-1 Receptor Agonists in Type 2 Diabetes in Asia
Ju-Ming Lu
Advances in Therapy.2019; 36(4): 798. CrossRef - A Review of Practical Issues on the Use of Glucagon-Like Peptide-1 Receptor Agonists for the Management of Type 2 Diabetes
Irene Romera, Ana Cebrián-Cuenca, Fernando Álvarez-Guisasola, Fernando Gomez-Peralta, Jesús Reviriego
Diabetes Therapy.2019; 10(1): 5. CrossRef - Glucagon-Like Peptide-1 Receptor Agonists for the Treatment of Type 2 Diabetes Mellitus: A Position Statement of the Korean Diabetes Association
Hyun Jin Kim
The Journal of Korean Diabetes.2018; 19(1): 35. CrossRef
- Anti-inflammatory effect of glucagon-like Peptide-1 receptor agonist on the neurosensory retina in an acute optic nerve injury rat model
- Islet Studies and Transplantation
- An Update on the Effect of Incretin-Based Therapies on β-Cell Function and Mass
- Suk Chon, Jean-François Gautier
- Diabetes Metab J. 2016;40(2):99-114. Published online April 25, 2016
- DOI: https://doi.org/10.4093/dmj.2016.40.2.99
- 4,975 View
- 111 Download
- 43 Web of Science
- 41 Crossref
-
 Abstract
Abstract
 PDF
PDF PubReader
PubReader Type 2 diabetes mellitus (T2DM) is a multifactorial disease with a complex and progressive pathogenesis. The two primary mechanisms of T2DM pathogenesis are pancreatic β-cell dysfunction and insulin resistance. Pancreatic β-cell dysfunction is recognized to be a prerequisite for the development of T2DM. Therapeutic modalities that improve β-cell function are considered critical to T2DM management; however, blood glucose control remains a challenge for many patients due to suboptimal treatment efficacy and the progressive nature of T2DM. Incretin-based therapies are now the most frequently prescribed antidiabetic drugs in Korea. Incretin-based therapies are a favorable class of drugs due to their ability to reduce blood glucose by targeting the incretin hormone system and, most notably, their potential to improve pancreatic β-cell function. This review outlines the current understanding of the incretin hormone system in T2DM and summarizes recent updates on the effect of incretin-based therapies on β-cell function and β-cell mass in animals and humans.
-
Citations
Citations to this article as recorded by- Harnessing gut cells for functional insulin production: Strategies and challenges
Kelvin Baafi, John C. March
Biotechnology Notes.2023; 4: 7. CrossRef - Incretin and Pancreatic β-Cell Function in Patients with Type 2 Diabetes
Chang Ho Ahn, Tae Jung Oh, Se Hee Min, Young Min Cho
Endocrinology and Metabolism.2023; 38(1): 1. CrossRef - Weight loss maintenance with exercise and liraglutide improves glucose tolerance, glucagon response, and beta cell function
Simon B. K. Jensen, Christian R. Juhl, Charlotte Janus, Julie R. Lundgren, Christoffer Martinussen, Christoffer Wiingaard, Cecilie Knudsen, Ruth Frikke‐Schmidt, Bente M. Stallknecht, Jens J. Holst, Sten Madsbad, Signe S. Torekov
Obesity.2023; 31(4): 977. CrossRef - How do parasitic worms prevent diabetes? An exploration of their influence on macrophage and β-cell crosstalk
Inah Camaya, Bronwyn O’Brien, Sheila Donnelly
Frontiers in Endocrinology.2023;[Epub] CrossRef - An Imbalance of Pathophysiologic Factors in Late Postprandial Hypoglycemia Post Bariatric Surgery: A Narrative Review
Marah Alsayed Hasan, Stanley Schwartz, Victoria McKenna, Richard Ing
Obesity Surgery.2023; 33(9): 2927. CrossRef - Therapeutic Dilemma in Personalized Medicine
Ehab S. EL Desoky
Current Reviews in Clinical and Experimental Pharmacology.2022; 17(2): 94. CrossRef - Lessons from neonatal β-cell epigenomic for diabetes prevention and treatment
Amar Abderrahmani, Cécile Jacovetti, Romano Regazzi
Trends in Endocrinology & Metabolism.2022; 33(6): 378. CrossRef - Beneficial metabolic effects of recurrent periods of beta‐cell rest and stimulation using stable neuropeptide Y1 and glucagon‐like peptide‐1 receptor agonists
Neil Tanday, Ryan A. Lafferty, Peter R. Flatt, Nigel Irwin
Diabetes, Obesity and Metabolism.2022; 24(12): 2353. CrossRef - A Randomized Controlled Trial of R-Form Verapamil Added to Ongoing Metformin Therapy in Patients with Type 2 Diabetes
Chih-Yuan Wang, Kuo-Chin Huang, Chia-Wen Lu, Chih-Hsun Chu, Chien-Ning Huang, Harn-Shen Chen, I-Te Lee, Jung-Fu Chen, Ching-Chu Chen, Chung-Sen Chen, Chang-Hsun Hsieh, Kai-Jen Tien, Hung-Yu Chien, Yu-Yao Huang, Jui-Pao Hsu, Guang-Tzuu Shane, Ai-Ching Chan
The Journal of Clinical Endocrinology & Metabolism.2022; 107(10): e4063. CrossRef - Exenatide, Metformin, or Both for Prediabetes in PCOS: A Randomized, Open-label, Parallel-group Controlled Study
Tao Tao, Yi Zhang, Yu-Chen Zhu, Jia-Rong Fu, Yu-Ying Wang, Jie Cai, Jing-Yu Ma, Yu Xu, Yi-Ning Gao, Yun Sun, WuQiang Fan, Wei Liu
The Journal of Clinical Endocrinology & Metabolism.2021; 106(3): e1420. CrossRef - The utility of assessing C-peptide in patients with insulin-treated type 2 diabetes: a cross-sectional study
Tuccinardi Dario, Giorgino Riccardo, Pieralice Silvia, Watanabe Mikiko, Maggi Daria, Palermo Andrea, Defeudis Giuseppe, Fioriti Elvira, Pozzilli Paolo, Manfrini Silvia
Acta Diabetologica.2021; 58(4): 411. CrossRef - Type 2 diabetes: evidence-based medicine approach to glucose-lowering therapy
E. V. Biryukova, I. A. Morozova, S. V. Rodionova
Meditsinskiy sovet = Medical Council.2021; (21): 160. CrossRef - Emerging Role of Caveolin-1 in GLP-1 Action
Alessandra Puddu, Davide Maggi
Frontiers in Endocrinology.2021;[Epub] CrossRef - Improvements in HOMA indices and pancreatic endocrinal tissues in type 2-diabetic rats by DPP-4 inhibition and antioxidant potential of an ethanol fruit extract of Withania coagulans
