- Current
- Browse
- Collections
-
For contributors
- For Authors
- Instructions to authors
- Article processing charge
- e-submission
- For Reviewers
- Instructions for reviewers
- How to become a reviewer
- Best reviewers
- For Readers
- Readership
- Subscription
- Permission guidelines
- About
- Editorial policy
Articles
- Page Path
- HOME > Diabetes Metab J > Volume 39(2); 2015 > Article
-
Original ArticleEpidemiology Clinical Features and Causes of Endogenous Hyperinsulinemic Hypoglycemia in Korea
- Chang-Yun Woo1, Ji Yun Jeong1, Jung Eun Jang1, Jaechan Leem1, Chang Hee Jung1, Eun Hee Koh1, Woo Je Lee1, Min-Seon Kim1, Joong-Yeol Park1, Jung Bok Lee2, Ki-Up Lee1
-
Diabetes & Metabolism Journal 2015;39(2):126-131.
DOI: https://doi.org/10.4093/dmj.2015.39.2.126
Published online: March 9, 2015
1Department of Internal Medicine, University of Ulsan College of Medicine, Seoul, Korea.
2Department of Statistics, University of Ulsan College of Medicine, Seoul, Korea.
- Corresponding author: Ki-Up Lee. Department of Internal Medicine, University of Ulsan College of Medicine, 88 Olympic-ro 43-gil, Songpa-gu, Seoul 138-736, Korea. kulee@amc.seoul.kr
- *Chang-Yun Woo and Ji Yun Jeong contributed equally to this study as first authors.
†Current affiliation: Department of Internal Medicine, Soonchunhyang University Gumi Hospital, Soonchunhyang University College of Medicine, Gumi, Korea.
Copyright © 2015 Korean Diabetes Association
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
ABSTRACT
-
Background
- Endogenous hyperinsulinemic hypoglycemia (EHH) is characterized by an inappropriately high plasma insulin level, despite a low plasma glucose level. Most of the EHH cases are caused by insulinoma, whereas nesidioblastosis and insulin autoimmune syndrome (IAS) are relatively rare.
-
Methods
- To evaluate the relative frequencies of various causes of EHH in Korea, we retrospectively analyzed 84 patients who were diagnosed with EHH from 1998 to 2012 in a university hospital.
-
Results
- Among the 84 EHH patients, 74 patients (88%), five (6%), and five (6%) were diagnosed with insulinoma, nesidioblastosis or IAS, respectively. The most common clinical manifestation of EHH was neuroglycopenic symptoms. Symptom duration before diagnosis was 14.5 months (range, 1 to 120 months) for insulinoma, 1.0 months (range, 6 days to 7 months) for nesidioblastosis, and 2.0 months (range, 1 to 12 months) for IAS. One patient, who was diagnosed with nesidioblastosis in 2006, underwent distal pancreatectomy but was later determined to be positive for insulin autoantibodies. Except for one patient who was diagnosed in 2007, the remaining three patients with nesidioblastosis demonstrated severe hyperinsulinemia (157 to 2,719 µIU/mL), which suggests that these patients might have had IAS, rather than nesidioblastosis.
-
Conclusion
- The results of this study suggest that the prevalence of IAS may be higher in Korea than previously thought. Therefore, measurement of insulin autoantibody levels is warranted for EHH patients, especially in patients with very high plasma insulin levels.
- Endogenous hyperinsulinemic hypoglycemia (EHH) is characterized by low blood glucose due to excessive endogenous insulin without injected insulin. Insulinoma, an islet cell tumor, is the most common cause of EHH [1]. The incidence of insulinomas are approximately 4 per one million person-years [2]. Approximately 90% of insulinomas are benign, single, <2 cm in diameter and can be cured by surgical resection [2,3]. Nesidioblastosis, a disease characterized by pancreatic β-cell hyperplasia and hypertrophy, is usually reported in infants but is rare in adults [4,5]. Insulin autoimmune syndrome (IAS) is another cause of EHH and is characterized by the presence of autoantibodies to native insulin or the insulin receptor [6]. IAS is also extremely rare, and only scattered cases have been reported worldwide [7,8].
- The clinical manifestations of EHH result from hypoglycemia. Hypoglycemic symptoms can be grouped into two major categories, adrenergic or neuroglycopenic. The release of catecholamines in response to glucose levels <55 mg/dL (3 mmol/L) causes adrenergic symptoms, such as diaphoresis, weakness, hunger, tremor, nausea, warm sensations, anxiety, and palpitations [2]. However, the most common and convincing symptom of EHH is neuroglycopenia, including loss of consciousness, diplopia, blurred vision, confusion, abnormal behavior, and amnesia. Although it was originally thought that these symptoms would only become evident during fasting or after exercise, a portion of patients with EHH also develop postprandial symptoms. Therefore, concomitant ≤45 mg/dL (2.5 mmol/L) plasma glucose and ≥3 µIU/mL plasma insulin on immunoradiometric assay (IRMA) is required to diagnose EHH, regardless of whether hypoglycemic symptoms occur in the fasting or postprandial state.
