- Current
- Browse
- Collections
-
For contributors
- For Authors
- Instructions to authors
- Article processing charge
- e-submission
- For Reviewers
- Instructions for reviewers
- How to become a reviewer
- Best reviewers
- For Readers
- Readership
- Subscription
- Permission guidelines
- About
- Editorial policy
Articles
- Page Path
- HOME > Diabetes Metab J > Volume 41(5); 2017 > Article
-
Original ArticleComplications Features of Long-Standing Korean Type 2 Diabetes Mellitus Patients with Diabetic Retinopathy: A Study Based on Standardized Clinical Data
-
Sejeong Park1, Sang Youl Rhee1, Su Jin Jeong2, Kiyoung Kim3, Suk Chon1, Seung-Young Yu3, Jeong-Taek Woo1

-
Diabetes & Metabolism Journal 2017;41(5):393-404.
DOI: https://doi.org/10.4093/dmj.2017.41.5.393
Published online: September 5, 2017
1Department of Endocrinology and Metabolism, Kyung Hee University School of Medicine, Seoul, Korea.
2Statistics Support Department, Medical Science Research Institute, Kyung Hee University Medical Center, Kyung Hee University School of Medicine, Seoul, Korea.
3Department of Ophthalmology, Kyung Hee University School of Medicine, Seoul, Korea.
- Corresponding author: Jeong-Taek Woo. Department of Endocrinology and Metabolism, Kyung Hee University School of Medicine, 23 Kyungheedae-ro, Dongdaemun-gu, Seoul 02447, Korea. jtwoomd@khmc.or.kr
- *Sejeong Park and Sang Youl Rhee contributed equally to this study as first authors.
Copyright © 2017 Korean Diabetes Association
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/4.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
ABSTRACT
-
Background
- This is part of a prospective study carried out as a national project to secure standardized public resources for type 2 diabetes mellitus (T2DM) patients in Korea. We compared various characteristics of long-standing T2DM patients with diabetic retinopathy (DR) and macular edema (ME).
-
Methods
- From September 2014 to July 2015, T2DM patients with disease duration of at least 15 years were recruited at a single university hospital. Clinical data and samples were collected according to the common data elements and standards of procedure developed by the Korean Diabetes Association Research Council. Each participant was assessed by ophthalmologists for DR and ME.
-
Results
- Among 220 registered patients, 183 completed the ophthalmologic assessment. DR was associated with longer disease duration (odds ratio [OR], 1.071; 95% confidence interval [CI], 1.001 to 1.147 for non-proliferative diabetic retinopathy [NPDR]) (OR, 1.142; 95% CI, 1.051 to 1.242 for proliferative diabetic retinopathy [PDR]) and the use of long-acting insulin (OR, 4.559; 95% CI, 1.672 to 12.427 for NPDR) (OR, 4.783; 95% CI, 1.581 to 14.474 for PDR), but a lower prevalence of a family history of cancer (OR, 0.310; 95% CI, 0.119 to 0.809 for NPDR) (OR, 0.206; 95% CI, 0.063 to 0.673 for PDR). ME was associated with higher glycosylated hemoglobin levels (OR, 1.380; 95% CI, 1.032 to 1.845) and the use of rapid-acting insulin (OR, 5.211; 95% CI, 1.445 to 18.794).
-
Conclusion
- Various clinical features were associated with DR and ME. Additional epidemiological and biorepository-based studies using this cohort are being conducted to deepen our understanding of diabetic complications in Korea.
- Diabetes mellitus is a leading cause of mortality, with an estimated 1.3 million diabetes-related deaths worldwide in 2010, which is twice as many as occurred in 1990 [1]. Diabetes and diabetes-related chronic complications, such as retinopathy, neuropathy, nephropathy, cardiovascular, and cerebrovascular diseases, peripheral artery occlusive diseases, and amputations, also significantly increase medical costs [234]. While the prevalence of diabetes is increasing in Korea, proper glycemic control has not been achieved in Korean type 2 diabetes mellitus (T2DM) patients [56]. The risk of diabetic complications is strongly associated with hyperglycemia, whereas proper glycemic control is likely to reduce complications [27].
- Recently, research based on -omics data has been on the rise following the rapid development of large-scale genome analysis techniques. Such studies may be particularly valuable in T2DM, which is a complex and multifactorial disease with variable clinical features and courses [8]. Success in metabolomics studies relies on proper sample preparation, innovative instrumentation, and bioinformatics tools [9]. The unmet need for a long-term prospective study based on -omics data might be partly due to the difficulty of collecting standardized clinical and biological data [10].
- In this context, the establishment of a structurally designed large-scale registry with standardized data collection methods is needed to draw clinically significant conclusions. This study is part of a national project to provide standardized clinical data and biospecimens for future long-term prospective studies. It is a fundamental study aimed at identifying the properties of diabetic retinopathy (DR) and macular edema (ME) in Korean T2DM patients. Our main goal was to compare the characteristics of long-standing T2DM patients with their DR and/or ME status. DR and ME were used as criteria because they are the most specific complications of T2DM [11].