Heera Ram, Pramod Kumar, Ashok Purohit, Priya Kashyap, Suresh Kumar, Shivani Kumar, Garima Singh, Abdulaziz A. Alqarawi, Abeer Hashem, Elsayed Fathi Abd-Allah, Al-Bandari Fahad Al-Arjani, Bhim Pratap Singh
Nutrition & Metabolism.2021;[Epub] CrossRef -
Chop
/
Ddit3
depletion in β cells alleviates ER stress and corrects hepatic steatosis in mice
Jing Yong, Vishal S. Parekh, Shannon M. Reilly, Jonamani Nayak, Zhouji Chen, Cynthia Lebeaupin, Insook Jang, Jiangwei Zhang, Thazha P. Prakash, Hong Sun, Sue Murray, Shuling Guo, Julio E. Ayala, Leslie S. Satin, Alan R. Saltiel, Randal J. Kaufman
Science Translational Medicine.2021;[Epub] CrossRef - Assessment of Insulin Secretion and Insulin Resistance in Human
So Young Park, Jean-François Gautier, Suk Chon
Diabetes & Metabolism Journal.2021; 45(5): 641. CrossRef - Targeted pharmacological therapy restores β-cell function for diabetes remission
Stephan Sachs, Aimée Bastidas-Ponce, Sophie Tritschler, Mostafa Bakhti, Anika Böttcher, Miguel A. Sánchez-Garrido, Marta Tarquis-Medina, Maximilian Kleinert, Katrin Fischer, Sigrid Jall, Alexandra Harger, Erik Bader, Sara Roscioni, Siegfried Ussar, Annett
Nature Metabolism.2020; 2(2): 192. CrossRef - The Relationship Between Timing of Initiation on a Glucagon-like Peptide-1 Receptor Agonist and Glycosylated Hemoglobin Values Among Patients With Type 2 Diabetes
Kristina S. Boye, Reema Mody, Maureen J. Lage, Raleigh E. Malik
Clinical Therapeutics.2020; 42(9): 1812. CrossRef - Short-term renal and metabolic effects of low dose vildagliptin treatment added-on insulin therapy in non-proteinuric patients with type 2 diabetes: open-label randomized prospective study
Valentina K. Bayrasheva, Ivan Y. Pchelin, Vladimir A. Dobronravov, Alina Yu. Babenko, Svetlana G. Chefu, Ivan S. Shatalov, Volha N. Vasilkova, Natalia V. Hudiakova, Alexandra N. Ivanova, Pavel A. Andoskin, Elena N. Grineva
Archives of Endocrinology and Metabolism.2020;[Epub] CrossRef - A Phenotypic Screen Identifies Calcium Overload as a Key Mechanism of β-Cell Glucolipotoxicity
Jennifer Vogel, Jianning Yin, Liansheng Su, Sharon X. Wang, Richard Zessis, Sena Fowler, Chun-Hao Chiu, Aaron C. Wilson, Amy Chen, Frederic Zecri, Gordon Turner, Thomas M. Smith, Brian DeChristopher, Heming Xing, Deborah M. Rothman, Xinming Cai, Alina Ber
Diabetes.2020; 69(5): 1032. CrossRef - Neuropeptide 26RFa (QRFP) is a key regulator of glucose homeostasis and its activity is markedly altered in obese/hyperglycemic mice
Gaëtan Prévost, Arnaud Arabo, Marie-Anne Le Solliec, Justine Bons, Marie Picot, Julie Maucotel, Hind Berrahmoune, Mouna El Mehdi, Saloua Cherifi, Alexandre Benani, Emmanuelle Nédélec, Moïse Coëffier, Jérôme Leprince, Anneli Nordqvist, Valéry Brunel, Pierr
American Journal of Physiology-Endocrinology and Metabolism.2019; 317(1): E147. CrossRef - Gastrin analogue administration adds no significant glycaemic benefit to a glucagon‐like peptide‐1 receptor agonist acutely or after washout of both analogues
Krister Bokvist, Ying Ding, William H. Landschulz, Vikram Sinha, Aleksandra Pastrak, Ruth M. Belin
Diabetes, Obesity and Metabolism.2019; 21(7): 1606. CrossRef - Effects of boschnaloside from Boschniakia rossica on dysglycemia and islet dysfunction in severely diabetic mice through modulating the action of glucagon-like peptide-1
Lie-Chwen Lin, Lin-Chien Lee, Cheng Huang, Chiung-Tong Chen, Jen-Shin Song, Young-Ji Shiao, Hui-Kang Liu
Phytomedicine.2019; 62: 152946. CrossRef - The future of new drugs for diabetes management
Clifford J. Bailey, Caroline Day
Diabetes Research and Clinical Practice.2019; 155: 107785. CrossRef - Compact fluidic system for functional assessment of pancreatic islets
Takeshi Hori, Kei Yamane, Takayuki Anazawa, Osamu Kurosawa, Hiroo Iwata
Biomedical Microdevices.2019;[Epub] CrossRef - Pharmacokinetics of Exenatide in nonhuman primates following its administration in the form of sustained-release PT320 and Bydureon
Yazhou Li, Kelli L. Vaughan, David Tweedie, Jin Jung, Hee Kyung Kim, Ho-Il Choi, Dong Seok Kim, Julie A. Mattison, Nigel H. Greig
Scientific Reports.2019;[Epub] CrossRef - Pharmacokinetics and efficacy of PT302, a sustained-release Exenatide formulation, in a murine model of mild traumatic brain injury
Miaad Bader, Yazhou Li, Daniela Lecca, Vardit Rubovitch, David Tweedie, Elliot Glotfelty, Lital Rachmany, Hee Kyung Kim, Ho-Il Choi, Barry J. Hoffer, Chaim G. Pick, Nigel H. Greig, Dong Seok Kim
Neurobiology of Disease.2019; 124: 439. CrossRef - Novel dual incretin agonist peptide with antidiabetic and neuroprotective potential
N.M. Pathak, V. Pathak, V.A. Gault, S. McClean, N. Irwin, P.R. Flatt
Biochemical Pharmacology.2018; 155: 264. CrossRef - Human EndoC-βH1 β-cells form pseudoislets with improved glucose sensitivity and enhanced GLP-1 signaling in the presence of islet-derived endothelial cells
Michael G. Spelios, Lauren A. Afinowicz, Regine C. Tipon, Eitan M. Akirav
American Journal of Physiology-Endocrinology and Metabolism.2018; 314(5): E512. CrossRef - Vildagliptin: ten years in the service for type 2 diabetes mellitus patients. The journey of discovery, innovation and success in clinical practice
Tatiana Yu. Demidova
Problems of Endocrinology.2018; 64(5): 336. CrossRef - Thromboxane-Dependent Platelet Activation in Obese Subjects with Prediabetes or Early Type 2 Diabetes: Effects of Liraglutide- or Lifestyle Changes-Induced Weight Loss
Paola Simeone, Rossella Liani, Romina Tripaldi, Augusto Di Castelnuovo, Maria Guagnano, Armando Tartaro, Riccardo Bonadonna, Virginia Federico, Francesco Cipollone, Agostino Consoli, Francesca Santilli
Nutrients.2018; 10(12): 1872. CrossRef - Nutrient regulation of β-cell function: what do islet cell/animal studies tell us?