- We previously reported the clinical manifestations of EHH patients who visited our hospital between 1990 to 1997 [9]. At that time, we reported that among 16 EHH patients, three patients had neisidioblastosis. Because nesidioblastosis is highly rare in adults, we extended this observation and reanalyzed the clinical characteristics and causes of EHH in patients who visited our hospital between 1998 and 2012. We confirmed that a relatively high proportion of patients did have noninsulinoma, but we observed that a large portion of these patients may have had IAS (which we did not consider in our previous report [9]), rather than nesidioblastosis.
INTRODUCTION
- Patients and classification
- In this retrospective study, we reviewed the medical records of 84 adult patients who were diagnosed with EHH between 1998 and 2012 at Asan Medical Center, a tertiary referral center in South Korea. The data collected included demographic features, clinical presentations, biochemical laboratory data, the findings of any localization studies, operative data, histological reports, and treatment outcomes. Since 2007, we have routinely examined the presence of anti-insulin antibodies and anti-insulin receptor antibodies in patients in whom tumors could not be localized using noninvasive studies, including computed tomography (CT) or magnetic resonance imaging (MRI). CT examinations were performed using either 8, 16, 64, or 128-row multidetector CT (Siemens Medical Systems, Erlangen, Germany) and MRI was performed using Abanto 1.5T (Siemens Medical Systems). Anti-insulin antibodies were measured using radioimmunoassay (Cobra 5010; Biosource Europe, Nivelles, Belgium), and insulin receptor antibodies were measured using a radioreceptor assay (BML, Tokyo, Japan). Determination of the duration of hypoglycemic symptoms and the amount of weight gained after symptom occurrence was based on each patient's self-reports.
- Procedures for diagnosis of endogenous hyperinsulinemic hypoglycemia
- All patients suspected of having EHH were biochemically diagnosed, except for patients who had a documented episode of hypoglycemia at another hospital prior to admission to our center. When the clinical symptoms of a patient suggested postprandial hypoglycemia, we first performed an extended 5-hour 75-g oral glucose tolerance test (OGTT). This was followed by 72-hour fasting test, which was conducted according to standardized procedures [3]. Plasma glucose, insulin, and C-peptide levels were measured every 4 hours. The fasting tests were terminated when the plasma glucose level dropped <45 mg/dL (2.5 mmol/L), or if hypoglycemic symptoms developed.
- Plasma insulin levels were measured using a commercial IRMA kit (TFB, Tokyo, Japan). The detection sensitivity of this kit is between 0.1 and 200 µIU/mL (0.7 and 1,435 mmol/L). The samples were diluted and re-measured when insulin concentrations were >200 µIU/mL (1,435 mmol/L).
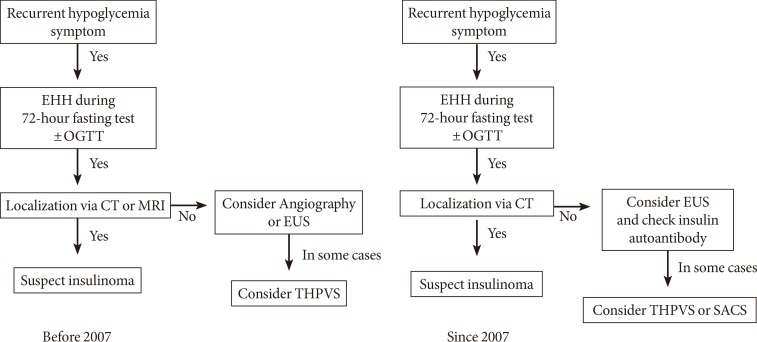
- To localize the source of hyperinsulinemia, CT or MRI was initially used to examine all patients. If these tests did not identify a tumor, endoscopic ultrasound (EUS; GF-UE 260; Olympus, Tokyo, Japan) or selective angiography was subsequently performed. Since 2007, we have preferred CT over MRI, and EUS over angiography. A portion of patients who had negative results with these studies underwent trans-hepatic portal vein sampling (THPVS) or selective arterial calcium stimulation test as alternative localization methods (Fig. 1) [1,2]. Intraoperative ultrasonography was performed on select patients when tumor localization failed. All procedures were performed by experienced radiologists.