INTRODUCTION
- Subjects and study design
- This cross-sectional study was carried out as part of a prospective study at Kyung Hee University Hospital in Korea. From September 2014 to July 2015, T2DM patients with disease duration of longer than 15 years were recruited. Clinical data, along with blood and urine samples, were collected from each participant after obtaining informed consent. The statuses of DR and ME were assessed in each participant through ophthalmological exams.
- Common data elements and standard operating procedure
- To secure standardized clinical and biochemical data and to establish a practical action plan, common data elements (CDEs) and standard operating procedures (SOPs) were established. CDEs were initially selected from the case report form (CRF) of a previous Korea National Diabetes Program (KNDP) cohort study [2]. Then the initial draft of the CRF was established by referring to the critical items recommended by the Korean Diabetes Association (KDA) and the American Diabetes Association clinical guidelines [1213]. The SOP was established referring to methods from Korean National Biobank guidelines [14].
- The final forms of the CRF and SOP were announced and confirmed at the opening symposium of the KDA clinical standardization group in January 2015. It was then approved by the board of directors of the KDA in July 2015 Participant clinical data were managed electronically through the iCReaT (Internet based Clinical Research and Trial management) System (Korea Centers for Disease Control and Prevention, Cheongju, Korea), which was developed by the Korea National Research Institute of Health for multicenter registration and long-term follow-up.
- Outcome definition
- The DR status of each participant was assessed through color fundus photographs (FF 450 Plus; Carl Zeiss Meditec, Jena, Germany). According to Early Treatment Diabetic Retinopathy Study (ETDRS) criteria, DR was graded into three categories: no DR, non-proliferative diabetic retinopathy (NPDR), or proliferative diabetic retinopathy (PDR) [1516]. If the eyes of a single patient were rated at different stages, the grade of the worse eye was used. The presence of DME was confirmed using thickness measurements as assessed by Cirrus High-Definition Optical Coherence Tomography (HD-OCT; Carl Zeiss Meditec, Dublin, CA, USA). Two or more ophthalmologists classified the DR and ME statuses based on the exam results. In cases of discordance between the evaluators, they reviewed the images again and agreed on the final interpretation.
- Trained interviewers assessed the presence of underlying comorbidities by surveying the participants and checking the medical records for the use of relevant medications. The status of physical activities and other socioeconomic statuses were also assessed through interviews. Obesity was defined as a body mass index at or above 25 kg/m2. Dyslipidemia was defined as raised triglyceride (≥150 mg/dL) and/or low high density lipoprotein cholesterol (<40 mg/dL in men and <50 mg/dL in women) or on a specific treatment, according to the Third Report of the National Cholesterol Education Program Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults [17].
- Ethics statement
- This study was approved by the Institutional Review Board of Kyung Hee University Hospital (IRB No. KMC IRB 1428-04). Written informed consent was obtained from all participants. Data from this study were registered at the Clinical Research Information Service (CRIS, No. KCT0001269). CRIS is a Korean National Service connected to the International Clinical Trials Registry Platform of the World Health Organization.
- Statistical analyses
- Means, proportions, and distributions of characteristics were compared between patients with or without DR and ME. Chi-square tests were used to measure the significance of associations for categorical variables, and Student t-tests for continuous variables. Multiple logistic regression analysis was performed to assess the independent contribution of variables to DR and/or ME. Variables that were significant in univariate analyses were included as covariates in the model. Statistical analyses were conducted independently by a specialized statistician at the Statistics Support Department of Kyung Hee University Medical Center Medical Science Research Institute. SAS software version 9.3 (SAS Institute Inc., Cary, NC, USA) was used to perform all statistical tests.
METHODS
- Study progression
- Among 220 patients recruited, clinical data and samples were collected from 198 patients who consented to the study. A total of 183 ophthalmologic exams were completed (Fig. 1). The mean age of the participants was 66.8 years, the median duration of DM was 22.6 years, and 49.7% of the participants were male.
- Comparison of characteristics with DR status
- Tables 1 and 2 show selected characteristics compared to the grade of DR. Among a total of 183 participants who underwent ophthalmological assessment, 124 (67.76%) were diagnosed with DR. A total of 72 patients (39.3%) had NPDR and 52 (28.4%) had PDR. Statistically significant differences were found for several factors.
- More severe DR was associated with a younger age (70±11 years old without DR, 66±12.25 years old with NPDR, and 62±17 years old with PDR, P=0.001) and longer duration of DM (20±6 years without DR, 22±7 years with NPDR, and 23.5±9 years with PDR, P=0.022). Urine microalbumin levels (7.9±14.8 µg/mg Cr without DR, 12.55±33.55 µg/mg Cr with NPDR, and 48.2±302.6 µg/mg Cr with PDR, P<0.001) and serum creatinine levels (0.7±0.4 mg/dL without DR, 0.8±0.4 mg/dL with NPDR, and 0.85±0.5 mg/dL with PDR, P=0.017) were higher and creatinine clearance was lower (101.4±42.75 mL/min without DR, 90±40.62 mL/min with NPDR, and 83.6±49.12 mL/min with PDR, P=0.044) in patients with more severe DR. In addition, patients with more severe DR had a lower γ-glutamyltransferase level (24±21 U/L without DR, 27±18.25 U/L with NPDR, and 18±13 U/L with PDR, P=0.001). Groups with more severe DR had a higher proportion of patients with a family history of cardiovascular disease (1.8% without DR, 2.8% with NPDR, and 12% with PDR, P=0.050), but less of a family history of cancer (36.2% without DR, 22.5% with NPDR, and 15.7% with PDR, P=0.045) (Table 1). A greater proportion of patients were using rapid-acting insulin (1.7% without DR, 18.1% with NPDR, and 25% with PDR, P<0.001), long-acting insulin (20.3% without DR, 38.9% with NPDR, and 50% with PDR, P=0.004), and β-blockers (3.4% without DR, 13.9% with NPDR, and 19.2% with PDR, P=0.024) in groups with more severe DR. A larger proportion of patients in the groups with more severe DR had less physical activity (P=0.041) and fewer incidences of exercise per week (P=0.021) (Table 2).
