- Current
- Browse
- Collections
-
For contributors
- For Authors
- Instructions to authors
- Article processing charge
- e-submission
- For Reviewers
- Instructions for reviewers
- How to become a reviewer
- Best reviewers
- For Readers
- Readership
- Subscription
- Permission guidelines
- About
- Editorial policy
Articles
- Page Path
- HOME > Diabetes Metab J > Volume 45(3); 2021 > Article
-
Original ArticleDrug/Regimen Effect of Dapagliflozin as an Add-on Therapy to Insulin on the Glycemic Variability in Subjects with Type 2 Diabetes Mellitus (DIVE): A Multicenter, Placebo-Controlled, Double-Blind, Randomized Study
-
Seung-Hwan Lee1,2
 , Kyung-Wan Min3, Byung-Wan Lee4, In-Kyung Jeong5, Soon-Jib Yoo6, Hyuk-Sang Kwon7, Yoon-Hee Choi1,8, Kun-Ho Yoon1,2
, Kyung-Wan Min3, Byung-Wan Lee4, In-Kyung Jeong5, Soon-Jib Yoo6, Hyuk-Sang Kwon7, Yoon-Hee Choi1,8, Kun-Ho Yoon1,2
-
Diabetes & Metabolism Journal 2021;45(3):339-348.
DOI: https://doi.org/10.4093/dmj.2019.0203
Published online: May 28, 2020
1Division of Endocrinology and Metabolism, Department of Internal Medicine, Seoul St. Mary's Hospital, College of Medicine, The Catholic University of Korea, Seoul, Korea
2Department of Medical Informatics, College of Medicine, The Catholic University of Korea, Seoul, Korea
3Division of Endocrinology and Metabolism, Department of Internal Medicine, Eulji General Hospital, Eulji University School of Medicine, Seoul, Korea
4Division of Endocrinology and Metabolism, Department of Internal Medicine, Yonsei University College of Medicine, Seoul, Korea
5Department of Endocrinology and Metabolism, Kyung Hee University Hospital at Gangdong, Kyung Hee University School of Medicine, Seoul, Korea
6Division of Endocrinology and Metabolism, Department of Internal Medicine, Bucheon St. Mary's Hospital, College of Medicine, The Catholic University of Korea, Bucheon, Korea
7Division of Endocrinology and Metabolism, Department of Internal Medicine, Uijeongbu St. Mary's Hospital, College of Medicine, The Catholic University of Korea, Seoul, Korea
8MedicalExcellence Inc., Seoul, Korea
-
Corresponding author: Kun-Ho Yoon
 Department of Internal Medicine, Seoul St. Mary's Hospital, College of Medicine, The Catholic University of Korea, 222 Banpo-daero, Seocho-gu, Seoul 06591, Korea. yoonk@catholic.ac.kr
Department of Internal Medicine, Seoul St. Mary's Hospital, College of Medicine, The Catholic University of Korea, 222 Banpo-daero, Seocho-gu, Seoul 06591, Korea. yoonk@catholic.ac.kr
Copyright © 2021 Korean Diabetes Association
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/4.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
ABSTRACT
-
Background
- Glycemic variability is associated with the development of diabetic complications and hypoglycemia. However, the effect of sodium-glucose transporter 2 (SGLT2) inhibitors on glycemic variability is controversial. We aimed to examine the effect of dapagliflozin as an add-on therapy to insulin on the glycemic variability assessed using continuous glucose monitoring (CGM) in subjects with type 2 diabetes mellitus.
-
Methods
- In this multicenter, placebo-controlled, double-blind, randomized study, 84 subjects received 10 mg of dapagliflozin (n=41) or the placebo (n=43) for 12 weeks. CGM was performed before and after treatment to compare the changes in glycemic variability measures (standard deviation [SD], mean amplitude of glycemic excursions [MAGEs]).
-
Results
- At week 12, significant reductions in glycosylated hemoglobin (−0.74%±0.66% vs. 0.01%±0.65%, P<0.001), glycated albumin (−3.94%±2.55% vs. −0.67%±2.48%, P<0.001), and CGM-derived mean glucose (−41.6±39.2 mg/dL vs. 1.1±46.2 mg/dL, P<0.001) levels were observed in the dapagliflozin group compared with the placebo group. SD and MAGE were significantly decreased in the dapagliflozin group, but not in the placebo group. However, the difference in ΔSD and ΔMAGE failed to reach statistical significance between two groups. No significant differences in the incidence of safety endpoints were observed between the two groups.
-
Conclusion
- Dapagliflozin effectively decreased glucose levels, but not glucose variability, after 12 weeks of treatment in participants with type 2 diabetes mellitus receiving insulin treatment. The role of SGLT2 inhibitors in glycemic variability warrants further investigations.
- Glucose variability, which refers to oscillations or fluctuations in blood glucose levels, is associated with various adverse health outcomes [12]. Although certain degree of variability in biological parameters is a physiological phenomenon, glucose variability is often excessively increased in patients with diabetes and has been identified as an independent driver of diabetic complications and mortality [345678]. Therefore, strategies that reduce glucose variability are becoming an emerging treatment target for patients with diabetes, in addition to controlling fasting hyperglycemia, postprandial hyperglycemia and increased glycosylated hemoglobin (HbA1c) levels. Based on this information, researchers are focusing on whether a specific class of glucose-lowering agents also stabilize glucose fluctuations.
- Sodium-glucose transporter 2 (SGLT2) inhibitors, including dapagliflozin, empagliflozin, canagliflozin, ertugliflozin, and ipragliflozin, have been approved as a new class of glucose-lowering compounds for the treatment of type 2 diabetes mellitus [9]. Inhibition of SGLT2 in renal proximal tubules improves glucose homeostasis by reducing the maximum renal glucose reabsorptive capacity and threshold at which glucose is excreted in the urine [10]. The effect of SGLT2 inhibitors depends on the degree of hyperglycemia and renal function, and is characterized by an insulin-independent mode of action [11]. The risk of hypoglycemia is low due to the compensatory role of SGLT1 in the downstream late proximal tubule and counterregulatory mechanisms, including an increase in plasma glucagon levels and hepatic gluconeogenesis [121314]. Therefore, SGLT2 inhibitors would conceivably be effective in reducing glucose variability.
- Several published studies have examined the effects of SGLT2 inhibitors on glucose variability, in both patients with type 1 and type 2 diabetes mellitus [1516171819202122232425]. However, most of the studies were performed with a relatively small number of subjects and short-term treatment, producing conflicting results. In this study, we aimed to examine the effect of dapagliflozin as an add-on to insulin on the glycemic variability assessed using continuous glucose monitoring (CGM) in subjects with type 2 diabetes mellitus and inadequate glycemic control.
INTRODUCTION
- Compliance with ethical standards
- The study protocol was approved by Institutional Review Board of each participating center (Seoul St. Mary's Hospital: XC14MIMV0103K), and all subjects provided written informed consent before participation. This study was conducted in accordance with the ethical principles of the Declaration of Helsinki.