- Statistical analysis
- The data presented in the text and tables are shown as the median and range values (minimum to maximum) because most of our data were not normally distributed, and we could enroll only a small number of patients with nesidioblastosis and IAS. The biochemical characteristics of the study population were compared using a Kruskal-Wallis and Fisher exact tests. All statistical analyses were performed using SPSS version 17.0 (SPSS Inc., Chicago, IL, USA). P<0.05 was considered to be statistically significant.
METHODS
- Demographic and biochemical data and diagnosis
- We diagnosed 84 patients with EHH. Of them, 74 patients had insulinoma, five patients had nesidioblastosis, and five patients had IAS. Among the 74 patients with insulinoma, four had multiple endocrine neoplasia type 1. One patient with insulinoma was diagnosed with low-grade malignancy. No patients with nesidioblastosis had a history of gastric bypass surgery [4].
- A total of 70 patients with insulinoma were surgically confirmed by pancreatic resection and immunohistochemical staining against insulin. Two patients were diagnosed by EUS-guided fine-needle aspiration, and the other two patients were diagnosed by confirming EHH and localizing of the insulinoma on EUS or THPVS. Nesidioblastosis was diagnosed in the pathological specimens obtained at surgery according to the following histological criteria: no evidence of insulinoma, hypertrophic β-cells within enlarged or normal-appearing islets, small scattered clusters of endocrine cells, and ductuloinsular complexes [10].
- The median duration of symptoms, which was defined as the time from symptom onset to diagnosis, was 14.5 months (range, 1 to 120 months), and the average was 24.3 months among insulinoma patients. Conversely, the median duration of symptoms was 1.0 month (range, 0.2 to 7 months) and 2.0 months (range, 1 to 12 months) in patients with nesidioblastosis and IAS, respectively (P=0.006) (Table 1). Of the 84 EHH patients, 76 (90.5%) presented with hypoglycemic symptoms, such as neuroglycopenia. There was no statistically significant difference in terms of the number of patients who presented with hypoglycemic symptoms among the three groups (Table 1).
- A total of 38 patients did not undergo the 72-hour fasting test because their relatively high insulin levels during spontaneous fasting-induced hypoglycemia clearly indicated EHH. Of the remaining 46 patients who underwent the 72-hour fasting test, 42 patients (91.3%) developed hypoglycemia. Of them, 39 (92.9%) developed hypoglycemia within 24 hours, 2 (4.8%) developed hypoglycemia during the next 24 hours and 1 (2.4%) developed hypoglycemia after 48 hours (Table 1). Interestingly, all patients with insulinomas developed hypoglycemia during the 72-hour fasting test, but only three of seven noninsulinoma patients developed fasting hypoglycemia.
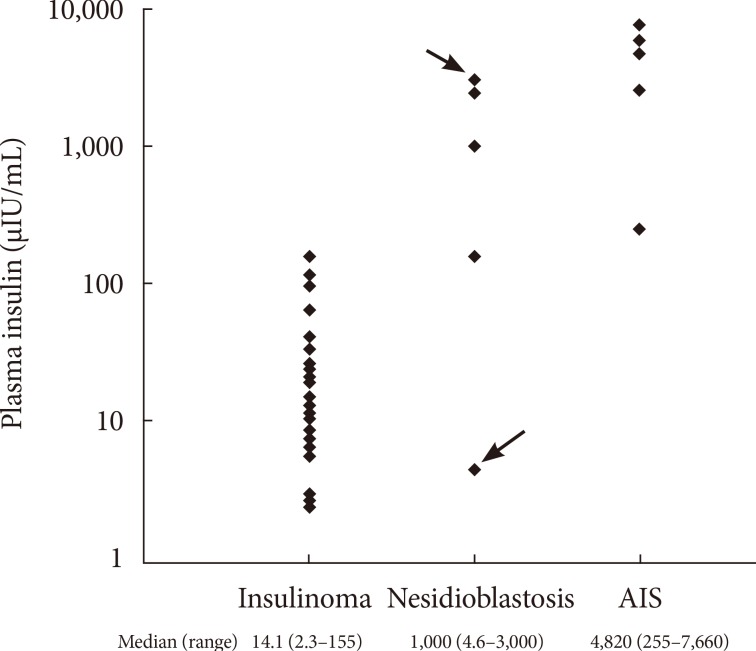
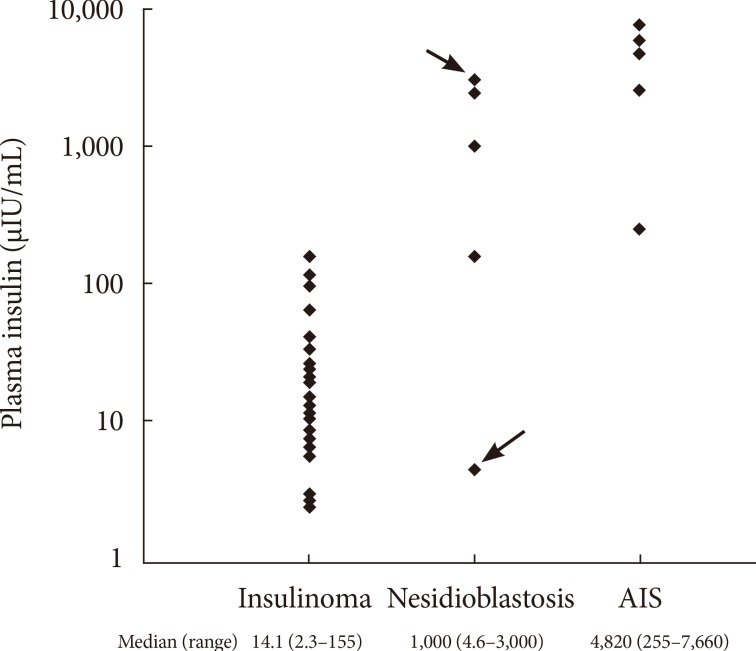
- As shown in Fig. 2, the median insulin levels were 14.1 µIU/mL (97.9 pmol/L) in patients with insulinoma, in comparison with 1,000.0 µIU/mL (6,945 pmol/L) and 4,820.0 µIU/mL (33,474.9 pmol/L) in patients with nesidioblastosis and IAS, respectively. The median insulin levels were markedly higher in patients with IAS and nesidioblastosis compared with insulinoma patients (P=0.002).