- Among microvascular complications, patients with more severe DR had a greater prevalence of microalbuminuria (30.5% without DR, 36.1% with NPDR, and 67.3% with PDR, P<0.001), overt proteinuria (10.2% without DR, 18.1% with NPDR, and 36.5% with PDR, P=0.003), and autonomic neuropathy (24.1% without DR, 29.9% with NPDR, and 46.8% with PDR, P=0.043). In addition, the prevalence of dyslipidemia was significantly different among the three groups (P=0.018) (Table 2).
- Comparison of characteristics with ME status
- Tables 3 and 4 show selected characteristics comparing values in the presence or absence of ME. Among a total of 182 participants assessed with OCT, ME was present in 46 (25.27%). As with DR, statistically significant differences were found for several factors (Tables 3 and 4). Patients with ME were younger (66.5±12 years old vs. 63.5±15.5 years old, P=0.049), had a longer duration of DM (21±7.5 years vs. 23.5±7 years, P=0.037), and a larger proportion of patients were female (45.6% vs. 65.2%, P=0.027). Glycosylated hemoglobin (7.75%±1.82% vs. 8.25%±2.35%, P=0.029) and urine microalbumin (11.45±27.55 µg/mg Cr vs. 32.45±210.03 µg/mg Cr, P=0.001) levels were higher, along with a higher prevalence of overt proteinuria (16.9% vs. 32.6%, P=0.035) in the ME group (Table 3).
- A greater proportion of patients underwent inpatient treatment (22.8% vs. 39.1%, P=0.036) and surgery (10.3% vs. 30.4%, P=0.002) within the past year. They had a higher prevalence of a family history of cardiovascular disease (1.5% vs. 15.9%, P=0.001). A significantly higher proportion of ME patients were using rapid-acting insulin (11.0% vs. 26.1%, P=0.017), but there was no significant difference in the use of long-acting (P=0.479) or premixed insulin (P=0.837) (Table 4).
- Multivariate analyses for DR
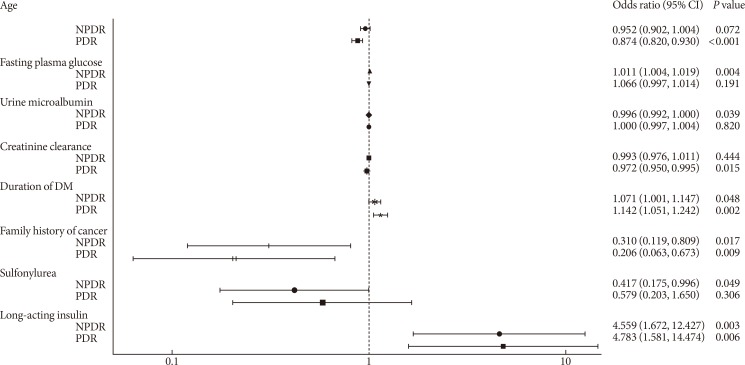
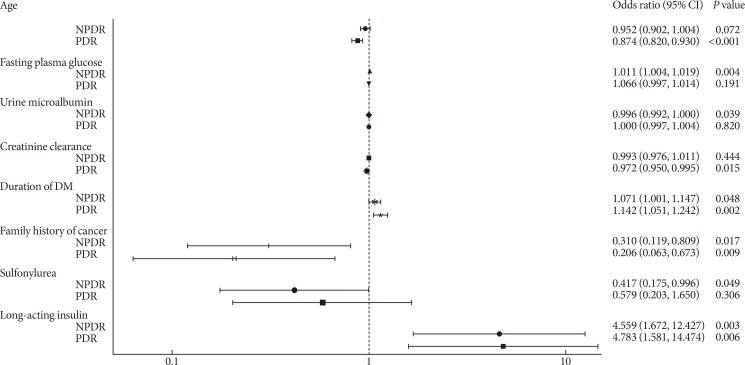
- Multivariate logistic regression analyses were conducted according to the characteristics of the variables shown in Tables 1 and 2. Several factors were found to be significantly associated with DR (Fig. 2). Younger age (odds ratio [OR], 0.874; 95% confidence interval [CI], 0.820 to 0.930; P<0.001) and lower creatinine clearance (OR, 0.972; 95% CI, 0.950 to 0.995; P=0.015) were associated with PDR. NPDR was associated with higher fasting plasma glucose levels (OR, 1.011; 95% CI, 1.004 to 1.019; P=0.004), lower urine microalbumin levels (OR, 0.996; 95% CI, 0.992 to 1.000; P=0.039), and less use of sulfonylurea (OR, 0.417; 95% CI, 0.175 to 0.996; P=0.049). In addition, longer duration of DM (OR, 1.071; 95% CI, 1.001 to 1.147; P=0.048 for NPDR) (OR, 1.142; 95% CI, 1.051 to 1.242; P=0.002 for PDR) and the use of long-acting insulin (OR, 4.559; 95% CI, 1.672 to 12.427; P=0.003 for NPDR) (OR, 4.783; 95% CI, 1.581 to 14.474; P=0.006 for PDR) were identified as independent risk factors for both NPDR and PDR, whereas a family history of cancer was identified as a negative risk factor (OR, 0.310; 95% CI, 0.119 to 0.809; P=0.017 for NPDR) (OR, 0.206; 95% CI, 0.063 to 0.673; P=0.009 for PDR).