- Study design and subjects
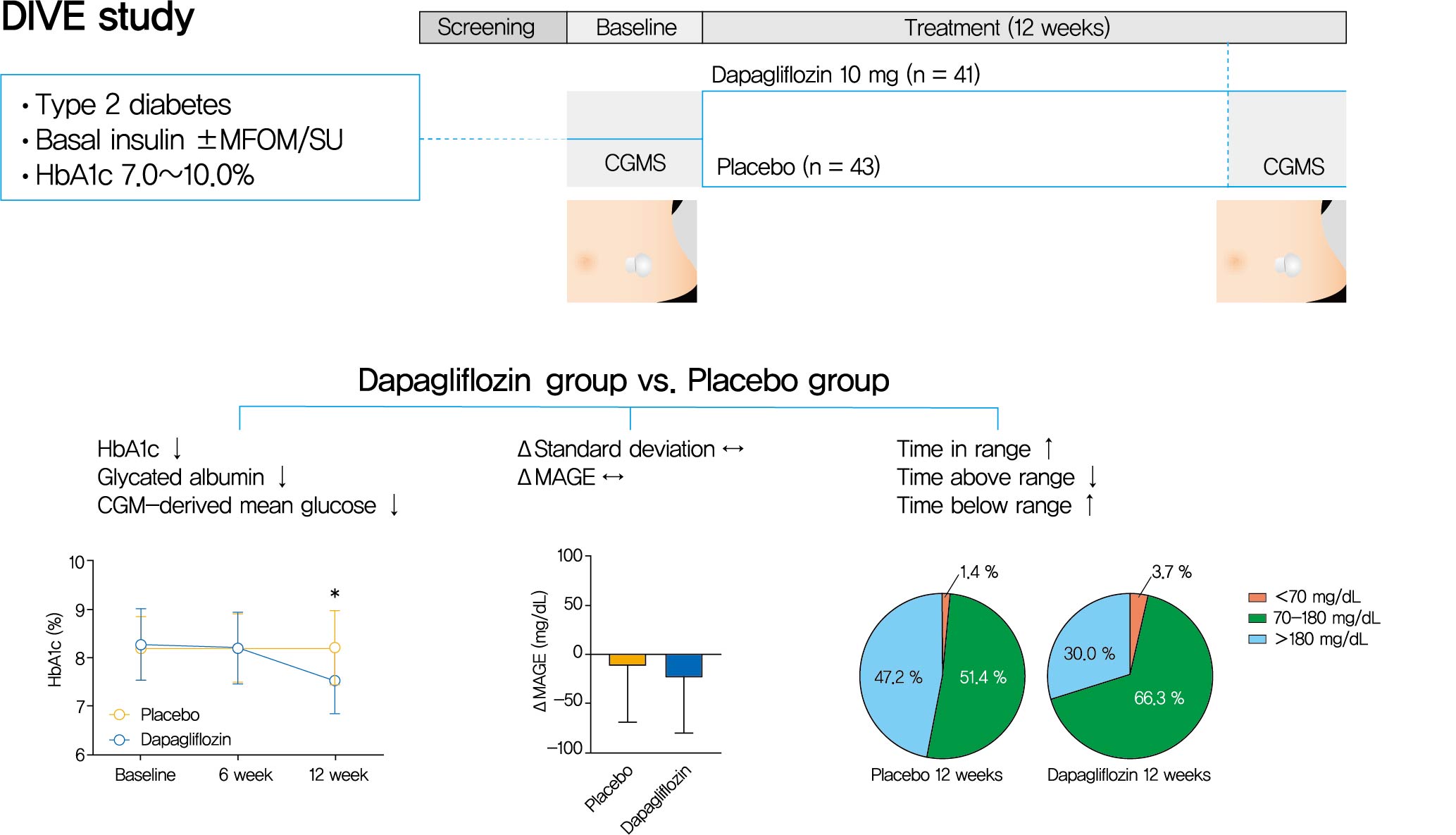
- This multicenter, placebo-controlled, double-blind, randomized clinical trial (ClinicalTrials.gov identifier: NCT02459353) was conducted at six centers in South Korea between August 2015 and December 2016. Eligible participants were patients with type 2 diabetes mellitus who were receiving greater than 0.2 IU/kg/day of basal insulin±metformin or sulfonylurea for at least 12 weeks. Patients aged 20 to 70 years with an HbA1c level of 7.0% to 10.0% at screening were recruited. Participants were randomized to receive 10 mg of dapagliflozin or placebo for 12 weeks. Randomization was stratified based on an HbA1c level of 8.0% and the study site. The maintenance of the same dose of insulin and oral hypoglycemic agents during the study period was recommended, although insulin could be titrated in the event of hypoglycemia or hyperglycemia and based on the physician's discretion. The key exclusion criteria included people with type 1 diabetes mellitus (fasting C-peptide ≤0.6 ng/dL), secondary diabetes or gestational diabetes; use of insulin other than basal insulin; a history of diabetic ketoacidosis or hyperglycemic hyperosmolar state; an estimated glomerular filtration rate <60 mL/min/1.73 m2; abnormal liver function as defined by aspartate aminotransferase (AST)/alanine aminotransferase (ALT) >3 times the upper normal limit; a history of chronic cystitis or recurrent urinary tract infection; use of loop diuretics or medications known to affect glucose metabolism; a history of malignancy within 5 years; a history of acute coronary syndrome, stroke or coronary artery bypass graft within 6 months; New York Heart Association class III or IV congestive heart failure; and participation in a weight loss program or use of anti-obesity medications.
- Continuous glucose monitoring
- CGM was performed two times, before randomization and after the completion of 12 weeks treatment, using the Minimed CGM System Gold or iPro2 (Medtronic, Northridge, CA, USA). Glucose data were obtained for 72 hours, and the mean amplitude of glycemic excursions (MAGE) and standard deviation (SD) of glucose were calculated. The sensor was inserted on day 1 and removed on day 4 at both visits. We also calculated MAGE and SD over 48 hours, from 00:00 hours on the second day of CGM sensor insertion to 24:00 hours on the third day. MAGE was defined as the mean of the absolute difference from nadir to peak or from peak to nadir for those differences that exceeded the SD [26]. For the efficacy variables derived from CGM, analyses using 48 hour data are mainly presented. Analyses using 72 hour data produced similar results (data not shown).
- Efficacy and safety assessments
- The primary endpoint was to evaluate the effect of dapagliflozin on glucose variability compared to the placebo after 12 weeks of treatment. The extent of changes in MAGE and SD were compared between the two groups. Secondary endpoints included a comparison of glycemic control variables (fasting plasma glucose, HbA1c, glycated albumin, and mean glucose levels, as well as the time-in-target range [70 to 180 mg/dL] obtained from CGM), blood pressure, lipid profiles, body weight and changes in body mass index (BMI) between two groups. Safety and tolerability were also assessed from overall adverse events, serious adverse events and adverse events of special interest, which included hypoglycemia, severe hypoglycemia, genital infection, urinary tract infection, dehydration and hypovolemia. Severe hypoglycemia was defined as hypoglycemic events requiring assistance. Anthropometric and laboratory data were collected at baseline and 6 and 12 weeks after treatment.
- Statistical analyses
- The sample size was calculated by assuming that the mean MAGE difference between the dapagliflozin group and placebo group would be at least 20 mg/dL (SD=30 mg/dL). Approximately 37 subjects in each group were needed to detect a significant difference with a power of 0.8 and statistical significance of 0.05 (α). Considering a 20% drop-out rate, the target enrollment number was 90 subjects. The intention-to-treat (ITT) population included all randomized patients who received at least one dose of the study drug. The per-protocol (PP) population was defined as a subset of the ITT population who completed the study without any major protocol violations. The efficacy analyses were performed both on ITT and PP populations. The last observation carried forward imputation technique was used to handle missing values. The PP population was analyzed for the presentation of efficacy variables derived from CGM. The safety analyses were performed on the ITT population. Data are presented as mean±SD or numbers (%). The independent t-test or Wilcoxon rank sum test were used to compare of continuous variables, and the chi-square or Fisher's exact test were used to compare categorical variables, as appropriate. A paired t-test or Wilcoxon signed rank test was also applied to compare pre- and post-treatment values. A P value <0.05 was considered statistically significant, and all analyses were performed using SAS software version 9.3 (SAS Institute, Cary, NC, USA).