R Carlessi, K N Keane, C Mamotte, P Newsholme
European Journal of Clinical Nutrition.2017; 71(7): 890. CrossRef - Effectiveness and safety of exenatide in Korean patients with type 2 diabetes inadequately controlled with oral hypoglycemic agents: an observational study in a real clinical practice
You-Cheol Hwang, Ari Kim, Euna Jo, Yeoree Yang, Jae-Hyoung Cho, Byung-Wan Lee
BMC Endocrine Disorders.2017;[Epub] CrossRef - Efficacy and safety of adding evogliptin versus sitagliptin for metformin‐treated patients with type 2 diabetes: A 24‐week randomized, controlled trial with open label extension
Sang‐Mo Hong, Cheol‐Young Park, Dong‐Min Hwang, Kyung Ah Han, Chang Beom Lee, Choon Hee Chung, Kun‐Ho Yoon, Ji‐Oh Mok, Kyong Soo Park, Sung‐Woo Park
Diabetes, Obesity and Metabolism.2017; 19(5): 654. CrossRef - The effects of vildagliptin compared with metformin on vascular endothelial function and metabolic parameters: a randomized, controlled trial (Sapporo Athero-Incretin Study 3)
Naoyuki Kitao, Hideaki Miyoshi, Tomoo Furumoto, Kota Ono, Hiroshi Nomoto, Aika Miya, Chiho Yamamoto, Atsushi Inoue, Kenichi Tsuchida, Naoki Manda, Yoshio Kurihara, Shin Aoki, Akinobu Nakamura, Tatsuya Atsumi
Cardiovascular Diabetology.2017;[Epub] CrossRef - Recent Advances in Effect‐directed Enzyme Assays based on Thin‐layer Chromatography
Sarah Bräm, Evelyn Wolfram
Phytochemical Analysis.2017; 28(2): 74. CrossRef - Efficacy and safety of gemigliptin, a dipeptidyl peptidase‐4 inhibitor, in patients with type 2 diabetes mellitus inadequately controlled with combination treatment of metformin and sulphonylurea: a 24‐week, multicentre, randomized, double‐blind, placebo‐
Chang Ho Ahn, Kyung Ah Han, Jae Myung Yu, Joo Young Nam, Kyu Jeung Ahn, Tae Keun Oh, Hyoung Woo Lee, Dae Ho Lee, Jaetaek Kim, Choon Hee Chung, Tae Sun Park, Byung Joon Kim, Seok Won Park, Hyeong Kyu Park, Kwang Jae Lee, Sang‐Wook Kim, Jeong Hyun Park, Kwa
Diabetes, Obesity and Metabolism.2017; 19(5): 635. CrossRef - Antihyperglycemic agent therapy for adult patients with type 2 diabetes mellitus 2017: a position statement of the Korean Diabetes Association
Seung-Hyun Ko, Kyu-Yeon Hur, Sang Youl Rhee, Nan-Hee Kim, Min Kyong Moon, Seok-O Park, Byung-Wan Lee, Hyun Jin Kim, Kyung Mook Choi, Jin Hwa Kim
The Korean Journal of Internal Medicine.2017; 32(6): 947. CrossRef - Antihyperglycemic Agent Therapy for Adult Patients with Type 2 Diabetes Mellitus 2017: A Position Statement of the Korean Diabetes Association
Seung-Hyun Ko, Kyu-Yeon Hur, Sang Youl Rhee, Nan-Hee Kim, Min Kyong Moon, Seok-O Park, Byung-Wan Lee, Hyun Jin Kim, Kyung Mook Choi, Jin Hwa Kim
Diabetes & Metabolism Journal.2017; 41(5): 337. CrossRef - DPP-4 inhibitors in diabetic complications: role of DPP-4 beyond glucose control
Eun Ju Bae
Archives of Pharmacal Research.2016; 39(8): 1114. CrossRef - Liraglutide Enhances the Efficacy of Human Mesenchymal Stem Cells in Preserving Islet ß-cell Function in Severe Non-obese Diabetic Mice
Li-rong Li, Jing Lu, Xiao-lei Jia, Hui Hui, Jie Zhang, Ying Liu, Wei-juan Cui, Qian-yue Xu, Da-long Zhu
Molecular Medicine.2016; 22(1): 800. CrossRef
- Harnessing gut cells for functional insulin production: Strategies and challenges
- Obesity and Metabolic Syndrome
- Perspective of Small-Molecule AdipoR Agonist for Type 2 Diabetes and Short Life in Obesity
- Miki Okada-Iwabu, Masato Iwabu, Kohjiro Ueki, Toshimasa Yamauchi, Takashi Kadowaki
- Diabetes Metab J. 2015;39(5):363-372. Published online October 22, 2015
- DOI: https://doi.org/10.4093/dmj.2015.39.5.363
- 6,810 View
- 61 Download
- 39 Web of Science
- 43 Crossref
-
 Abstract
Abstract
 PDF
PDF PubReader
PubReader Obesity associated with unhealthy diet and lack of exercise is shown to contribute to the onset and/or aggravation of the metabolic syndrome and diabetes, thus placing affected individuals at increased risk of cardiovascular disease and cancer. Plasma adiponectin levels are decreased in obesity, which causes insulin resistance and diabetes. Therefore, we identified adiponectin receptors (AdipoRs) as the therapeutic target. It was suggested that, similarly to caloric restriction and exercise, activation of the AdipoRs may have the potential not only to improve lifestyle-related diseases but to contribute to prolonged the shortened lifespan on a high caloric unhealthy diet. To this end, we have identified "AdipoRon" as an adiponectin receptor agonist. Indeed, AdipoRon ameliorated diabetes associated with obesity as well as to increase exercise endurance, thus prolonging shortened lifespan of obese mice fed on a high fat diet. Additionally, we have recently determined the crystal structures of the human AdipoRs. The seven-transmembrane helices of AdipoRs are structurally distinct from those of G-protein coupled receptors. It is expected that these findings will contribute not only to the elucidation of the AdipoR-related signal transduction but to the development and optimization of AdipoR-targeted therapeutics for obesity-related diseases such as diabetes.