- Multivariate analyses for ME
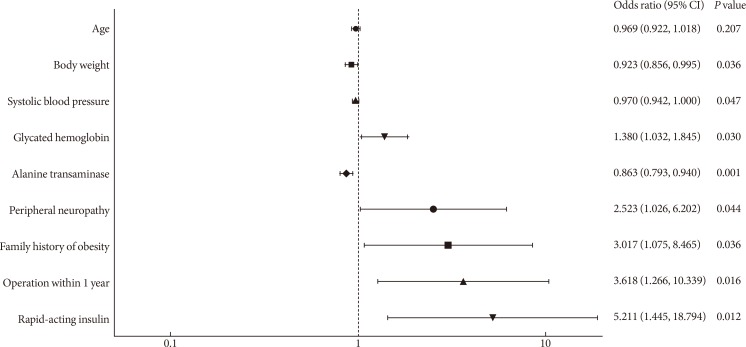
- Several factors were found to be significantly associated with ME in multivariate logistic regression analysis (Fig. 3). Lower body weight (OR, 0.923; 95% CI, 0.856 to 0.995; P=0.036), lower systolic blood pressure (OR, 0.970; 95% CI, 0.942 to 1.000; P=0.047), and lower levels of alanine transaminase (OR, 0.863; 95% CI, 0.793 to 0.940; P<0.001) were associated with ME. Higher glycosylated hemoglobin levels (OR, 1.380, 95% CI, 1.032 to 1.845; P=0.030), peripheral neuropathy (OR, 2.523; 95% CI, 1.026 to 6.202; P=0.044), family history of obesity (OR, 3.017; 95% CI, 1.075 to 8.465; P=0.036), history of surgery within the past year (OR, 3.618; 95% CI, 1.266 to 10.339; P=0.016), and the use of rapid-acting insulin (OR, 5.211; 95% CI, 1.445 to 18.794; P=0.012) were confirmed as independent risk factors for ME.
RESULTS
- A registry-based study for standardized clinical research
- The authors have been running the KNDP since May 2006 [2]. The KNDP is a large-scale multicenter prospective cohort study that was established to observe the long-term clinical course of Korean T2DM patients [18]. It consists of 4,600 T2DM patients collected from 13 Korean university hospitals. A well-structured systemically designed registry was established based on the knowledge and skills obtained through experiences with the KNDP cohort. Initially, it was designed as a case-control study on DR and a control group, but it was changed to a cohort-based study, which provides a higher level of evidence in clinical medicine. The goal of further studies is to perform long-term observations with a larger sample size based on the cohort of this study.
- Summary of findings
- In this study, we confirmed various clinical features associated with the occurrence of DR and ME. DR was associated with higher levels of fasting plasma glucose, longer duration of DM, and greater use of long-acting insulin, but a lower prevalence of a family history of cancer. ME was associated with higher levels of glycosylated hemoglobin, a greater prevalence of surgery within the past year, and greater use of rapid-acting insulin.
- Comparison with previous studies (1): diabetic retinopathy
- The duration of DM, hyperglycemia, and hypertension are known to be major risk factors for DR [1920]. Diabetic nephropathy is also strongly associated with DR [21]. These findings are also valid in the Korean population [2223]. Our results were consistent with previous findings in many ways, but some were inconsistent. For instance, we could not find a significant association of blood pressure with the presence or grade of DR. However, the use of β-blockers was significantly higher in the NPDR and PDR groups than in the group without DR.
- We obtained significant results for several factors that showed unclear significance or inconsistent results in previous studies. A younger age was associated with DR in our study. This might imply the association of DR with the diagnosis of T2DM at a younger age because our study only included T2DM patients with disease duration of longer than 15 years. The use of rapid- and long-acting insulin was associated with DR, which showed inconsistent results as a risk factor in previous studies [21].
- Comparison with previous studies (2): macular edema
- In addition to the major risk factors identified for DR, dyslipidemia is also known to be a strong risk factor for ME [22]. As with DR, some of the findings in our study were inconsistent with previous findings. Systolic blood pressure, underlying hypertension, and the use of any antihypertensive medication did not differ between the non-ME group and the ME group.