METHODS
- Participant disposition and characteristics
- One hundred seventeen subjects were screened and 84 were randomized (41 to the dapagliflozin group and 43 to the placebo group). Of the randomized subjects, 66 completed the study (Supplementary Fig. 1). Baseline demographics and efficacy parameters were generally balanced between the treatment groups (Table 1). The mean age and BMI were 57.7 years and 26.6 kg/m2 in the placebo group, and 59.7 years and 27.3 kg/m2 in the dapagliflozin group, respectively. The mean duration of diabetes was 15.1 years in both groups. No significant differences in fasting glucose (137.4±53.5 mg/dL vs. 145.6±56.4 mg/dL), HbA1c (8.25%±0.69% vs. 8.30%±0.77%) and fasting C-peptide (1.78±1.27 ng/dL vs. 1.60±1.19 ng/dL) levels were observed between the placebo and dapagliflozin groups. Only systolic BP and background medication differed between two groups.
- Efficacy
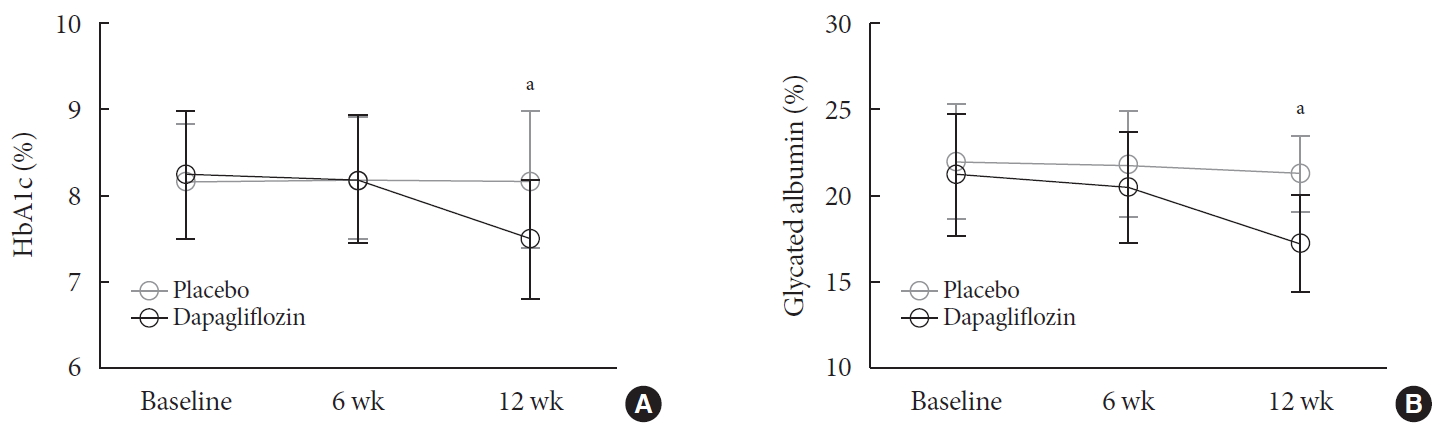
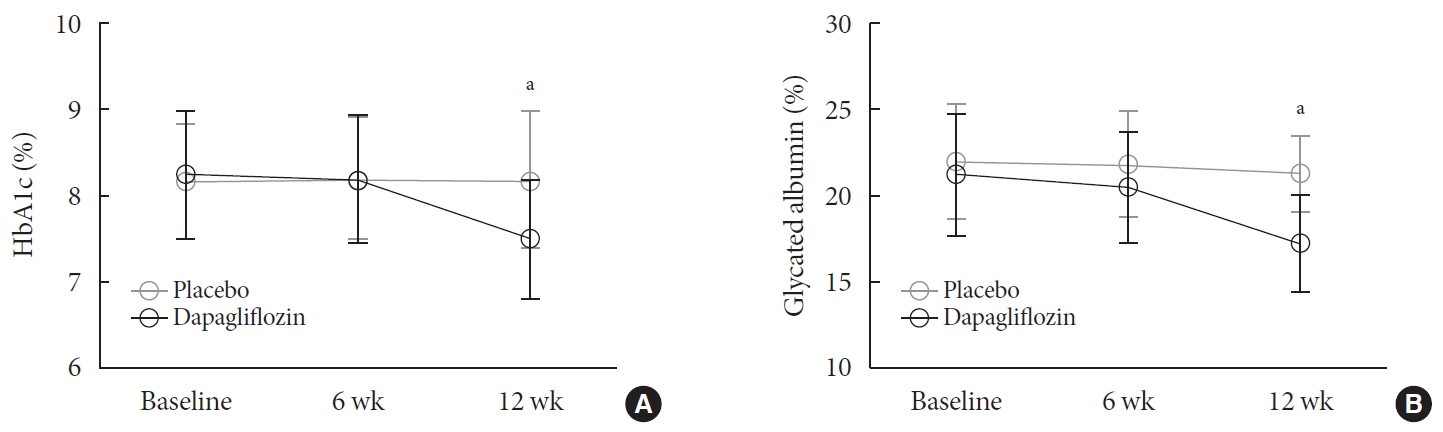
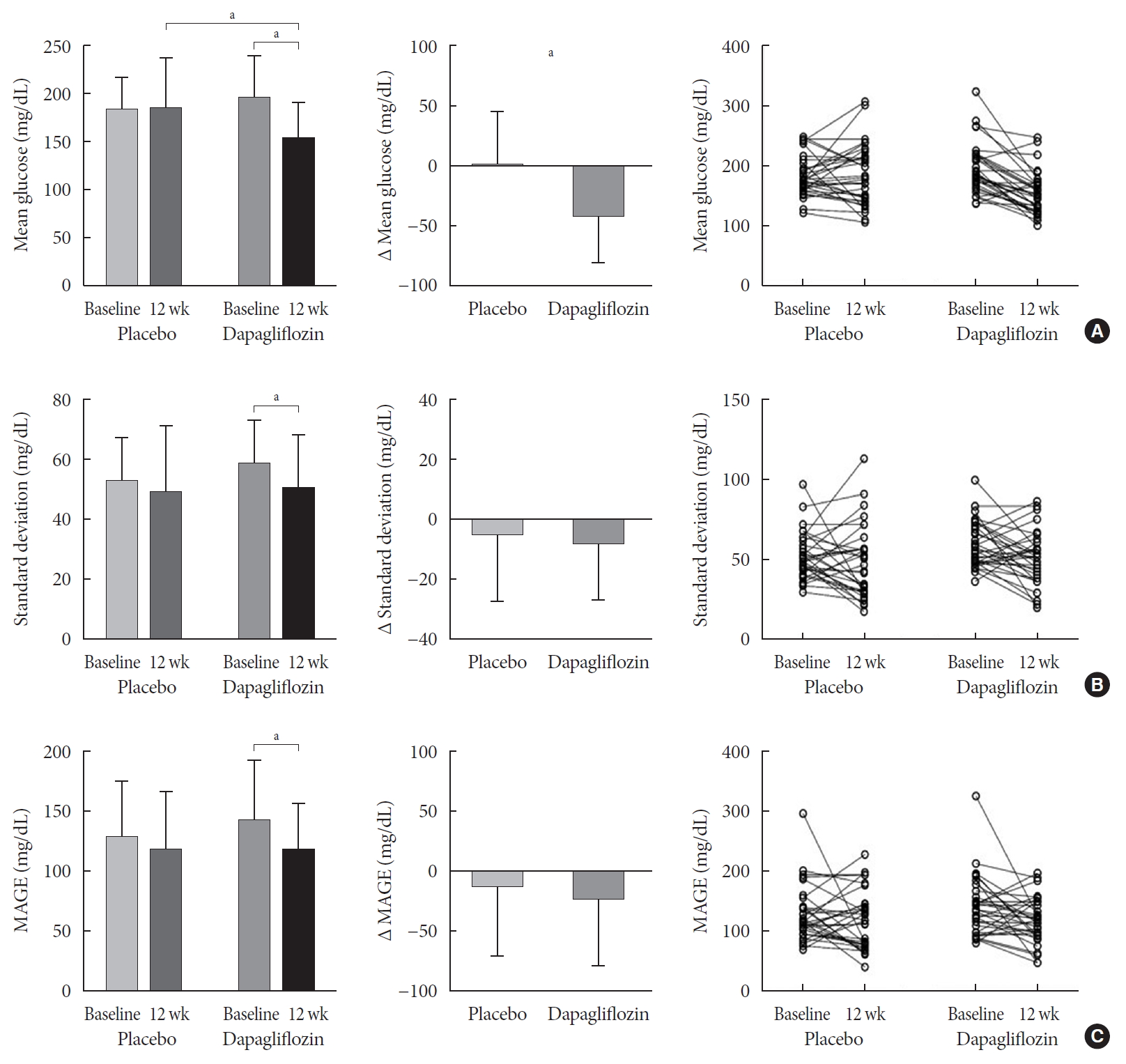
- The 24 hours urine glucose excretion was significantly increased in the dapagliflozin group (Supplementary Fig. 2). After 12 weeks of treatment, the mean changes in the HbA1c (−0.74%±0.66% vs. 0.01%±0.65%, P<0.001) and glycated albumin (−3.94%±2.55% vs. −0.67%±2.48%, P<0.001) levels from the baseline values were significantly greater in the group treated with dapagliflozin compared with the placebo (Fig. 1). Fasting glucose levels were significantly decreased by the dapagliflozin treatment, but not the placebo (Table 2). The 48 hours mean glucose levels derived from CGM were also significantly decreased in the dapagliflozin group, but not in the