- The characteristics of the individual IAS and nesidioblastosis patients are shown in Table 2. Our center began to measure the levels of insulin autoantibody in 2007, which is reported as negative if the titer is <7%. After 2007, five patients were diagnosed with IAS and only one patient was diagnosed with nesidioblastosis. The examination of the stored samples from a patient who was diagnosed with nesidioblastosis in 2006 by surgical biopsy (case no. 9 in Table 2) were actually positive for insulin autoantibody.
RESULTS
- In this retrospective study, we determined that the incidence of noninsulinoma or IAS was higher than that previously reported in other studies [5,11]. The reason for the relatively high prevalence of noninsulinoma at our center is uncertain. Our hospital is a tertiary referral center, and this may have resulted in selection bias. Specifically, patients with negative localization results at other hospitals may have been transferred to our hospital. However, we cannot exclude the possibility that the incidence of noninsulinoma EHH/IAS in Korea is higher than in other countries, or that the incidence is higher than previously thought [7,8]. Comparisons with EHH patients treated at other centers in Korea and countries are needed to answer this question.
- IAS is caused by autoantibodies to insulin or the insulin receptor [8,12]. This syndrome is a relatively common cause of hypoglycemia in Japan but has rarely been reported in other populations [7]. As reported here, we identified five IAS patients after we started to check insulin autoantibodies in 2007. On the other hand, only one patient was diagnosed with nesidioblastosis during the same period.
- Consistent with previous studies [6], our current IAS patients demonstrated markedly higher plasma insulin levels than insulinoma patients. The binding of endogenous insulin to antibodies may decrease the level of bioavailable insulin. This may cause hyperglycemia, but in subjects whose pancreatic β-cell function is intact, insulin is over-secreted to compensate for the decreased bioavailability of insulin. In some instances, insulin bound to antibodies is subsequently released, resulting in inappropriately high free insulin concentrations and causing hypoglycemia [6,13]. Insulin secreted immediately after a meal may be bound to autoantibodies and may cause early postprandial hyperglycemia and exaggerated insulin secretion from the pancreas. If insulin is then dissolved from the complex, this may cause late postprandial hypoglycemia [6,14]. In our series of five IAS patients, this phenomenon was observed in two patients. However, as other IAS patients presented with fasting hypoglycemia, this may not apply to all IAS patients. Although 42.9% of insulinoma patients (three of seven patients) also developed postprandial hypoglycemia, 75% of noninsulinoma patients (three of four patients) developed postprandial hypoglycemia during OGTT (Table 1). Interestingly, the plasma insulin level of the patient with nesidioblastosis who had hypoglycemia during OGTT was 2,719 µIU/mL, which is markedly higher than the highest insulin level observed in insulinoma.