- As with DR, a younger age and the use of long-acting insulin were associated with ME. The proportion of females was higher and body weight was lower in patients with ME. In addition, levels of alanine transaminase and γ-glutamyltransferase were lower in the ME group. A history of hospitalization and surgery within the past year was associated with ME. The mechanisms underlying most of these findings are not yet fully understood.
- Clinical implications and future research plans
- Various environmental factors are considered to be significantly associated with the development of T2DM and related chronic complications. Genetic factors are also considered important in disease progression and aggravation [21]. Although much effort has been expended on elucidating significant genetic factors and their influences in the clinical course of T2DM and its complications, the results thus far have not been fully satisfying. One possible reason for this is the difficulty of managing various types of clinical information and biospecimens by a consensus-based methodology, despite recent advances in genotyping technology. Therefore, we should pay more attention to obtaining detailed and accurate information in clinical phenotyping for breakthroughs in relevant research.
- To overcome the above problem, we established the research group on clinical data standardization for diabetes research at the KDA and prepared the draft of standardized data elements and standard procedures for multicenter patient registration and biospecimen collection. With this system, we organized a multicenter, prospective cohort for DR, initiated a genome-wide association study to verify performance, and encouraged participation by other investigators. Recently, this system was approved by the members of the KDA's board of directors.
- In addition, we only included patients with long-standing T2DM in our study, which is distinct from previous studies. This unique study design was based on the hypothesis that there are several genetic and environmental factors that are protective against the development of diabetic complications. In addition, outcome definitions were clarified via ophthalmologic assessment by ophthalmology specialists, enhancing our confidence in the results.
- Some of the results were inconsistent with the conventional risk factors established in previous studies. This might have been due to the limitations of our study. First, it was a single-center study with a small sample size. Second, causal relationships could not be identified since it was a cross-sectional study performed on baseline data. Third, the main reason for withdrawal was difficulty in performing complete ophthalmologic exams. This might have led to selection bias by excluding patients with poor performance status.
- We are planning a 10-year follow-up of the data collected in this study, and additional recruitment is in progress. The limitations of the current study can be overcome by integrating data with other centers. This can be easily done through the electronic CRFs, CDEs, and SOPs established in this study. Future longitudinal studies will focus on identifying biomarkers associated with diabetic complications and assessing responses to medical treatment.
DISCUSSION
-
Acknowledgements
- All of the authors thank Professor Emeritus Young Seol Kim at Kyung Hee University for his teaching and inspiration, which encouraged us to conduct the present study. This research was supported by a grant from the Korea Health Technology R&D Project through the Korea Health Industry Development Institute (KHIDI), funded by the Ministry of Health and Welfare, Republic of Korea (HD14B1246 and HI16C2048).
ACKNOWLEDGMENTS
-
CONFLICTS OF INTEREST: No potential conflict of interest relevant to this article was reported.
NOTES