placebo group (−41.6±39.2 mg/dL vs. 1.1±46.2 mg/dL, P<0.001) (Fig. 2A). Glucose variability indices, SD and MAGE, were significantly decreased by dapagliflozin after 12 weeks of treatment compared with baseline values. However, ΔSD (−7.64± 18.75 mg/dL vs. −4.09±22.62 mg/dL, P=0.494) and ΔMAGE (−23.09±55.18 mg/dL vs. −11.34±57.61 mg/dL, P=0.341) were not significantly different between the dapagliflozin and placebo groups (Fig. 2B and C). Analyses of groups stratified by baseline HbA1c levels (8%), BMI (25 kg/m2), duration of diabetes (10 years), insulin dose (0.6 IU/kg), median value of homeostasis model assessment of insulin resistance or beta-cell function, and the presence or absence of hypoglycemia identified using CGM revealed similar results in all subgroups (data not shown). Analyses using the ITT dataset also produced similar results (data not shown). In the dapagliflozin group, subjects with improved glucose variability were characterized by a lower BMI than subjects with increased variability (26.5±3.4 kg/m2 vs. 29.4±4.1 kg/m2, P=0.044).
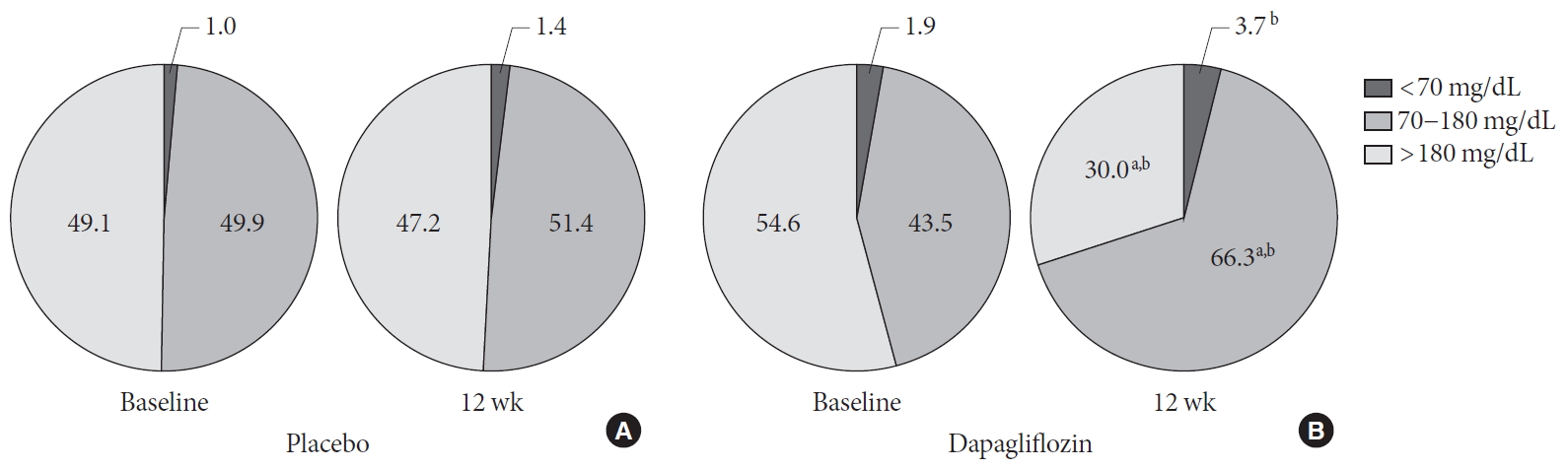
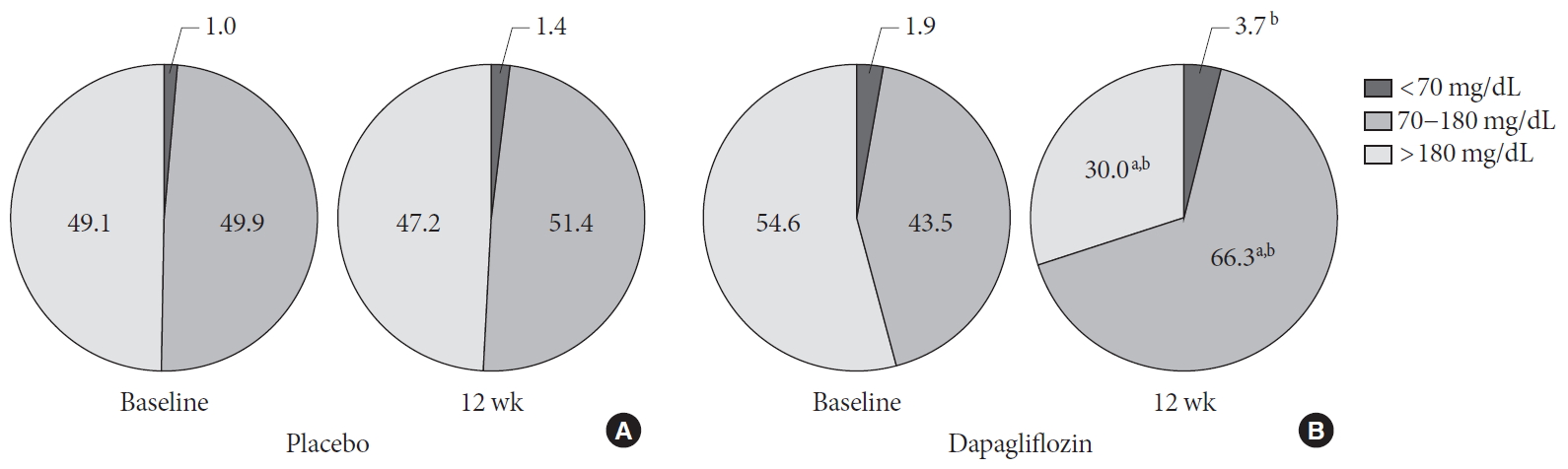
- Compared with baseline values, the mean percentage of time in which CGM glucose values were within the target range (70 to 180 mg/dL) increased, whereas time within the hyperglycemic range (>180 mg/dL) decreased following the dapagliflozin treatment. No significant changes were noted in the placebo group. At 12 weeks after the treatment, the dapagliflozin group showed higher percentage of time within the target range and with hypoglycemic range, but a lower percentage of time within the hyperglycemic range, than placebo group (Fig. 3).