- Nesidioblastosis is a common entity in neonates with EHH, but is considered very rare in adults, representing only approximately 0.5% of EHH cases [15,16,17,18]. Nesidioblastosis is frequently found in patients who have undergone gastrectomy [4,19,20]. However, none of the patients with nesidioblastosis identified in this series had a history of gastric surgery. It should be noted that the number of patients diagnosed with nesidioblastosis decreased after our center began measuring insulin autoantibody levels. One patient was diagnosed with nesidioblastosis by surgical biopsy in 2006 but had positive insulin autoantibody results. Although hyperplasia of the islet β-cells was previously reported in some IAS patients [21], there have been no case(s) reporting nesidioblastosis and IAS in a patient. Therefore, in the case of this patient, IAS was most likely misdiagnosed as nesidioblastosis.
- Among the four remaining cases of nesidioblastosis, three patients demonstrated notably higher plasma insulin levels (Fig. 2). Although insulin autoantibody levels were not measured in these patients, it is also possible that these patients may have had IAS. This assumption is supported by previous studies showing modest hyperinsulinemia in nesidioblastosis [4,5].
- In our current series, all patients with insulinomas demonstrated hypoglycemia on prolonged fasting tests. On the other hand, only three of seven noninsulinoma patients demonstrated hypoglycemia during the fasting test. Thus, if hypoglycemia is not confirmed by 72-hour fasting test, noninsulinoma can be considered an alternative cause of EHH. In support of this, a previous study performed in the United States reported that 19 of 44 patients with IAS demonstrated postprandial hypoglycemia, but not fasting hypoglycemia [6].
- Our present study has obvious limitations, in that all data were retrospectively collected. However, to the best of our knowledge, this study is the first to report that the prevalence of IAS may be higher in Korea than previously thought. Among EHH patients whose tumors cannot be easily localized, and particularly among patients who show inappropriately high plasma insulin levels, the measurement of insulin autoantibody is recommended.
DISCUSSION
-
CONFLICTS OF INTEREST: No potential conflict of interest relevant to this article was reported.
NOTES
- 1. Service FJ. Diagnostic approach to adults with hypoglycemic disorders. Endocrinol Metab Clin North Am 1999;28:519-532. ArticlePubMed
- 2. Grant CS. Insulinoma. Best Pract Res Clin Gastroenterol 2005;19:783-798. ArticlePubMed
- 3. Service FJ. Hypoglycemic disorders. N Engl J Med 1995;332:1144-1152. ArticlePubMed
- 4. Service GJ, Thompson GB, Service FJ, Andrews JC, Collazo-Clavell ML, Lloyd RV. Hyperinsulinemic hypoglycemia with nesidioblastosis after gastric-bypass surgery. N Engl J Med 2005;353:249-254. ArticlePubMed
- 5. Anlauf M, Wieben D, Perren A, Sipos B, Komminoth P, Raffel A, Kruse ML, Fottner C, Knoefel WT, Monig H, Heitz PU, Kloppel G. Persistent hyperinsulinemic hypoglycemia in 15 adults with diffuse nesidioblastosis: diagnostic criteria, incidence, and characterization of beta-cell changes. Am J Surg Pathol 2005;29:524-533. PubMed
- 6. Lupsa BC, Chong AY, Cochran EK, Soos MA, Semple RK, Gorden P. Autoimmune forms of hypoglycemia. Medicine (Baltimore) 2009;88:141-153. ArticlePubMed
- 7. Uchigata Y, Hirata Y, Iwamoto Y. Insulin autoimmune syndrome (Hirata disease): epidemiology in Asia, including Japan. Diabetol Int 2010;1:21-25.ArticlePDF
- 8. Uchigata Y, Eguchi Y, Takayama-Hasumi S, Omori Y. Insulin autoimmune syndrome (Hirata disease): clinical features and epidemiology in Japan. Diabetes Res Clin Pract 1994;22:89-94. ArticlePubMed
- 9. Park HJ, Park JY, Shong YK, Hong SK, Lee KU, Han DJ, Sung KB, Kim GS. Clinical manifestation of 16 cases with insulinoma or nesidioblastosis. Korean J Med 1999;56:189-195.