- 1. Lozano R, Naghavi M, Foreman K, Lim S, Shibuya K, Aboyans V, Abraham J, Adair T, Aggarwal R, Ahn SY, Alvarado M, Anderson HR, Anderson LM, Andrews KG, Atkinson C, Baddour LM, Barker-Collo S, Bartels DH, Bell ML, Benjamin EJ, Bennett D, Bhalla K, Bikbov B, Bin Abdulhak A, Birbeck G, Blyth F, Bolliger I, Boufous S, Bucello C, Burch M, Burney P, Carapetis J, Chen H, Chou D, Chugh SS, Coffeng LE, Colan SD, Colquhoun S, Colson KE, Condon J, Connor MD, Cooper LT, Corriere M, Cortinovis M, de Vaccaro KC, Couser W, Cowie BC, Criqui MH, Cross M, Dabhadkar KC, Dahodwala N, De Leo D, Degenhardt L, Delossantos A, Denenberg J, DesJarlais DC, Dharmaratne SD, Dorsey ER, Driscoll T, Duber H, Ebel B, Erwin PJ, Espindola P, Ezzati M, Feigin V, Flaxman AD, Forouzanfar MH, Fowkes FG, Franklin R, Fransen M, Freeman MK, Gabriel SE, Gakidou E, Gaspari F, Gillum RF, Gonzalez-Medina D, Halasa YA, Haring D, Harrison JE, Havmoeller R, Hay RJ, Hoen B, Hotez PJ, Hoy D, Jacobsen KH, James SL, Jasrasaria R, Jayaraman S, Johns N, Karthikeyan G, Kassebaum N, Keren A, Khoo JP, Knowlton LM, Kobusingye O, Koranteng A, Krishnamurthi R, Lipnick M, Lipshultz SE, Ohno SL, Mabweijano J, MacIntyre MF, Mallinger L, March L, Marks GB, Marks R, Matsumori A, Matzopoulos R, Mayosi BM, McAnulty JH, McDermott MM, McGrath J, Mensah GA, Merriman TR, Michaud C, Miller M, Miller TR, Mock C, Mocumbi AO, Mokdad AA, Moran A, Mulholland K, Nair MN, Naldi L, Narayan KM, Nasseri K, Norman P, O'Donnell M, Omer SB, Ortblad K, Osborne R, Ozgediz D, Pahari B, Pandian JD, Rivero AP, Padilla RP, Perez-Ruiz F, Perico N, Phillips D, Pierce K, Pope CA 3rd, Porrini E, Pourmalek F, Raju M, Ranganathan D, Rehm JT, Rein DB, Remuzzi G, Rivara FP, Roberts T, De Leon FR, Rosenfeld LC, Rushton L, Sacco RL, Salomon JA, Sampson U, Sanman E, Schwebel DC, Segui-Gomez M, Shepard DS, Singh D, Singleton J, Sliwa K, Smith E, Steer A, Taylor JA, Thomas B, Tleyjeh IM, Towbin JA, Truelsen T, Undurraga EA, Venketasubramanian N, Vijayakumar L, Vos T, Wagner GR, Wang M, Wang W, Watt K, Weinstock MA, Weintraub R, Wilkinson JD, Woolf AD, Wulf S, Yeh PH, Yip P, Zabetian A, Zheng ZJ, Lopez AD, Murray CJ, AlMazroa MA, Memish ZA. Global and regional mortality from 235 causes of death for 20 age groups in 1990 and 2010: a systematic analysis for the Global Burden of Disease Study 2010. Lancet 2012;380:2095-2128. ArticlePubMedPMC
- 2. Rhee SY, Chon S, Kwon MK, Park IeB, Ahn KJ, Kim IJ, Kim SH, Lee HW, Koh KS, Kim DM, Baik SH, Lee KW, Nam MS, Park YS, Woo JT, Kim YS. Prevalence of chronic complications in Korean patients with type 2 diabetes mellitus based on the Korean National Diabetes Program. Diabetes Metab J 2011;35:504-512. ArticlePubMedPMC
- 3. Rhee SY, Guan H, Liu ZM, Cheng SW, Waspadji S, Palmes P, Tai TY, Suwanwalaikorn S, Kim YS. PAD-SEARCH Study Group. Multi-country study on the prevalence and clinical features of peripheral arterial disease in Asian type 2 diabetes patients at high risk of atherosclerosis. Diabetes Res Clin Pract 2007;76:82-92. ArticlePubMed
- 4. Moon EJ, Jo YE, Park TC, Kim YK, Jung SH, Kim HJ, Kim DJ, Chung YS, Lee KW. Clinical characteristics and direct medical costs of type 2 diabetic patients. Korean Diabetes J 2008;32:358-365.Article
- 5. Rhee SY, Kim YS, Oh S, Choi WH, Park JE, Jeong WJ. Diabcare Asia 2001: Korea country report on outcome data and analysis. Korean J Intern Med 2005;20:48-54. ArticlePubMedPMC
- 6. Jung JH, Lee JH, Noh JW, Park JE, Kim HS, Yoo JW, Song BR, Lee JR, Hong MH, Jang HM, Na Y, Lee HJ, Lee JM, Kang YG, Kim SY, Sim KH. Current status of management in type 2 diabetes mellitus at general hospitals in South Korea. Diabetes Metab J 2015;39:307-315. ArticlePubMedPMC
- 7. Stratton IM, Adler AI, Neil HA, Matthews DR, Manley SE, Cull CA, Hadden D, Turner RC, Holman RR. Association of glycaemia with macrovascular and microvascular complications of type 2 diabetes (UKPDS 35): prospective observational study. BMJ 2000;321:405-412. ArticlePubMedPMC
- 8. Kussmann M, Morine MJ, Hager J, Sonderegger B, Kaput J. Perspective: a systems approach to diabetes research. Front Genet 2013;4:205ArticlePubMedPMC
- 9. Fernie AR, Trethewey RN, Krotzky AJ, Willmitzer L. Metabolite profiling: from diagnostics to systems biology. Nat Rev Mol Cell Biol 2004;5:763-769. ArticlePubMedPDF
- 10. Houle D, Govindaraju DR, Omholt S. Phenomics: the next challenge. Nat Rev Genet 2010;11:855-866. ArticlePubMedPDF
- 11. Cheung N, Mitchell P, Wong TY. Diabetic retinopathy. Lancet 2010;376:124-136. ArticlePubMed
- 12. Ko SH, Kim SR, Kim DJ, Oh SJ, Lee HJ, Shim KH, Woo MH, Kim JY, Kim NH, Kim JT, Kim CH, Kim HJ, Jeong IK, Hong EK, Cho JH, Mok JO, Yoon KH; Committee of Clinical Practice Guidelines, Korean Diabetes Association. 2011 Clinical practice guidelines for type 2 diabetes in Korea. Diabetes Metab J 2011;35:431-436. ArticlePubMedPMC
- 13. American Diabetes Association. Standards of medical care in diabetes: 2013. Diabetes Care 2013;36(Suppl 1):S11-S66. ArticlePubMedPDF
- 14. National Biobank of Korea. Guidelines for deposit and consignment of human bioresources. Cheongju: National Biobank of Korea; 2014.