- In the dapagliflozin group, body weight, BMI, insulin dose, and uric acid, AST, and ALT levels were significantly decreased at 12 weeks compared with the baseline values. Furthermore, uric acid and ALT levels were significantly reduced by the dapagliflozin treatment compared with the placebo. A subtle but significant decrease in eGFR levels was observed in the dapagliflozin group. No significant changes were detected in the blood pressure and lipid profiles between the two groups or before and after treatment (Table 2).
- Safety
- Measures of adverse events were generally similar between groups. The percentage of subjects experiencing one or more adverse events was 17.1% in the dapagliflozin group and 11.6% in the placebo group (P=0.476) (Supplementary Table 1). No significant difference in the occurrence of hypoglycemia was noted between two groups, and no severe hypoglycemic episodes occurred during the study (Supplementary Table 2).
RESULTS
- In this randomized, placebo-controlled trial, we tested whether 12 weeks of treatment with dapagliflozin improved glucose variability in patients with type 2 diabetes mellitus and inadequate glycemic control who were receiving insulin. The dapagliflozin treatment significantly decreased the fasting glucose, HbA1c, glycated albumin, and mean glucose levels obtained from CGM, and increased the time-in-target range. However, changes in the degree of glucose variability were insignificant compared with the placebo group.
- Glucose variability and HbA1c variability, which reflect short-term and long-term glycemic fluctuation, respectively, have been linked to various complications and mortality in both subjects with type 1 and type 2 diabetes mellitus [45678]. An analysis of glucose and HbA1c data obtained from the Action in Diabetes and Vascular Disease: Preterax and Diamicron MR Controlled Evaluation (ADVANCE) trial revealed that visit-to-visit glucose variability was associated with both micro- and macrovascular events and HbA1c variability was associated with both vascular events and mortality [4]. A cohort study in China showed an independent association between fasting plasma glucose variability and all-cause mortality in patients with type 2 diabetes mellitus and poor glycemic control [7]. A meta-analysis of seven studies of patients with type 1 diabetes mellitus and thirteen studies of patients with type 2 diabetes mellitus also confirmed that HbA1c variability represents a possible independent risk factor for micro- and macrovascular complications and mortality. Although most previous studies employed a retrospective design, and intentional control of glucose variability is difficult to achieve in a prospective study, the consistency observed from multiple studies strengthens the relationship between glucose variability and adverse health outcomes. Mechanistically, apoptosis was increased in human umbilical vein endothelial cells exposed to alternating 5 or 20 mmol/L glucose which was more deleterious than constant exposure to high glucose [27]. This difference was associated with elevated levels of nitrotyrosine and 8-hydroxydeoxyguanosine, suggesting that glucose fluctuations induce oxidative stress [28]. In humans, 24-hour urinary excretion rates of free 8-iso prostaglandin F2α, another estimate of oxidative stress, were significantly correlated with MAGE after adjustment for multiple factors [29]. Collectively, data from clinical trials and in vitro studies clearly emphasize the importance of targeting glucose variability for achieving better outcomes in patients with diabetes.
- From this perspective, the effects of different classes of antidiabetic agents on glucose variability have been studied in various clinical trials. Glucagon-like peptide-1 receptor agonists and dipeptidyl peptidase-4 (DPP-4) inhibitors, which act in a glucose-dependent manner, or α-glucosidase inhibitors and prandial insulins, which lowers postprandial glucose levels have shown to be more effective in stabilizing glucose fluctuations than other agents, such as sulfonylurea or basal insulin [330313233]. A clear conclusion regarding the effect of SGLT2 inhibitors on glucose variability appears to be premature. In patients with type 1 diabetes mellitus, short-term treatment with empagliflozin or sotagliflozin as an add-on to insulin improves glucose variability [1820]. Eighteen to 24 weeks of treatment with canagliflozin or sotagliflozin also improve the time-in-range measured using CGM [2325]. However, an exploratory study in which patients were treated with dapagliflozin for 2 weeks failed to show a significant difference in changes in SD or MAGE compared with the placebo, although a dose-dependent reduction in glucose variability was observed [15]. In patients with type 2 diabetes mellitus, short-term treatment with empagliflozin or luseogliflozin as a monotherapy improves mean glucose levels or time-in-range, but a significant difference in glucose variability indices is not observed compared with the placebo [1617]. A single-arm study in which canagliflozin was added to insulin therapy revealed reductions in SD, MAGE, and the mean of the daily difference in blood glucose levels (MODD), whereas switching from a DPP-4 inhibitor to dapagliflozin had no effect on reducing glucose variability in patients with type 2 diabetes mellitus receiving insulin [2122]. A recent study evaluating the effect of a 4 weeks dapagliflozin treatment showed improvements in glucose variability when dapagliflozin was added to metformin monotherapy, but not when added to insulin therapy [24]. Our results are consistent with some of the previous studies [1524] showing that the extent to which dapagliflozin decreases glucose variability is insignificant compared with the placebo, although changes compared with the baseline values are observed in patients when this treatment is added to insulin treatment.
- Various explanations may account for the inconsistent results, including differences in characteristics of study subjects, background medications, treatment duration, type of comparator, and the selectivity of SGLT2 inhibitors. In particular, when SGLT2 inhibitors are used in combination with insulin or sulfonylurea, the risk of hypoglycemia is potentially increased. In a study examining the kinetics of renal glucose transport using hyperglycemic clamp, empagliflozin treatment reduced maximal renal glucose transport and the plasma glucose concentration threshold for glucose spillage in the urine to <40 mg/dL in both healthy individuals and patients with type 2 diabetes mellitus [34]. Based on this finding, SGLT2 inhibitors might induce renal glucose excretion at levels less than the normal plasma glucose concentration. Another study using the hyperglycemic clamp in patients with type 2 diabetes mellitus reported a proportional increase in renal glucose excretion with increasing blood glucose levels. Glucosuria persisted when the blood glucose concentration decreased to the euglycemic level, suggesting that the classical concept of a renal threshold for glucose excretion is not supported in subjects with diabetes [35]. Notably, we and other groups showed a slight increase in the hypoglycemic range in the CGM analysis over time in patients treated with SGLT2 inhibitors, which might exert a negative effect on glucose variability [24]. Researchers have proposed that the actions of SGLT2 inhibitors are mitigated by compensatory glucose reabsorption by SGLT1, glucagon secretion from pancreatic alpha cells and increased endogenous glucose production, which might exert positive effects on reducing the hypoglycemia risk. Further investigations are needed to determine whether these findings are class effects.
- The strength of this study is that we recruited a large number of subjects compared to previous studies examining the effects of SGLT2 inhibitors on glucose variability in patients with type 2 diabetes mellitus receiving insulin. However, one important limitation is that we were unable to introduce a standardized diet during CGM because this study was performed in an ambulatory care setting, which might have exaggerated the glucose variability.