-
Citations
Citations to this article as recorded by- Identification of a novel adiponectin receptor and opioid receptor dual acting agonist as a potential treatment for diabetic neuropathy
Oscar Ka-Fai Ma, Simone Ronsisvalle, Livia Basile, Ariya Weiman Xiang, Cristina Tomasella, Federica Sipala, Matteo Pappalardo, Koon-Ho Chan, Danilo Milardi, Roy Chun-Laam Ng, Salvatore Guccione
Biomedicine & Pharmacotherapy.2023; 158: 114141. CrossRef - AdipoRon Effect on Expression of Lipid Metabolism Genes in Cultured Human Primary Macrophages
I. A. Pobozheva, K. V. Dracheva, S. N. Pchelina, V. V. Miroshnikova
Molecular Biology.2023; 57(4): 616. CrossRef - The Antiviral Potential of AdipoRon, an Adiponectin Receptor Agonist, Reveals the Ability of Zika Virus to Deregulate Adiponectin Receptor Expression
Daed El Safadi, Grégorie Lebeau, Jonathan Turpin, Christian Lefebvre d’Hellencourt, Nicolas Diotel, Wildriss Viranaicken, Pascale Krejbich-Trotot
Viruses.2023; 16(1): 24. CrossRef - The Effect of Adiporon on Lipid Metabolism Genes Expression in Human Macrophages
I. A. Pobozheva, K. V. Dracheva, S. N. Pchelina, V. V. Miroshnikova
Молекулярная биология.2023; 57(4): 623. CrossRef - The effects of high intensity-interval training on vaspin, adiponectin and leptin levels in women with polycystic ovary syndrome
H. Ş. Aktaş, Y. E. Uzun, O. Kutlu, H. H. Pençe, F. Özçelik, E. Ö. Çil, L. Irak, Ö. Altun, M. Özcan, N. Özsoy, Ş. Aydın Yoldemir, S. Kalyon, Y. Arman, T. Tükek
Archives of Physiology and Biochemistry.2022; 128(1): 37. CrossRef - G protein-coupled receptors that influence lifespan of human and animal models
Francisco Alejandro Lagunas-Rangel
Biogerontology.2022; 23(1): 1. CrossRef - Protective effects of AdipoRon on the liver of Huoyan goose fed a high-fat diet
Zhongzan Cao, Ben Ma, Chengyu Cui, Jiahui Zhao, Sidi Liu, Yunqiao Qiu, Yan Zheng, Ming Gao, Xinhong Luan
Poultry Science.2022; 101(4): 101708. CrossRef - AdipoRon exerts opposing effects on insulin sensitivity via fibroblast growth factor 21–mediated time-dependent mechanisms
Yongliang Wang, Huan Liu, Ruixin Zhang, Yuyao Xiang, Junfeng Lu, Bo Xia, Liang Peng, Jiangwei Wu
Journal of Biological Chemistry.2022; 298(3): 101641. CrossRef - Compendious Review on Adipokines of Corpulence
Feryal Hashim
Research Journal of Pharmacy and Technology.2022; : 4315. CrossRef - AdipoR agonist increases insulin sensitivity and exercise endurance in AdipoR-humanized mice
Masato Iwabu, Miki Okada-Iwabu, Hiroaki Tanabe, Nozomi Ohuchi, Keiko Miyata, Toshiko Kobori, Sara Odawara, Yuri Kadowaki, Shigeyuki Yokoyama, Toshimasa Yamauchi, Takashi Kadowaki
Communications Biology.2021;[Epub] CrossRef - ADIPOR1 regulates genes involved in milk fat metabolism in goat mammary epithelial cells
Wangsheng Zhao, Michael Adjei, Hongmei Wang, Yueling Yangliu, Jiangjiang Zhu, Huijuan Wu
Research in Veterinary Science.2021; 137: 194. CrossRef - Differentiation of THP-1 monocytes to macrophages increased mitochondrial DNA copy number but did not increase expression of mitochondrial respiratory proteins or mitochondrial transcription factor A
Mizuho Okamoto, Masanori Shimogishi, Akari Nakamura, Yusuke Suga, Kyosuke Sugawara, Michio Sato, Ryotaro Nishi, Akio Fujisawa, Yorihiro Yamamoto, Misato Kashiba
Archives of Biochemistry and Biophysics.2021; 710: 108988. CrossRef - Clinical features, treatment and rehabilitation of new coronavirus infection in patients with metabolic syndrome
Dmitry O. Ivanov, Yury P. Uspenskiy, Andrey M. Sarana, Yulia A. Fominykh, Iana V. Sousova, Dmitry V. Zakharov
Pediatrician (St. Petersburg).2021; 12(5): 5. CrossRef - AdipoRon promotes diabetic fracture repair through endochondral ossification-based bone repair by enhancing survival and differentiation of chondrocytes
Zhongyi Wang, Jinxin Tang, Ying Li, Yu Wang, Yanyang Guo, Qisheng Tu, Jake Chen, Chen Wang
Experimental Cell Research.2020; 387(2): 111757. CrossRef - Discovery of AdipoRon analogues as novel AMPK activators without inhibiting mitochondrial complex I
Geng Sun, Yanping You, Haobin Li, Yalong Cheng, Ming Qian, Xinyu Zhou, Haoliang Yuan, Qing-Long Xu, Liang Dai, Pengfei Wang, Keguang Cheng, Xiaoan Wen, Caiping Chen
European Journal of Medicinal Chemistry.2020; 200: 112466. CrossRef - Insights Into the Controversial Aspects of Adiponectin in
Cardiometabolic Disorders
Emilio Antonio Francischetti, Rômulo Sperduto Dezonne, Cláudia Maria Pereira, Cyro José de Moraes Martins, Bruno Miguel Jorge Celoria, Patrícia Aguiar Cardoso de Oliveira, Virgínia Genelhu de Abreu
Hormone and Metabolic Research.2020; 52(10): 695. CrossRef - Adiponectin and Its Mimics on Skeletal Muscle: Insulin Sensitizers, Fat Burners, Exercise Mimickers, Muscling Pills … or Everything Together?