- 15. Wilkinson CP, Ferris FL 3rd, Klein RE, Lee PP, Agardh CD, Davis M, Dills D, Kampik A, Pararajasegaram R, Verdaguer JT. Global Diabetic Retinopathy Project Group. Proposed international clinical diabetic retinopathy and diabetic macular edema disease severity scales. Ophthalmology 2003;110:1677-1682. ArticlePubMed
- 16. Wu L, Fernandez-Loaiza P, Sauma J, Hernandez-Bogantes E, Masis M. Classification of diabetic retinopathy and diabetic macular edema. World J Diabetes 2013;4:290-294. ArticlePubMedPMC
- 17. Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults. Executive summary of the third report of the National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III). JAMA 2001;285:2486-2497. ArticlePubMed
- 18. Rhee SY, Hong SM, Chon S, Ahn KJ, Kim SH, Baik SH, Park YS, Nam MS, Lee KW, Woo JT, Kim YS. Hypoglycemia and medical expenses in patients with type 2 diabetes mellitus: an analysis based on the Korea National Diabetes Program Cohort. PLoS One 2016;11:e0148630ArticlePubMedPMC
- 19. Ding J, Wong TY. Current epidemiology of diabetic retinopathy and diabetic macular edema. Curr Diab Rep 2012;12:346-354. ArticlePubMedPDF
- 20. Yau JW, Rogers SL, Kawasaki R, Lamoureux EL, Kowalski JW, Bek T, Chen SJ, Dekker JM, Fletcher A, Grauslund J, Haffner S, Hamman RF, Ikram MK, Kayama T, Klein BE, Klein R, Krishnaiah S, Mayurasakorn K, O'Hare JP, Orchard TJ, Porta M, Rema M, Roy MS, Sharma T, Shaw J, Taylor H, Tielsch JM, Varma R, Wang JJ, Wang N, West S, Xu L, Yasuda M, Zhang X, Mitchell P, Wong TY. Meta-Analysis for Eye Disease (META-EYE) Study Group. Global prevalence and major risk factors of diabetic retinopathy. Diabetes Care 2012;35:556-564. ArticlePubMedPMCPDF
- 21. Lee R, Wong TY, Sabanayagam C. Epidemiology of diabetic retinopathy, diabetic macular edema and related vision loss. Eye Vis (Lond) 2015;2:17ArticlePubMedPMC
- 22. Jee D, Lee WK, Kang S. Prevalence and risk factors for diabetic retinopathy: the Korea National Health and Nutrition Examination Survey 2008-2011. Invest Ophthalmol Vis Sci 2013;54:6827-6833. ArticlePubMed
- 23. Kim YJ, Kim JG, Lee JY, Lee KS, Joe SG, Park JY, Kim MS, Yoon YH. Development and progression of diabetic retinopathy and associated risk factors in Korean patients with type 2 diabetes: the experience of a tertiary center. J Korean Med Sci 2014;29:1699-1705. ArticlePubMedPMCPDF
REFERENCES
Odds ratio of diabetic retinopathy based on various demographic, lifestyle, social, and clinical factors. NPDR, non-proliferative diabetic retinopathy; PDR, proliferative diabetic retinopathy; DM, diabetes mellitus; CI, confidence interval.
Odds ratio of macular edema based on various demographic, lifestyle, social, and clinical factors. CI, confidence interval.

Comparison of characteristics with DR status
Values are presented as number (%) or mean±standard deviation. P value for the chi-square test comparing the groups with and without DR. Statistical comparisons were performed using the chi-square test (nominal data) or the Mann-Whitney U test (continuous data).
DR, diabetic retinopathy; NPDR, non-proliferative diabetic retinopathy; PDR, proliferative diabetic retinopathy; DM, diabetes mellitus; LDL-C, low density lipoprotein cholesterol.
Comparison of the underlying comorbidities and the use of medications with a grade of DR
Values are presented as number (%). P value for the chi-square test comparing the groups with and without DR. Statistical comparisons were performed using the chi-square test (nominal data) or the Mann-Whitney U test (continuous data).
DR, diabetic retinopathy; NPDR, non-proliferative diabetic retinopathy; PDR, proliferative diabetic retinopathy; SGLT-2, sodium/glucose co-transporter 2; GLP-1, glucagon-like peptide-1; ACEi, angiotensin-converting enzyme inhibitor.
Comparison of characteristics with ME status
Values are presented as number (%) or mean±standard deviation. P value for the chi-square test comparing the groups with and without ME. Statistical comparisons were performed using the chi-square test (nominal data) or the Mann-Whitney U test (continuous data).
ME, macular edema; DM, diabetes mellitus; LDL-C, low density lipoprotein cholesterol.
Comparison of the underlying comorbidities and medication use with the presence of ME
Values are presented as number (%). P value for the chi-square test comparing the groups with and without ME. Statistical comparisons were performed using the chi-square test (nominal data) or the Mann-Whitney U test (continuous data).
ME, macular edema; SGLT-2, sodium/glucose co-transporter 2; GLP-1, glucagon-like peptide-1; ACEi, angiotensin-converting enzyme inhibitor.