- 10. Rindi G, Solcia E. Endocrine hyperplasia and dysplasia in the pathogenesis of gastrointestinal and pancreatic endocrine tumors. Gastroenterol Clin North Am 2007;36:851-865. ArticlePubMed
- 11. Raffel A, Krausch MM, Anlauf M, Wieben D, Braunstein S, Kloppel G, Roher HD, Knoefel WT. Diffuse nesidioblastosis as a cause of hyperinsulinemic hypoglycemia in adults: a diagnostic and therapeutic challenge. Surgery 2007;141:179-184. ArticlePubMed
- 12. Virally ML, Guillausseau PJ. Hypoglycemia in adults. Diabetes Metab 1999;25:477-490. PubMed
- 13. Redmon JB, Nuttall FQ. Autoimmune hypoglycemia. Endocrinol Metab Clin North Am 1999;28:603-618. ArticlePubMed
- 14. Ma WY, Won JG, Tang KT, Lin HD. Severe hypoglycemic coma due to insulin autoimmune syndrome. J Chin Med Assoc 2005;68:82-86. ArticlePubMed
- 15. Kaczirek K, Niederle B. Nesidioblastosis: an old term and a new understanding. World J Surg 2004;28:1227-1230. ArticlePubMedPDF
- 16. McHenry C, Newell K, Chejfec G, Barbato A, Lawrence AM, Brooks M, Emanuele MA, Paloyan E. Adult nesidioblastosis. An unusual cause of fasting hypoglycemia. Am Surg 1989;55:366-369. PubMed
- 17. Fong TL, Warner NE, Kumar D. Pancreatic nesidioblastosis in adults. Diabetes Care 1989;12:108-114. ArticlePubMedPDF
- 18. Jabri AL, Bayard C. Nesidioblastosis associated with hyperinsulinemic hypoglycemia in adults: review of the literature. Eur J Intern Med 2004;15:407-410. ArticlePubMed
- 19. Patti ME, Goldfine AB. Hypoglycaemia following gastric bypass surgery: diabetes remission in the extreme? Diabetologia 2010;53:2276-2279. ArticlePubMedPDF
- 20. Goldfine AB, Mun EC, Devine E, Bernier R, Baz-Hecht M, Jones DB, Schneider BE, Holst JJ, Patti ME. Patients with neuroglycopenia after gastric bypass surgery have exaggerated incretin and insulin secretory responses to a mixed meal. J Clin Endocrinol Metab 2007;92:4678-4685. ArticlePubMed
- 21. Uchigata Y, Hirata Y. Insulin autoimmune syndrome (IAS, Hirata disease). Ann Med Interne (Paris) 1999;150:245-253. PubMed
REFERENCES
Flow diagram of the localization strategies used to identify endogenous hyperinsulinemic hypoglycemia (EHH) patients. OGTT, oral glucose tolerance test; CT, computed tomography; MRI, magnetic resonance imaging; EUS, endoscopic ultrasound; THPVS, trans-hepatic portal vein sampling; SACS, selective arterial calcium stimulation.

Plasma insulin levels in patients diagnosed with insulinoma, nesidioblastosis, and insulin autoimmune syndrome (IAS). The case indicted by the arrow demonstrated positive results on the insulin autoantibody test (3,000 µIU/mL, case no. 9 in Table 2). The other case indicated by the head of arrow demonstrated negative results on the insulin autoantibody test (5 µIU/mL, case no. 10 in Table 2).
Clinical and biochemical characteristics of patients with insulinoma, nesidioblastosis, and insulin autoimmune syndrome
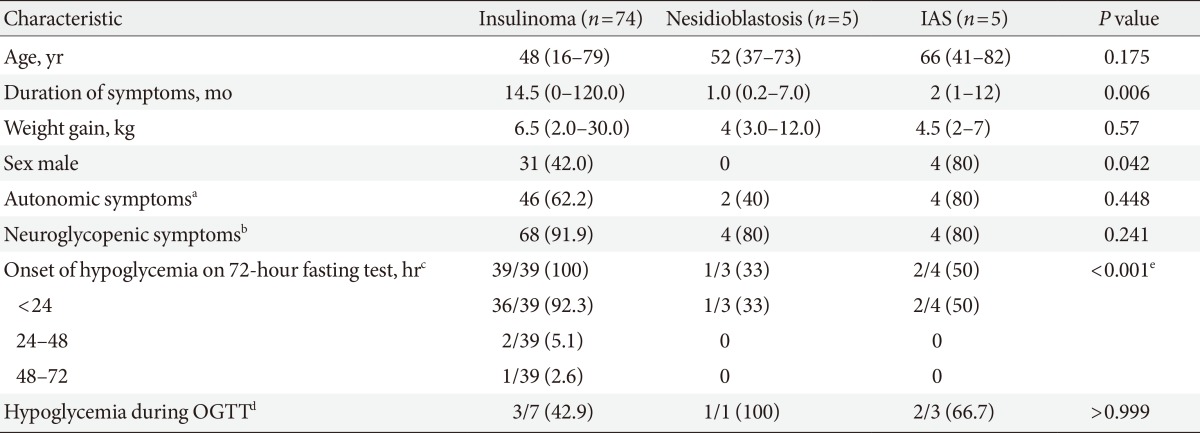
Values are presented as median (range) or number (%).