Michel Abou-Samra, Camille M. Selvais, Nicolas Dubuisson, Sonia M. Brichard
International Journal of Molecular Sciences.2020; 21(7): 2620. CrossRef - AdipoRon, a new therapeutic prospect for Duchenne muscular dystrophy
Michel Abou‐Samra, Camille M. Selvais, Raphael Boursereau, Sophie Lecompte, Laurence Noel, Sonia M. Brichard
Journal of Cachexia, Sarcopenia and Muscle.2020; 11(2): 518. CrossRef - Targeting perivascular and epicardial adipose tissue inflammation: therapeutic opportunities for cardiovascular disease
Rim Rafeh, Anissa Viveiros, Gavin Y. Oudit, Ahmed F. El-Yazbi
Clinical Science.2020; 134(7): 827. CrossRef - Therapeutic effects of AdipoRon on liver inflammation and fibrosis induced by CCl4 in mice
Min Sha, Yaru Gao, Can Deng, Yuemeng Wan, Yuan Zhuang, Xiaochuan Hu, Ying Wang
International Immunopharmacology.2020; 79: 106157. CrossRef - AdipoRon: A Novel Insulin Sensitizer in Various Complications and the Underlying Mechanisms: A Review
Ishfaq Ahmad Bhat, Shaheen Wasil Kabeer, Mohammad Irshad Reza, Reyaz Hassan Mir, Muhammad Ovais Dar
Current Molecular Pharmacology.2020; 13(2): 94. CrossRef - Case Report: Concurrent Resistance and Aerobic Training Regulate Adiponectin Expression and Disease Severity in Multiple Sclerosis: A Case Study
Elisa Grazioli, Ersilia Nigro, Claudia Cerulli, Giovanna Borriello, Annamaria Mancini, Eliana Tranchita, Rita Polito, Attilio Parisi, Pasqualina Buono, Aurora Daniele
Frontiers in Neuroscience.2020;[Epub] CrossRef - Mechanisms of Adiponectin Action: Implication of Adiponectin Receptor Agonism in Diabetic Kidney Disease
Yaeni Kim, Cheol Whee Park
International Journal of Molecular Sciences.2019; 20(7): 1782. CrossRef - Drug development research for novel adiponectin receptor-targeted antidiabetic drugs contributing to healthy longevity
Miki Okada-Iwabu, Masato Iwabu, Toshimasa Yamauchi, Takashi Kadowaki
Diabetology International.2019; 10(4): 237. CrossRef - Potential Role of Adiponectin Receptor Agonist, AdipoRon in Cardiometabolic Disease
Eunhee Cho, Sewon Lee
Exercise Science.2019; 28(2): 102. CrossRef - Adiponectin for the treatment of diabetic nephropathy
Jun Young Lee, Jae Won Yang, Byoung Geun Han, Seung Ok Choi, Jae Seok Kim
The Korean Journal of Internal Medicine.2019; 34(3): 480. CrossRef - Adiponectin receptor agonist AdipoRon relieves endotoxin-induced acute hepatitis in mice
Wen-Ze Xiao, Li Zhang
Chinese Medical Journal.2019; 132(20): 2438. CrossRef - Adiponectin/AdipoR Research and Its Implications for Lifestyle-Related Diseases
Masato Iwabu, Miki Okada-Iwabu, Toshimasa Yamauchi, Takashi Kadowaki
Frontiers in Cardiovascular Medicine.2019;[Epub] CrossRef - Examining the Potential of Developing and Implementing Use of Adiponectin-Targeted Therapeutics for Metabolic and Cardiovascular Diseases
Ying Liu, Vivian Vu, Gary Sweeney
Frontiers in Endocrinology.2019;[Epub] CrossRef - The role of immune cells in the development of adipose tissue dysfunction in cardiovascular diseases
E. G. Uchasova, O. V. Gruzdeva, Yu. A. Dyleva, E. V. Belik, O. L. Barbarash
Russian Journal of Cardiology.2019; (4): 92. CrossRef - Adiponectin and Inflammation in Health and Disease: An Update
Alice G. Geagea, Samir Mallat, Charbel F. Matar, Raymond Zerbe, Estelle Filfili, Maria Francis, Hanine Haidar, Abdo Jurjus
Open Medicine Journal.2018; 5(1): 20. CrossRef - Structure and function analysis of adiponectin receptors toward development of novel antidiabetic agents promoting healthy longevity
Miki Okada-Iwabu, Masato Iwabu, Toshimasa Yamauchi, Takashi Kadowaki
Endocrine Journal.2018; 65(10): 971. CrossRef - Adiponectin induced AMP-activated protein kinase impairment mediates insulin resistance in Bama mini-pig fed high-fat and high-sucrose diet
Miaomiao Niu, Lei Xiang, Yaqian Liu, Yuqiong Zhao, Jifang Yuan, Xin Dai, Hua Chen
Asian-Australasian Journal of Animal Sciences.2017; 30(8): 1190. CrossRef - The role of adiponectin receptors in the regulation of synaptic transmission in the hippocampus
Filippo Weisz, Sonia Piccinin, Dalila Mango, Richard Teke Ngomba, Nicola B. Mercuri, Ferdinando Nicoletti, Robert Nisticò
Synapse.2017;[Epub] CrossRef - Unravelling the adiponectin paradox: novel roles of adiponectin in the regulation of cardiovascular disease
Lavinia Woodward, Ioannis Akoumianakis, Charalambos Antoniades
British Journal of Pharmacology.2017; 174(22): 4007. CrossRef - Annual banned‐substance review: analytical approaches in human sports drug testing
Mario Thevis, Tiia Kuuranne, Hans Geyer, Wilhelm Schänzer
Drug Testing and Analysis.2017; 9(1): 6. CrossRef - Discovery of Novel Insulin Sensitizers: Promising Approaches and Targets
Yadan Chen, Haiming Ma, Dasheng Zhu, Guowei Zhao, Lili Wang, Xiujuan Fu, Wei Chen
PPAR Research.2017; 2017: 1. CrossRef - Adiponectin and Its Receptors in Diabetic Kidney Disease: Molecular Mechanisms and Clinical Potential
Dongqing Zha, Xiaoyan Wu, Ping Gao
Endocrinology.2017; 158(7): 2022. CrossRef - Role of pro- and anti-inflammatory phenomena in the physiopathology of type 2 diabetes and obesity
Luciano Pirola, José Candido Ferraz
World Journal of Biological Chemistry.2017; 8(2): 120. CrossRef - Perivascular adipose tissue as a regulator of vascular disease pathogenesis: identifying novel therapeutic targets
Ioannis Akoumianakis, Akansha Tarun, Charalambos Antoniades
British Journal of Pharmacology.2017; 174(20): 3411. CrossRef - Insight into the Dissociation of Behavior from Histology in Synucleinopathies and in Related Neurodegenerative Diseases
Kazunari Sekiyama, Yoshiki Takamatsu, Wakako Koike, Masaaki Waragai, Takato Takenouchi, Shuei Sugama, Makoto Hashimoto
Journal of Alzheimer's Disease.2016; 52(3): 831. CrossRef - Castration induced browning in subcutaneous white adipose tissue in male mice
Osamu Hashimoto, Tatsuya Noda, Atsushi Morita, Masahiro Morita, Hirofumi Ohtsuki, Makoto Sugiyama, Masayuki Funaba
Biochemical and Biophysical Research Communications.2016; 478(4): 1746. CrossRef - Aldosterone Production and Signaling Dysregulation in Obesity
Andrea Vecchiola, Carlos F. Lagos, Cristian A. Carvajal, Rene Baudrand, Carlos E. Fardella
Current Hypertension Reports.2016;[Epub] CrossRef
- Identification of a novel adiponectin receptor and opioid receptor dual acting agonist as a potential treatment for diabetic neuropathy
- The Role of Glucagon-Like Peptide-1 Receptor Agonists in Type 2 Diabetes: Understanding How Data Can Inform Clinical Practice in Korea
- Seungjoon Oh, Suk Chon, Kyu Jeong Ahn, In-Kyung Jeong, Byung-Joon Kim, Jun Goo Kang
- Diabetes Metab J. 2015;39(3):177-187. Published online June 15, 2015
- DOI: https://doi.org/10.4093/dmj.2015.39.3.177
- 4,209 View
- 47 Download
- 3 Web of Science
- 3 Crossref
-
 Abstract
Abstract
 PDF
PDF PubReader
PubReader Glucagon-like peptide-1 receptor agonists (GLP-1RAs) reduce glycosylated hemoglobin (HbA1c, 0.5% to 1.0%), and are associated with moderate weight loss and a relatively low risk of hypoglycemia. There are differences between Asian and non-Asian populations. We reviewed available data on GLP-1RAs, focusing on Korean patients, to better understand their risk/benefit profile and help inform local clinical practice. Control of postprandial hyperglycemia is important in Asians in whom the prevalence of post-challenge hyperglycemia is higher (vs. non-Asians). The weight lowering effects of GLP-1RAs are becoming more salient as the prevalence of overweight and obesity among Korean patients increases. The higher rate of gastrointestinal adverse events amongst Asian patients in clinical trials may be caused by higher drug exposure due to the lower body mass index of the participants (vs. non-Asian studies). Data on the durability of weight loss, clinically important health outcomes, safety and optimal dosing in Korean patients are lacking. Use of GLP-1RAs is appropriate in several patient groups, including patients whose HbA1c is uncontrolled, especially if this is due to postprandial glucose excursions and patients who are overweight or obese due to dietary problems (e.g., appetite control). The potential for gastrointestinal adverse events should be explained to patients at treatment initiation to facilitate the promotion of better compliance.