Figure & Data
References
Citations

- Diabetic retinopathy related homeostatic dysregulation and its association with mortality among diabetes patients: A cohort study from NHANES
Wenxiang Li, Zhaoguang Lai, Ningning Tang, Fen Tang, Guangyi Huang, Peng Lu, Li Jiang, Daizai Lei, Fan Xu
Diabetes Research and Clinical Practice.2024; 207: 111081. CrossRef - Study on the Interaction Between C3 Gene Polymorphism and Environment in Patients with Type 2 Diabetes Combined with Coronary Artery Disease
Haitang Qiu, Shajidan Abudureyimu, Mengjia Liu, Fen Liu, Ying Gao
Diabetes, Metabolic Syndrome and Obesity.2024; Volume 17: 1467. CrossRef - Cluster analysis of patient characteristics, treatment modalities, renal impairments, and inflammatory markers in diabetes mellitus
Milena Cojic, Aleksandra Klisic, Amina Sahmanovic, Nemanja Petrovic, Gordana Kocic
Scientific Reports.2024;[Epub] CrossRef - Amino acid analysis as a method of discovering biomarkers for diagnosis of diabetes and its complications
Dan Cai, Biao Hou, Song Lin Xie
Amino Acids.2023; 55(5): 563. CrossRef - The correlation between peripheral complete blood count parameters and diabetic macular edema in proliferative diabetic retinopathy patients: a cross-sectional study
Chunyan Lei, Jinyue Gu, Lili Liu, Keren Zhang, Meixia Zhang
Frontiers in Endocrinology.2023;[Epub] CrossRef - The association between different hypoglycemic regimens and postoperative diabetic macular edema after vitrectomy in the Japanese patients with proliferative diabetic retinopathy
Chunyan Lei, Yun Zhang, Meixia Zhang
Frontiers in Endocrinology.2022;[Epub] CrossRef - Plasma amino acids and oxylipins as potential multi-biomarkers for predicting diabetic macular edema
Sang Youl Rhee, Eun Sung Jung, Dong Ho Suh, Su Jin Jeong, Kiyoung Kim, Suk Chon, Seung-Young Yu, Jeong-Taek Woo, Choong Hwan Lee
Scientific Reports.2021;[Epub] CrossRef - SYSTEMIC FACTORS AND EARLY TREATMENT RESPONSE TO INTRAVITREAL INJECTION FOR DIABETIC MACULAR EDEMA
Hyeseong Hwang, Hwanho Lee, Jin Young Kim, Suhwan Lee, Eoi Jong Seo, Ju Byung Chae, Dong Yoon Kim
Retina.2021; 41(6): 1275. CrossRef - Relationship between Clinical Features of Diabetic Retinopathy and Systemic Factors in Patients with Newly Diagnosed Type II Diabetes Mellitus
Hyeseong Hwang, Jin Young Kim, Tae Keun Oh, Ju Byung Chae, Dong Yoon Kim
Journal of Korean Medical Science.2020;[Epub] CrossRef - Clinical Characteristics and Prevalence of Comorbidities according to Metformin Use in Korean Patients with Type 2 Diabetes
Sang Ouk Chin, In Gyoon Ha, Sang Youl Rhee, Su Jin Jeong, Suk Chon, Sung Hoon Kim, Kyu Jeung Ahn, Sei Hyun Baik, Yongsoo Park, Moon Suk Nam, Kwan Woo Lee, Jeong Taek Woo
International Journal of Endocrinology.2020; 2020: 1. CrossRef - Letter: Dipeptidyl Peptidase-4 Inhibitors versus Other Antidiabetic Drugs Added to Metformin Monotherapy in Diabetic Retinopathy Progression: A Real World-Based Cohort Study (Diabetes Metab J 2019;43:640–8)
Jun Sung Moon
Diabetes & Metabolism Journal.2019; 43(6): 911. CrossRef - Prevalence and epidemiological characteristics of patients with diabetic retinopathy in Slovakia: 12-month results from the DIARET SK study
Marta Ondrejkova, Peter Jackuliak, Emil Martinka, Marian Mokan, James Foley, Jana Fabkova, Karol Gecik, Iveta Tvrda, Miroslav Helbich, Monika Gajdosova, Tomislav Bulum
PLOS ONE.2019; 14(12): e0223788. CrossRef - Past and Current Status of Adult Type 2 Diabetes Mellitus Management in Korea: A National Health Insurance Service Database Analysis
Seung-Hyun Ko, Kyungdo Han, Yong-ho Lee, Junghyun Noh, Cheol-Young Park, Dae-Jung Kim, Chang Hee Jung, Ki-Up Lee, Kyung-Soo Ko
Diabetes & Metabolism Journal.2018; 42(2): 93. CrossRef - Letter: Features of Long-Standing Korean Type 2 Diabetes Mellitus Patients with Diabetic Retinopathy: A Study Based on Standardized Clinical Data (Diabetes Metab J 2017;41:393-404)
Jae-Seung Yun
Diabetes & Metabolism Journal.2017; 41(6): 492. CrossRef - Response: Features of Long-Standing Korean Type 2 Diabetes Mellitus Patients with Diabetic Retinopathy: A Study Based on Standardized Clinical Data (Diabetes Metab J 2017;41:393-404)
Sang Youl Rhee, Jeong-Taek Woo
Diabetes & Metabolism Journal.2017; 41(6): 494. CrossRef

 KDA
KDA
 PubReader
PubReader Cite
Cite