IAS, insulin autoimmune syndrome; OGTT, oral glucose tolerance test.
aNumber of patients who developed autonomic hypoglycemic symptoms (e.g., sweating, palpitation, anxiety, hunger, nausea, and paresthesia), bNumber of patients who developed neuroglycopenic hypoglycemic symptoms (e.g., loss of consciousness, disorientation, fatigue, seizure, hypersomnia, blurred vision, and change in behavior), cNumber of patients who developed hypoglycemia on 72-hour fasting test/total number of patients who received a 72-hour fasting test, dNumber of patients who developed hypoglycemia during OGTT/total number of patients who underwent OGTT, eStatistically significant development of hypoglycemia on 72-hour fasting test.
Figure & Data
References
Citations

- Case report: Insulinomatosis: description of four sporadic cases and review of the literature
Delmar Muniz Lourenço, Maria Lucia Corrêa-Giannella, Sheila Aparecida Coelho Siqueira, Marcia Nery, Flavio Galvão Ribeiro, Elizangela Pereira de Souza Quedas, Manoel de Souza Rocha, Ramon Marcelino do Nascimento, Maria Adelaide Albergaria Pereira
Frontiers in Endocrinology.2024;[Epub] CrossRef - Insulin Autoimmune Syndrome: A Systematic Review
MingXu Lin, YuHua Chen, Jie Ning, Tatsuya Kin
International Journal of Endocrinology.2023; 2023: 1. CrossRef - Diffuse, Adult-Onset Nesidioblastosis/Non-Insulinoma Pancreatogenous Hypoglycemia Syndrome (NIPHS): Review of the Literature of a Rare Cause of Hyperinsulinemic Hypoglycemia
Martin Philipp Dieterle, Ayman Husari, Sophie Nicole Prozmann, Hendrik Wiethoff, Albrecht Stenzinger, Manuel Röhrich, Uwe Pfeiffer, Wolfgang Rüdiger Kießling, Helena Engel, Harald Sourij, Thorsten Steinberg, Pascal Tomakidi, Stefan Kopf, Julia Szendroedi
Biomedicines.2023; 11(6): 1732. CrossRef - An Uncommon Cause of Recurrent Presyncope, Dizziness, and Tachycardia: A Case Report of Diffuse, Adult-Onset Nesidioblastosis/Non-Insulinoma Pancreatogenous Hypoglycemia Syndrome (NIPHS)
Martin Philipp Dieterle, Ayman Husari, Sophie Nicole Prozmann, Hendrik Wiethoff, Albrecht Stenzinger, Manuel Röhrich, Uwe Pfeiffer, Wolfgang Rüdiger Kießling, Helena Engel, Harald Sourij, Thorsten Steinberg, Pascal Tomakidi, Stefan Kopf, Julia Szendroedi
Biomedicines.2023; 11(6): 1741. CrossRef - An Uncommon Case of Recurrent Hypoglycemic Episodes in a Healthy Non-diabetic Male: Insulin Autoimmune Syndrome
Kanwarpal K Dhaliwal, Gaurav Bector, Saurabh Arora, Amanpreet Singh, Sanjay Kalra
Cureus.2023;[Epub] CrossRef - The After-Dinner Dip
Caren G. Solomon, Gertrud L.G. Haverkamp, Richard G. Ijzerman, Jos Kooter, Yvonne H.M. Krul-Poel
New England Journal of Medicine.2022; 386(22): 2130. CrossRef - Hirata's disease (insulin autoimmune syndrome) following envenomation by a common krait
Subramanian Senthilkumaran, Stephen W. Miller, Harry F. Williams, Ponniah Thirumalaikolundusubramanian, Sakthivel Vaiyapuri, Ketan Patel