-
Citations
Citations to this article as recorded by- Tolerability and Effectiveness of Switching to Dulaglutide in Patients With Type 2 Diabetes Inadequately Controlled With Insulin Therapy
Youngsook Kim, Ji Hye Huh, Minyoung Lee, Eun Seok Kang, Bong-Soo Cha, Byung-Wan Lee
Frontiers in Endocrinology.2022;[Epub] CrossRef - Antihyperglycemic Agent Therapy for Adult Patients with Type 2 Diabetes Mellitus 2017: A Position Statement of the Korean Diabetes Association
Seung-Hyun Ko, Kyu-Yeon Hur, Sang Youl Rhee, Nan-Hee Kim, Min Kyong Moon, Seok-O Park, Byung-Wan Lee, Hyun Jin Kim, Kyung Mook Choi, Jin Hwa Kim
Diabetes & Metabolism Journal.2017; 41(5): 337. CrossRef - Antihyperglycemic agent therapy for adult patients with type 2 diabetes mellitus 2017: a position statement of the Korean Diabetes Association
Seung-Hyun Ko, Kyu-Yeon Hur, Sang Youl Rhee, Nan-Hee Kim, Min Kyong Moon, Seok-O Park, Byung-Wan Lee, Hyun Jin Kim, Kyung Mook Choi, Jin Hwa Kim
The Korean Journal of Internal Medicine.2017; 32(6): 947. CrossRef
- Tolerability and Effectiveness of Switching to Dulaglutide in Patients With Type 2 Diabetes Inadequately Controlled With Insulin Therapy
- Refocusing Peroxisome Proliferator Activated Receptor-α: A New Insight for Therapeutic Roles in Diabetes
- Hannah Seok, Bong Soo Cha
- Diabetes Metab J. 2013;37(5):326-332. Published online October 17, 2013
- DOI: https://doi.org/10.4093/dmj.2013.37.5.326
- 2,759 View
- 30 Download
- 19 Crossref
-
 Abstract
Abstract
 PDF
PDF PubReader
PubReader Although glucose-lowering treatment shows some risk lowering effects in cardiovascular diseases, risks of macrovascular and microvascular complications have still remained, and development of new therapeutic strategies is needed. Recent data have shown that peroxisome proliferator activated receptor-α (PPAR-α) plays a pivotal role in the regulation of lipid homeostasis, fatty acid oxidation, cellular differentiation, and immune response such as inflammation or vascularization related to diabetic complication. This review will re-examine the metabolic role of PPAR-α, summarize data from clinical studies on the effect of PPAR-α agonist in diabetes, and will discuss the possible therapeutic role of PPAR-α activation.
-
Citations
Citations to this article as recorded by- Recent Insights into the Etiopathogenesis of Diabetic Retinopathy and Its Management
Arpon Biswas, Abhijit Deb Choudhury, Sristi Agrawal, Amol Chhatrapati Bisen, Sachin Nashik Sanap, Sarvesh Kumar Verma, Mukesh Kumar, Anjali Mishra, Shivansh Kumar, Mridula Chauhan, Rabi Sankar Bhatta
Journal of Ocular Pharmacology and Therapeutics.2024; 40(1): 13. CrossRef - Counteracting health risks by Modulating Homeostatic Signaling
Junqiang J. Tian, Mark Levy, Xuekai Zhang, Robert Sinnott, Rolando Maddela
Pharmacological Research.2022; 182: 106281. CrossRef - Moringa oleifera leaf extract influences oxidative metabolism in C2C12 myotubes through SIRT1-PPARα pathway
Guglielmo Duranti, Mariateresa Maldini, Domenico Crognale, Stefania Sabatini, Federica Corana, Katy Horner, Roberta Ceci
Phytomedicine Plus.2021; 1(1): 100014. CrossRef - Diabetes risk associated with plasma epoxylipid levels
John D. Imig
eBioMedicine.2021; 66: 103331. CrossRef - Genome-Wide Characterization of Alternative Splicing Events and Their Responses to Cold Stress in Tilapia
Bi Jun Li, Zong Xian Zhu, Hui Qin, Zi Ning Meng, Hao Ran Lin, Jun Hong Xia
Frontiers in Genetics.2020;[Epub] CrossRef - PPARγ provides anti-inflammatory and protective effects in intrahepatic cholestasis of pregnancy through NF-κB pathway
Yan Zhang, Xiaoping Huang, Jie Zhou, Yongxiang Yin, Ting Zhang, Daozhen Chen
Biochemical and Biophysical Research Communications.2018; 504(4): 834. CrossRef - Fermented Ginseng Contains an Agonist of Peroxisome Proliferator Activated Receptors α and γ
Kentaro Igami, Yosuke Shimojo, Hisatomi Ito, Toshitsugu Miyazaki, Fusako Nakano, Yoshiki Kashiwada
Journal of Medicinal Food.2016; 19(9): 817. CrossRef - Long-term oral administration of osteocalcin induces insulin resistance in male mice fed a high-fat, high-sucrose diet
Yu Yasutake, Akiko Mizokami, Tomoyo Kawakubo-Yasukochi, Sakura Chishaki, Ichiro Takahashi, Hiroshi Takeuchi, Masato Hirata
American Journal of Physiology-Endocrinology and Metabolism.2016; 310(8): E662. CrossRef - Peroxisome Proliferator-Activated Receptor-γGene Expression and Its Association with Oxidative Stress in Patients with Metabolic Syndrome
Mehdi Hatami, Massoud Saidijam, Reza Yadegarzari, Shiva Borzuei, Alireza Soltanian, Marzieh Safi Arian, Mohammad Taghi Goodarzi
Chonnam Medical Journal.2016; 52(3): 201. CrossRef - Short Exposure to a High-Sucrose Diet and the First ‘Hit' of Nonalcoholic Fatty Liver Disease in Mice