- In conclusion, the addition of dapagliflozin therapy to insulin in subjects with type 2 diabetes mellitus and inadequate glycemic control was effective in lowering glucose levels and generally tolerable, consistent with previous trials. Although reductions in glucose variability parameters were observed within the dapagliflozin group, the values were not significantly different from the placebo group. Larger clinical trials employing a diet-controlled CGM analysis would be helpful to better characterize the relationship between SGLT2 inhibitors and glucose variability.
DISCUSSION
-
Acknowledgements
- None
ACKNOWLEDGMENTS
-
CONFLICTS OF INTEREST
This work was supported by an AstraZeneca Ltd. investigator initiated study grant. The funder had no role in study design, data collection and analysis, interpretation of the data, decision to publish, or preparation of the manuscript. The funder had the opportunity to review the final manuscript prior to submission. The corresponding author had full access to all the data in the study and final responsibility for the decision to submit for publication
-
AUTHOR CONTRIBUTIONS
Conception or design: S.H.L., K.H.Y.
Acquisition, analysis, or interpretation of data: S.H.L., K.W.M., B.W.L., I.K.J., S.J.Y., H.S.K., Y.H.C., K.H.Y.
Drafting the work or revising: S.H.L.
Final approval of the manuscript: S.H.L., K.W.M., B.W.L., I.K.J., S.J.Y., H.S.K., Y.H.C., K.H.Y.
-
FUNDING
None
NOTES
SUPPLEMENTARY MATERIALS
Supplementary Fig. 2
- 1. Suh S, Kim JH. Glycemic variability: how do we measure it and why is it important? Diabetes Metab J 2015;39:273-82.ArticlePubMedPMC
- 2. Jung HS. Clinical implications of glucose variability: chronic complications of diabetes. Endocrinol Metab (Seoul) 2015;30:167-74.ArticlePubMedPMC
- 3. Umpierrez GE, P Kovatchev B. Glycemic variability: how to measure and its clinical implication for type 2 diabetes. Am J Med Sci 2018;356:518-27.ArticlePubMedPMC
- 4. Hirakawa Y, Arima H, Zoungas S, Ninomiya T, Cooper M, Hamet P, et al. Impact of visit-to-visit glycemic variability on the risks of macrovascular and microvascular events and all-cause mortality in type 2 diabetes: the ADVANCE trial. Diabetes Care 2014;37:2359-65.ArticlePubMedPDF
- 5. Gorst C, Kwok CS, Aslam S, Buchan I, Kontopantelis E, Myint PK, et al. Long-term glycemic variability and risk of adverse outcomes: a systematic review and meta-analysis. Diabetes Care 2015;38:2354-69.ArticlePubMedPDF
- 6. Hirsch IB. Glycemic variability and diabetes complications: does it matter? Of course it does! Diabetes Care 2015;38:1610-4.ArticlePubMedPDF
- 7. Xu D, Fang H, Xu W, Yan Y, Liu Y, Yao B. Fasting plasma glucose variability and all-cause mortality among type 2 diabetes patients: a dynamic cohort study in Shanghai, China. Sci Rep 2016;6:39633.ArticlePubMedPMCPDF
- 8. Cardoso CRL, Leite NC, Moram CBM, Salles GF. Long-term visit-to-visit glycemic variability as predictor of micro- and macrovascular complications in patients with type 2 diabetes: the Rio de Janeiro Type 2 Diabetes Cohort Study. Cardiovasc Diabetol 2018;17:33.ArticlePubMedPMCPDF
- 9. Chao EC, Henry RR. SGLT2 inhibition: a novel strategy for diabetes treatment. Nat Rev Drug Discov 2010;9:551-9.ArticlePubMedPDF
- 10. DeFronzo RA, Hompesch M, Kasichayanula S, Liu X, Hong Y, Pfister M, et al. Characterization of renal glucose reabsorption in response to dapagliflozin in healthy subjects and subjects with type 2 diabetes. Diabetes Care 2013;36:3169-76.ArticlePubMedPMCPDF
- 11. Lee JY, Cho Y, Lee M, Kim YJ, Lee YH, Lee BW, et al. Predictors of the therapeutic efficacy and consideration of the best combination therapy of sodium-glucose co-transporter 2 inhibitors. Diabetes Metab J 2019;43:158-73.ArticlePubMedPMCPDF
- 12. Rieg T, Vallon V. Development of SGLT1 and SGLT2 inhibitors. Diabetologia 2018;61:2079-86.ArticlePubMedPMCPDF
- 13. Bonner C, Kerr-Conte J, Gmyr V, Queniat G, Moerman E, Thevenet J, et al. Inhibition of the glucose transporter SGLT2 with dapagliflozin in pancreatic alpha cells triggers glucagon secretion. Nat Med 2015;21:512-7.ArticlePubMedPDF
- 14. Merovci A, Solis-Herrera C, Daniele G, Eldor R, Fiorentino TV, Tripathy D, et al. Dapagliflozin improves muscle insulin sensitivity but enhances endogenous glucose production. J Clin Invest 2014;124:509-14.ArticlePubMedPMC
- 15. Henry RR, Rosenstock J, Edelman S, Mudaliar S, Chalamandaris AG, Kasichayanula S, et al. Exploring the potential of the SGLT2 inhibitor dapagliflozin in type 1 diabetes: a randomized, double-blind, placebo-controlled pilot study. Diabetes Care 2015;38:412-9.ArticlePubMedPDF
- 16. Nishimura R, Osonoi T, Kanada S, Jinnouchi H, Sugio K, Omiya H, et al. Effects of luseogliflozin, a sodium-glucose co-transporter 2 inhibitor, on 24-h glucose variability assessed by continuous glucose monitoring in Japanese patients with type 2 diabetes mellitus: a randomized, double-blind, placebo-controlled, crossover study. Diabetes Obes Metab 2015;17:800-4.ArticlePubMedPMCPDF
- 17. Nishimura R, Tanaka Y, Koiwai K, Inoue K, Hach T, Salsali A, et al. Effect of empagliflozin monotherapy on postprandial glucose and 24-hour glucose variability in Japanese patients with type 2 diabetes mellitus: a randomized, double-blind, placebo-controlled, 4-week study. Cardiovasc Diabetol 2015;14:11.ArticlePubMedPMCPDF
- 18. Sands AT, Zambrowicz BP, Rosenstock J, Lapuerta P, Bode BW, Garg SK, et al. Sotagliflozin, a dual SGLT1 and SGLT2 inhibitor, as adjunct therapy to insulin in type 1 diabetes. Diabetes Care 2015;38:1181-8.ArticlePubMedPMCPDF
- 19. Nishimura R, Omiya H, Sugio K, Ubukata M, Sakai S, Samukawa Y. Sodium- glucose cotransporter 2 inhibitor luseogliflozin improves glycaemic control, assessed by continuous glucose monitoring, even on a low-carbohydrate diet. Diabetes Obes Metab 2016;18:702-6.ArticlePubMedPMCPDF
- 20. Famulla S, Pieber TR, Eilbracht J, Neubacher D, Soleymanlou N, Woerle HJ, et al. Glucose exposure and variability with empagliflozin as adjunct to insulin in patients with type 1 diabetes: continuous glucose monitoring data from a 4-week, randomized, placebo-controlled trial (EASE-1). Diabetes Technol Ther 2017;19:49-60.ArticlePubMed
- 21. Matsumura M, Nakatani Y, Tanka S, Aoki C, Sagara M, Yanagi K, et al. Efficacy of additional canagliflozin administration to type 2 diabetes patients receiving insulin therapy: examination of diurnal glycemic patterns using continuous glucose monitoring (CGM). Diabetes Ther 2017;8:821-7.ArticlePubMedPMCPDF