Toxicon.2022; 219: 106923. CrossRef - Analysis of the clinical characteristics of insulin autoimmune syndrome induced by methimazole
Linli Sun, Weijin Fang, Dan Yi, Wei Sun, Chunjiang Wang
Journal of Clinical Pharmacy and Therapeutics.2021; 46(2): 470. CrossRef - Continuous glucose monitoring and Rituximab treatment in insulin autoimmune syndrome
Hiya Boro, Uttio Gupta, Charandeep Singh, Rakhi Malhotra, Rajesh Khadgawat
Diabetes & Metabolic Syndrome: Clinical Research & Reviews.2021; 15(6): 102294. CrossRef Insulin Autoimmune Syndrome (Hirata Disease): A Comprehensive Review Fifty Years After Its First Description
Daniele Cappellani, Enrico Macchia, Alberto Falorni, Piero Marchetti
Diabetes, Metabolic Syndrome and Obesity: Targets and Therapy.2020; Volume 13: 963. CrossRef- Nationwide survey of endogenous hyperinsulinemic hypoglycemia in Japan (2017–2018): Congenital hyperinsulinism, insulinoma, non‐insulinoma pancreatogenous hypoglycemia syndrome and insulin autoimmune syndrome (Hirata’s disease)
Yuki Yamada, Kana Kitayama, Maki Oyachi, Shinji Higuchi, Rie Kawakita, Yutaka Kanamori, Tohru Yorifuji
Journal of Diabetes Investigation.2020; 11(3): 554. CrossRef - Insulin Autoimmune Syndrome – A Case Series
Hiya Boro, Uttio Gupta, Charandeep Singh, Rakhi Malhotra, Rajesh Khadgawat
European Endocrinology.2020; 16(2): 168. CrossRef - Management of Insulin Autoimmune Hypoglycaemia: Single‐centre experience from Western India with systematic review of world literature
Muniraj Patel, Ravikumar Shah, Swati Ramteke‐Jadhav, Virendra Patil, Shivendra Kumar Patel, Anurag Lila, Nalini Shah, Tushar Bandgar
Clinical Endocrinology.2020; 92(5): 409. CrossRef - Is insulin intoxication still the perfect crime? Analysis and interpretation of postmortem insulin: review and perspectives in forensic toxicology
Charline Bottinelli, Nathalie Cartiser, Fabien Bévalot, Laurent Fanton, Jérôme Guitton
Critical Reviews in Toxicology.2020; 50(4): 324. CrossRef - Insulin autoimmune syndrome induced by exogenous insulin injection: a four-case series
Yimin Shen, Xiaoxiao Song, Yuezhong Ren
BMC Endocrine Disorders.2019;[Epub] CrossRef - An observational analysis of insulinoma from a single institution
S Shao, Z Zeng, S Hu
QJM: An International Journal of Medicine.2018; 111(4): 237. CrossRef - Anti-tuberculosis Treatment-Induced Insulin Autoimmune Syndrome
Jung Suk Han, Han Ju Moon, Jin Seo Kim, Hong Il Kim, Cheol Hyeon Kim, Min Joo Kim
The Ewha Medical Journal.2016; 39(4): 122. CrossRef - Spontaneous hypoglycemia: diagnostic evaluation and management
Leelavathy Kandaswamy, Rajeev Raghavan, Joseph M. Pappachan
Endocrine.2016; 53(1): 47. CrossRef - Hypoglycemia due to Insulin Autoimmune Syndrome: A rare cause not to be forgotten
Sarah Alam, Maaz Ozair, Jamal Ahmad
Journal of Clinical and Translational Endocrinology: Case Reports.2016; 2: 7. CrossRef

 KDA
KDA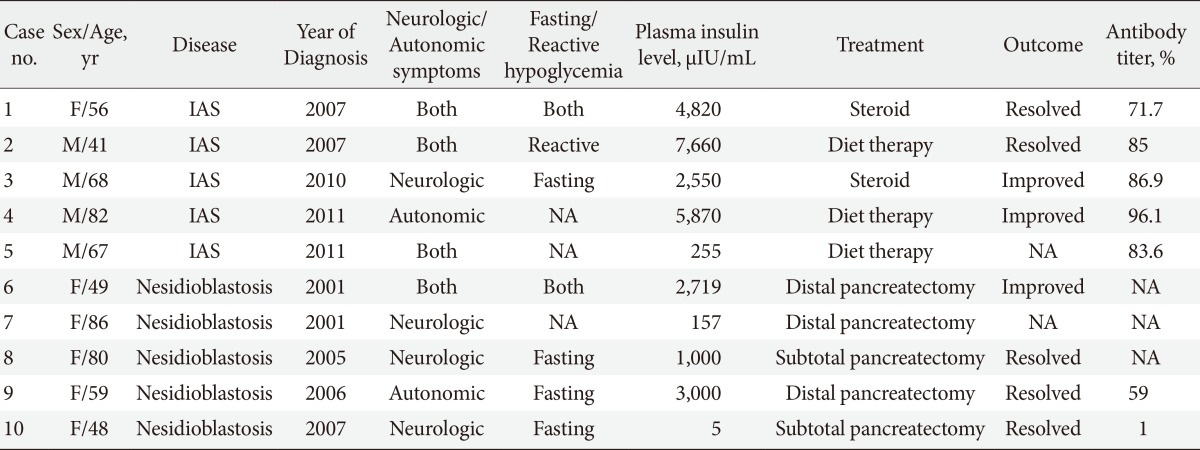
 PubReader
PubReader Cite
Cite