Flavia Fernandes-Lima, Thiago Luís Ribeiro Gomes Monte, Fernanda Amorim de Morais Nascimento, Bianca Martins Gregório
Cells Tissues Organs.2016; 201(6): 464. CrossRef - In vitro evaluation of dual agonists for PPARγ/β from the flower of Edgeworthia gardneri (wall.) Meisn
Die Gao, Yong-lan Zhang, Pan Xu, Ye-xin Lin, Feng-qing Yang, Jian-hui Liu, Hai-wen Zhu, Zhi-ning Xia
Journal of Ethnopharmacology.2015; 162: 14. CrossRef - PPARα Agonist Fenofibrate Ameliorates Learning and Memory Deficits in Rats Following Global Cerebral Ischemia
Ai-Guo Xuan, Yan Chen, Da-Hong Long, Meng Zhang, Wei-Dong Ji, Wen-Juan Zhang, Ji-Hong Liu, Le-Peng Hong, Xiao-Song He, Wen-Liang Chen
Molecular Neurobiology.2015; 52(1): 601. CrossRef - Modulation of Cardiac Connexin-43 by Omega-3 Fatty Acid Ethyl-Ester Supplementation Demonstrated in Spontaneously Diabetic Rats
J. RADOSINSKA, L. H. KURAHARA, K. HIRAISHI, C. VICZENCZOVA, T. EGAN BENOVA, B. SZEIFFOVA BACOVA, V. DOSENKO, J. NAVAROVA, B. OBSITNIK, I. IMANAGA, T. SOUKUP, N. TRIBULOVA
Physiological Research.2015; : 795. CrossRef - Does Bosentan Protect Diabetic Brain Alterations in Rats? The Role of Endothelin‐1 in the Diabetic Brain
Recep Demir, Elif Cadirci, Erol Akpinar, Yasemin Cayir, Hasan Tarik Atmaca, Harun Un, Celalettin Semih Kunak, Muhammed Yayla, Zafer Bayraktutan, Ilknur Demir
Basic & Clinical Pharmacology & Toxicology.2015; 116(3): 236. CrossRef - The effect of pioglitazone on aldosterone and cortisol production in HAC15 human adrenocortical carcinoma cells
Zhi-qiang Pan, Ding Xie, Vivek Choudhary, Mutsa Seremwe, Ying-Ying Tsai, Lawrence Olala, Xunsheng Chen, Wendy B. Bollag
Molecular and Cellular Endocrinology.2014; 394(1-2): 119. CrossRef - Icariin Is A PPARα Activator Inducing Lipid Metabolic Gene Expression in Mice
Yuan-Fu Lu, Yun-Yan Xu, Feng Jin, Qin Wu, Jing-Shan Shi, Jie Liu
Molecules.2014; 19(11): 18179. CrossRef - Canola Oil Influence on Azoxymethane-induced Colon Carcinogenesis, Hypertriglyceridemia and Hyperglycemia in Kunming Mice
Xiao-Qiong He, Simon Angelo Cichello, Jia-Li Duan, Jin Zhou
Asian Pacific Journal of Cancer Prevention.2014; 15(6): 2477. CrossRef - Cardiovascular risks and benefits with oral drugs for Type 2 diabetes mellitus
Emily Weidman-Evans, Steven M Metz, Jeffery D Evans
Expert Review of Clinical Pharmacology.2014; 7(2): 225. CrossRef - Enhanced pan‐peroxisome proliferator‐activated receptor gene and protein expression in adipose tissue of diet‐induced obese mice treated with telmisartan
Aline Penna‐de‐Carvalho, Francielle Graus‐Nunes, Júlia Rabelo‐Andrade, Carlos Alberto Mandarim‐de‐Lacerda, Vanessa Souza‐Mello
Experimental Physiology.2014; 99(12): 1663. CrossRef
- Recent Insights into the Etiopathogenesis of Diabetic Retinopathy and Its Management
- Role of Glucocorticoid Receptor on Insulin Secretion and Synthesis in INS-1 Cells.
- Ju Yeon Yang, Myong Su Kang, Tak Ho Song, In Kook Jeong, Pyong Ju Seo, Hee Jin Kim
- Korean Diabetes J. 2006;30(6):428-434. Published online November 1, 2006
- DOI: https://doi.org/10.4093/jkda.2006.30.6.428
- 1,589 View
- 23 Download
-
 Abstract
Abstract
 PDF
PDF - BACKGROUND
Glucocorticoids play important roles in the regulation of glucose homeostasis. It is well known that glucocorticoids reduce hepatic and peripheral tissue sensitivity to insulin, but the roles of glucocorticoids on insulin secretion and synthesis in pancreatic beta cells are still unclear. We have investigated the direct effects of glucocorticoids on insulin secretion and synthesis in rat insulinoma (INS-1) cells. METHODS: Insulin content and 11.2 mM glucose-stimulated insulin secretion (GSIS) were measured in INS-1 cells after culture with or without 1 micrometer dexamethasone (DEX). Preproinsulin mRNA levels were analyzed by real-time RT-PCR and normalized to the internal control. Effect of RU486 on DEX-induced inhibition of GSIS and preproinsulin mRNA synthesis was evaluated. RESULTS: Insulin content of INS-1 cells cultured in RPMI containing 11.2 mM glucose in the presence of DEX was not different from that of control cells. After 1-h preincubation in 2.8 mM glucose, basal insulin secretion from cells treated with DEX did not differ from that of controls, but GSIS was significantly reduced in the cells treated with DEX in comparison to control cells. The expression of preproinsulin mRNA relative to beta-actin mRNA was also lower in the cells treated with DEX. Glucocorticoid receptor antagonist improved DEX-induced inhibition of GSIS and preproinsulin mRNA synthesis. CONCLUSION: DEX inhibited GSIS and preproinsulin mRNA synthesis in INS-1 cells. Glucocorticoid receptor antagonist ameliorated the reduced GSIS and preproinsulin mRNA synthesis induced by DEX.