- 22. Nomoto H, Miyoshi H, Sugawara H, Ono K, Yanagiya S, Oita M, et al. A randomized controlled trial comparing the effects of dapagliflozin and DPP-4 inhibitors on glucose variability and metabolic parameters in patients with type 2 diabetes mellitus on insulin. Diabetol Metab Syndr 2017;9:54.ArticlePubMedPMCPDF
- 23. Rodbard HW, Peters AL, Slee A, Cao A, Traina SB, Alba M. The effect of canagliflozin, a sodium glucose cotransporter 2 inhibitor, on glycemic end points assessed by continuous glucose monitoring and patient-reported outcomes among people with type 1 diabetes. Diabetes Care 2017;40:171-80.ArticlePubMedPDF
- 24. Henry RR, Strange P, Zhou R, Pettus J, Shi L, Zhuplatov SB, et al. Effects of dapagliflozin on 24-hour glycemic control in patients with type 2 diabetes: a randomized controlled trial. Diabetes Technol Ther 2018;20:715-24.ArticlePubMedPMC
- 25. Danne T, Cariou B, Buse JB, Garg SK, Rosenstock J, Banks P, et al. Improved time in range and glycemic variability with sotagliflozin in combination with insulin in adults with type 1 diabetes: a pooled analysis of 24-week continuous glucose monitoring data from the intandem program. Diabetes Care 2019;42:919-30.ArticlePubMedPMCPDF
- 26. Service FJ, Molnar GD, Rosevear JW, Ackerman E, Gatewood LC, Taylor WF. Mean amplitude of glycemic excursions, a measure of diabetic instability. Diabetes 1970;19:644-55.ArticlePubMedPDF
- 27. Risso A, Mercuri F, Quagliaro L, Damante G, Ceriello A. Intermittent high glucose enhances apoptosis in human umbilical vein endothelial cells in culture. Am J Physiol Endocrinol Metab 2001;281:E924-30.ArticlePubMed
- 28. Quagliaro L, Piconi L, Assaloni R, Martinelli L, Motz E, Ceriello A. Intermittent high glucose enhances apoptosis related to oxidative stress in human umbilical vein endothelial cells: the role of protein kinase C and NAD(P)H-oxidase activation. Diabetes 2003;52:2795-804.PubMed
- 29. Monnier L, Mas E, Ginet C, Michel F, Villon L, Cristol JP, et al. Activation of oxidative stress by acute glucose fluctuations compared with sustained chronic hyperglycemia in patients with type 2 diabetes. JAMA 2006;295:1681-7.ArticlePubMed
- 30. Park SE, Lee BW, Kim JH, Lee WJ, Cho JH, Jung CH, et al. Effect of gemigliptin on glycaemic variability in patients with type 2 diabetes (STABLE study). Diabetes Obes Metab 2017;19:892-6.ArticlePubMedPDF
- 31. Kim NH, Kim DL, Kim KJ, Kim NH, Choi KM, Baik SH, et al. Effects of vildagliptin or pioglitazone on glycemic variability and oxidative stress in patients with type 2 diabetes inadequately controlled with metformin monotherapy: a 16-week, randomised, open label, pilot study. Endocrinol Metab (Seoul) 2017;32:241-7.ArticlePubMedPMCPDF
- 32. Kim HS, Shin JA, Lee SH, Kim ES, Cho JH, Son HY, et al. A comparative study of the effects of a dipeptidyl peptidase-IV inhibitor and sulfonylurea on glucose variability in patients with type 2 diabetes with inadequate glycemic control on metformin. Diabetes Technol Ther 2013;15:810-6.ArticlePubMed
- 33. FLAT-SUGAR Trial Investigators. Glucose variability in a 26-week randomized comparison of mealtime treatment with rapid-acting insulin versus GLP-1 agonist in participants with type 2 diabetes at high cardiovascular risk. Diabetes Care 2016;39:973-81.ArticlePubMedPDF
- 34. Al-Jobori H, Daniele G, Cersosimo E, Triplitt C, Mehta R, Norton L, et al. Empagliflozin and kinetics of renal glucose transport in healthy individuals and individuals with type 2 diabetes. Diabetes 2017;66:1999-2006.ArticlePubMedPMCPDF
- 35. Rave K, Nosek L, Posner J, Heise T, Roggen K, van Hoogdalem EJ. Renal glucose excretion as a function of blood glucose concentration in subjects with type 2 diabetes: results of a hyperglycaemic glucose clamp study. Nephrol Dial Transplant 2006;21:2166-71.ArticlePubMedPDF
REFERENCES
(A) Glycosylated hemoglobin (HbA1c) and (B) glycated albumin levels recorded during the study. Values are presented as mean±standard deviation. aP<0.05 for the comparison between groups.
(A) Mean glucose levels, (B) standard deviation, and (C) mean amplitude of glycemic excursion (MAGE) derived from continuous glucose monitoring. The right panel shows individual plots from participants. Values are presented as mean±standard deviation. aP<0.05.
Percentage of time in which glucose levels were within the target range, hyperglycemia, and hypoglycemia in the (A) placebo and (B) dapagliflozin groups. aP<0.05 compared with baseline, bP<0.05 compared with the placebo group.
|
Placebo (n=43) |
Dapagliflozin (n=41) |
|||||
|---|---|---|---|---|---|---|
| Baseline | 6 wk | 12 wk | Baseline | 6 wk | 12 wk | |
| Body weight, kg | 68.3±11.0 | 68.1±11.2 | 67.9±11.6 | 71.3±13.8 | 70.8±14.0a | 70.1±14.0a |
| Body mass index, kg/m2 | 26.6±3.0 | 26.6±3.1 | 26.5±3.3 | 27.3±3.9 | 27.1±3.9a | 26.8±4.0a |
| Fasting glucose, mg/dL | 137.4±53.5 | 136.2±48.7 | 136.3±41.6 | 145.6±56.4 | 142.7±55.8 | 124.0±29.3a,b |
| Insulin dose, U/day | 29.9±10.7 | 29.3±10.8a | 29.4±11.2 | 35.0±16.7 | 32.3±16.9a | 31.5±16.9a |
| Systolic BP, mm Hg | 121.8±11.9 | 123.7±11.6 | 122.4±11.8 | 125.2±10.9 | 125.0±9.3 | 123.0±9.6 |
| Diastolic BP, mm Hg | 76.3±8.4 | 76.7±7.6 | 77.5±7.9 | 77.7±7.7 | 78.0±7.4 | 77.4±7.9 |
| Hemoglobin, g/dL | 14.2±1.4 | 14.2±1.4 | 14.2±1.5 | 13.7±1.3 | 13.8±1.3 | 14.5±1.6a,b |
| BUN, mg/dL | 15.1±4.0 | 15.2±3.6 | 16.1±4.0 | 15.0±4.5 | 15.6±4.6 | 16.7±4.4a |
| Creatinine, mg/dL | 0.80±0.17 | 0.80±0.17 | 0.79±0.16 | 0.77±0.16 | 0.78±0.16 | 0.80±0.18a |
| Uric acid, mg/dL | 4.52±1.28 | 4.49±1.17 | 4.62±1.29 | 4.59±1.30 | 4.56±1.30 | 4.46±1.31a,b |
| eGFR, mL/min/1.73 m2 | 85.3±12.8 | 85.3±12.9 | 86.1±12.7 | 87.7±16.2 | 87.5±16.6 | 85.4±19.6a,b |
| AST, IU/L | 25.3±8.7 | 25.2±8.7 | 27.1±14.7 | 26.6±13.7 | 26.5±13.7 | 23.6±9.8a |
| ALT, IU/L | 27.6±11.4 | 27.2±11.3 | 29.3±14.4 | 28.4±16.8 | 29.6±19.1 | 23.1±9.5a,b |
| Total cholesterol, mg/dL | 158.8±34.4 | 159.4±32.3 | 159.2±36.9 | 157.1±33.5 | 154.6±30.9 | 160.6±32.9 |
| Triglycerides, mg/dL | 137.4±65.6 | 147.1±84.1 | 146.4±110.9 | 140.9±77.3 | 136.3±72.4 | 144.7±80.9 |
| HDL-C, mg/dL | 46.3±8.8 | 46.4±8.9 | 46.7±8.4 | 44.1±7.9 | 44.1±8.0 | 45.0±8.8 |
| LDL-C, mg/dL | 86.8±28.6 | 86.1±26.0 | 85.2±29.0 | 85.3±25.0 | 83.7±24.5 | 88.1±28.1 |
Values are presented as mean±standard deviation.