- Effects of PPAR-alpha and-gamma Agonists on Fatty Acid Metabolism of Muscle Cells in Hyperlipidemic and Hyperglycemic Conditions.
- Yong jik Lee, Zheng Shan Zhao, Soo Kyung Kim, Hae Jin Kim, Wan Sub Shim, Chul Woo Ahn, Hyun Chul Lee, Bong Soo Cha
- Korean Diabetes J. 2006;30(5):324-335. Published online September 1, 2006
- DOI: https://doi.org/10.4093/jkda.2006.30.5.324
- 2,090 View
- 24 Download
- 2 Crossref
-
 Abstract
Abstract
 PDF
PDF - BACKGROUND
Studies for the regulation of fatty acid metabolism are deficient relatively in skeletal muscle and heart. The investigations in pathological conditions for malonyl-CoA decarboxylase (MCD) and for the relation of MCD and PPAR-alpha.-gamma agonists are insufficient in particular. METHODS: In the current study, fully differentiated H9c2 muscle cells were exposed to pathological conditions such as hyperlipidemic (0.1 mM Palmitate) and hyperglycemic (16.5 mM Glucose) condition with 5 uM PPAR-gamma agonist (rosiglitazone) and 10 uM PPAR-alpha agonist (WY14,643) and then experiments such as MCD activity assay, MCD real-time RT-PCR, MCD reporter gene assay, MCD Western blotting, PPAR-alpha Western blotting, and palmitate oxidation test were carried out. RESULTS: Only PPAR-alpha agonist increased MCD activity. In the result of real-time RT-PCR, both PPAR-alpha and PPAR-gamma agonists elevated MCD mRNA expression in hyperlipidemic condition. MCD protein expression was decreased in hyperlipidemic condition, however, increased in rosiglitazone, or WY14,643 treated conditions. Rosiglitazone, and WY14,643 treated groups showed incresed MCD protein expression in hyperglycemic condition. Hyperlipidemic control group and PPAR-alpha.-gamma agonists treated groups presented about 3.8 times more increased palmitate oxidation level than normolipidemic control group in hyperlipidemic condition. PPAR-alpha agonist treated group showed 49% more increased palmitate oxidation rate than hyperlipidemic control group in primary cultured rat skeletal muscle cells. The amount of palmitate oxidation from differentiated H9c2 muscle cells that had overexpressed PPAR-alpha structural genes was more increased than control group. CONCLUSION: This study suggests that PPAR-alpha agonist ameliorates the defects induced by hyperlipidemic condition through the regulation of MCD. In summary, a closely reciprocal relation among PPAR-alpha agonist, MCD, and fatty acid oxidation existed distinctly in hyperlipidemic condition, but not in hyperglycemic condition. -
Citations
Citations to this article as recorded by- Beneficial effect of Combination with Korean Red Ginseng and Morus alba in metabolic syndrome
Yun Jung Lee, Hye Yoom Kim, Jung Joo Yoon, So Min Lee, You Mee Ahn, Joung Hyun Kho, Min Chul Kho, Ho Sub Lee, Kyung Min Choi, Dae Gill Kang
The Korea Journal of Herbology.2012; 27(6): 99. CrossRef - Effects of Mixed Extract from Lycium chinense, Cordyceps militaris, and Acanthopanax senticosus on Glucose-Regulating Enzymes of HepG2 in Hyperglycemic Conditions
Dae-Jung Kim, Jeong-Mi Kim, Tae-Hyuk Kim, Jong-Mi Baek, Hyun-Sook Kim, Myeon Choe
Journal of the Korean Society of Food Science and Nutrition.2010; 39(9): 1257. CrossRef
- Beneficial effect of Combination with Korean Red Ginseng and Morus alba in metabolic syndrome
- Effect of Peroxisome Proliferator Activated Receptor-gamma Agonist, Angiotensin II Receptor Blocker and alpha-lipoic Acid on Renal VEGF Expression in Diabetic Nephropathy.
- Jang Hyun Koh, Yeon Lee, Mi Jin Kim, Young Goo Shin, Eun Young Lee, Choon Hee Chung
- Korean Diabetes J. 2004;28(5):367-376. Published online October 1, 2004
- 1,076 View
- 20 Download
-
 Abstract
Abstract
 PDF
PDF - BACKGROUND
Diabetic nephropathy is one of the most serious complications in diabetes mellitus, and it is the leading cause of end stage renal disease. It has been reported that angiotensin converting enzyme inhibitor (ACEi) reduces the vascular endothelial growth factor (VEGF) expression, and so it plays an important role in reducing the renal damage. Peroxisome proliferator activated receptor-gamma (PPAR-gamma) agonist is known to reduce insulin resistance in type 2 diabetic patients. In the previous study, PPAR-gamma agonist was shown to lower VEGF expression in the retina, but it increased the plasma VEGF level. Alpha-lipoic acid (alpha-LA), which is an antioxidant, lowers the increased level of VEGF in retina as well. The precise role of PPAR-gamma agonist and alpha-LA on renal VEGF expression in diabetic nephropathy is still uncertain. We studied the effect of PPAR-gamma agonist, angiotensin II receptor blocker (ATIIRB) and alpha-LA on the renal VEGF expression in diabetic rats. METHODS: We used 60 Sprague-Dawley male rats, those were 8 weeks old and weighted about 300 g each as the study subjects. Among them, 48 rats were chosen and injected with streptozotocin (70 mg/kg) into peritoneal cavity to induce diabetes mellitus. The rast were than divided into 5 groups. Group I was a normal control group (n=12), group II was diabetic control group (n=12), group III was diabetic group that was given with PPAR-gamma agonist (n=12), group IV was the diabetic group that was given ATIIRB (n=12), and group V was the diabetic rats that were given alpha-LA (n=12). We measured their body weight, blood glucose levels, 24 hour urine protein and albumin levels at the baseline, the 8th and the 16th weeks of the experiment. On the 16th weeks of our experiment we extracted the kidneys to measure the glomerular volume, the optical density of the VEGF staining and VEGF mRNA expression. RESULTS: At the beginning of the study, the 5 groups all showed similar 24 hour urine albumin levels. At the 8th week, group II showed an increased urine albumin level of 143.4 +/- 117.2 mg/day; this was greater than that of group IV (60.7+/-30.6 mg/day) (p<0.05). The glomerular volume and optical densities of VEGF expression were significantly reduced in group III, IV and V compared to group II. For group IV and V, the renal VEGF mRNA expression was significantly lower than that of group II, but group III showed no significant difference. from group II. CONCLUSION: Angiotensin II receptor blocker delayed the progression of diabetic nephropathy. PPAR-gamma agonist and alpha-lipoic acid did not have any protective effect against the progression of diabetic nephropathy in spite of the decreased VEGF expression noted in this study.

 KDA
KDA
 First
First Prev
Prev