BP, blood pressure; BUN, blood urea nitrogen; eGFR, estimated glomerular filtration rate; AST, aspartate aminotransferase; ALT, alanine aminotransferase; HDL-C, high-density lipoprotein cholesterol; LDL-C, low-density lipoprotein cholesterol.
a P<0.05 compared with baseline values,
b P<0.05 compared with the placebo group.
Figure & Data
References
Citations

- Selective sodium-glucose cotransporter-2 inhibitors in the improvement of hemoglobin and hematocrit in patients with type 2 diabetes mellitus: a network meta-analysis
Yuanyuan Luo, Ruojing Bai, Wei Zhang, Guijun Qin
Frontiers in Endocrinology.2024;[Epub] CrossRef - Continuous Glucose Monitoring Profiles and Health Outcomes After Dapagliflozin Plus Saxagliptin vs Insulin Glargine
Donald C Simonson, Marcia A Testa, Ella Ekholm, Maxwell Su, Tina Vilsbøll, Serge A Jabbour, Marcus Lind
The Journal of Clinical Endocrinology & Metabolism.2024;[Epub] CrossRef - Impact of empagliflozin on insulin needs in patients with heart failure and diabetes: An EMPEROR‐Pooled analysis
Khawaja M. Talha, Jennifer Green, Gerasimos Filippatos, Stuart Pocock, Faiez Zannad, Martina Brueckmann, Elke Schueler, Anne Pernille Ofstad, João Pedro Ferreira, Stefan D. Anker, Javed Butler, Julio Rosenstock, Milton Packer
Diabetes, Obesity and Metabolism.2024;[Epub] CrossRef - Risk of Urinary Tract Infection in Patients with Type 2 Diabetes Mellitus Treated with Dapagliflozin: A Systematic Review and Meta-Analysis of Randomized Controlled Trials
Zhigui Zheng, Dongyuan He, Jianguo Chen, Xiaohui Xie, Yunan Lu, Binbin Wu, Xinxin Jiang
Clinical Drug Investigation.2023; 43(4): 209. CrossRef - Effect of SGLT2 Inhibitors and Metformin on Inflammatory and Prognostic
Biomarkers in Type 2 Diabetes Patients
Yang Cao, Ning Liang, Ting Liu, Jingai Fang, Xiaodong Zhang
Endocrine, Metabolic & Immune Disorders - Drug Targets.2023; 23(4): 530. CrossRef - What is Glycaemic Variability and which Pharmacological Treatment Options are Effective? A Narrative Review
Juan Miguel Huertas Cañas, Maria Alejandra Gomez Gutierrez, Andres Bedoya Ossa
European Endocrinology.2023; 19(2): 4. CrossRef - La variabilité glycémique : un facteur de risque singulier à conjuguer au pluriel
Louis Monnier, Claude Colette, Fabrice Bonnet, David Owens
Médecine des Maladies Métaboliques.2022; 16(1): 15. CrossRef - Association between Variability of Metabolic Risk Factors and Cardiometabolic Outcomes
Min Jeong Park, Kyung Mook Choi
Diabetes & Metabolism Journal.2022; 46(1): 49. CrossRef - Effect of sodium-glucose cotransporter-2 (SGLT2) inhibitors on serum urate levels in patients with and without diabetes: a systematic review and meta-regression of 43 randomized controlled trials
Alicia Swee Yan Yip, Shariel Leong, Yao Hao Teo, Yao Neng Teo, Nicholas L. X. Syn, Ray Meng See, Caitlin Fern Wee, Elliot Yeung Chong, Chi-Hang Lee, Mark Y. Chan, Tiong-Cheng Yeo, Raymond C. C. Wong, Ping Chai, Ching-Hui Sia
Therapeutic Advances in Chronic Disease.2022; 13: 204062232210835. CrossRef - Hypoglycemic agents and glycemic variability in individuals with type 2 diabetes: A systematic review and network meta-analysis
SuA Oh, Sujata Purja, Hocheol Shin, Minji Kim, Eunyoung Kim
Diabetes and Vascular Disease Research.2022; 19(3): 147916412211068. CrossRef - The Clinical Effect of Dapagliflozin in Patients with Angiographically Confirmed Coronary Artery Disease and Concomitant Type 2 Diabetes Mellitus
Yana Yu. Dzhun, Yevhen Yu. Marushko, Yanina A. Saienko, Nadiya M. Rudenko, Borys M. Mankovsky
Ukrainian Journal of Cardiovascular Surgery.2022; 30(3): 35. CrossRef - Stress-Induced Hyperglycaemia in Non-Diabetic Patients with Acute Coronary Syndrome: From Molecular Mechanisms to New Therapeutic Perspectives
Alessandro Bellis, Ciro Mauro, Emanuele Barbato, Antonio Ceriello, Antonio Cittadini, Carmine Morisco
International Journal of Molecular Sciences.2021; 22(2): 775. CrossRef - Glycemic Variability Impacted by SGLT2 Inhibitors and GLP 1 Agonists in Patients with Diabetes Mellitus: A Systematic Review and Meta-Analysis
Heeyoung Lee, Se-eun Park, Eun-Young Kim
Journal of Clinical Medicine.2021; 10(18): 4078. CrossRef - Effect of Dapagliflozin on Glycemic Variability in Patients with Type 2 Diabetes under Insulin Glargine Combined with Other Oral Hypoglycemic Drugs
Menghui Luo, Xiaocen Kong, Huiying Wang, Xiaofang Zhai, Tingting Cai, Bo Ding, Yun Hu, Ting Jing, Xiaofei Su, Huiqin Li, Jianhua Ma, Yoshifumi Saisho
Journal of Diabetes Research.2020; 2020: 1. CrossRef - Time in Range from Continuous Glucose Monitoring: A Novel Metric for Glycemic Control
Jee Hee Yoo, Jae Hyeon Kim
Diabetes & Metabolism Journal.2020; 44(6): 828. CrossRef
- Figure
- Related articles
-
- Efficacy and Safety of Evogliptin Add-on Therapy to Dapagliflozin/Metformin Combinations in Patients with Poorly Controlled Type 2 Diabetes Mellitus: A 24-Week Multicenter Randomized Placebo-Controlled Parallel-Design Phase-3 Trial with a 28-Week Extension
- Efficacy and Safety of Enavogliflozin versus Dapagliflozin as Add-on to Metformin in Patients with Type 2 Diabetes Mellitus: A 24-Week, Double-Blind, Randomized Trial

 KDA
KDA
 PubReader
PubReader ePub Link
ePub Link Cite
Cite