
- Current
- Browse
- Collections
-
For contributors
- For Authors
- Instructions to authors
- Article processing charge
- e-submission
- For Reviewers
- Instructions for reviewers
- How to become a reviewer
- Best reviewers
- For Readers
- Readership
- Subscription
- Permission guidelines
- About
- Editorial policy
Search
- Page Path
- HOME > Search
Review
- Basic Research
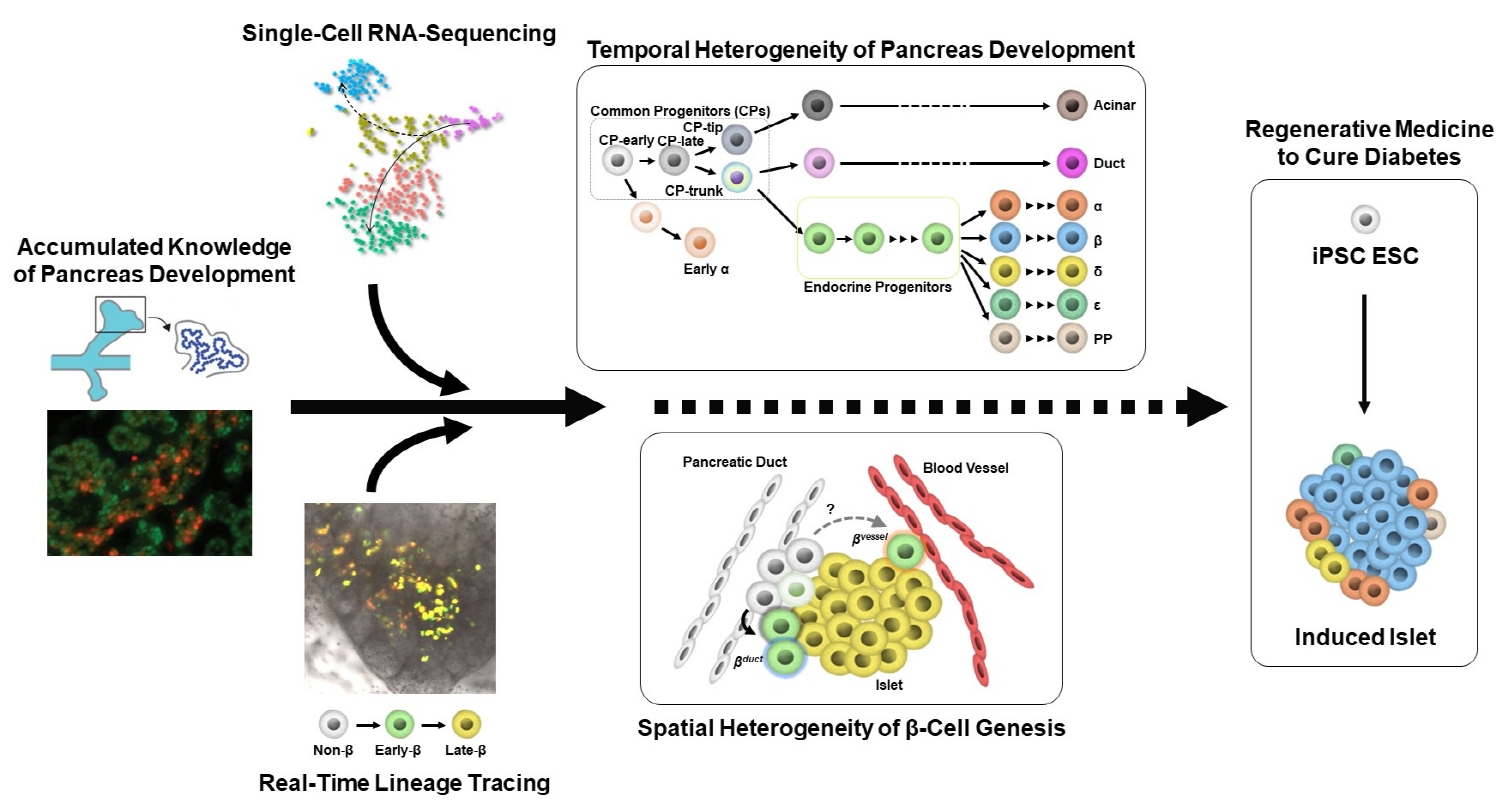
- Heterogeneity of Islet Cells during Embryogenesis and Differentiation
- Shugo Sasaki, Takeshi Miyatsuka
- Diabetes Metab J. 2023;47(2):173-184. Published online January 12, 2023
- DOI: https://doi.org/10.4093/dmj.2022.0324
- 3,773 View
- 248 Download
- 1 Web of Science
- 1 Crossref
-
 Abstract
Abstract
 PDF
PDF PubReader
PubReader  ePub
ePub - Diabetes is caused by insufficient insulin secretion due to β-cell dysfunction and/or β-cell loss. Therefore, the restoration of functional β-cells by the induction of β-cell differentiation from embryonic stem (ES) and induced-pluripotent stem (iPS) cells, or from somatic non-β-cells, may be a promising curative therapy. To establish an efficient and feasible method for generating functional insulin-producing cells, comprehensive knowledge of pancreas development and β-cell differentiation, including the mechanisms driving cell fate decisions and endocrine cell maturation is crucial. Recent advances in single-cell RNA sequencing (scRNA-seq) technologies have opened a new era in pancreas development and diabetes research, leading to clarification of the detailed transcriptomes of individual insulin-producing cells. Such extensive high-resolution data enables the inference of developmental trajectories during cell transitions and gene regulatory networks. Additionally, advancements in stem cell research have not only enabled their immediate clinical application, but also has made it possible to observe the genetic dynamics of human cell development and maturation in a dish. In this review, we provide an overview of the heterogeneity of islet cells during embryogenesis and differentiation as demonstrated by scRNA-seq studies on the developing and adult pancreata, with implications for the future application of regenerative medicine for diabetes.
-
Citations
Citations to this article as recorded by- Newly discovered knowledge pertaining to glucagon and its clinical applications
Dan Kawamori, Shugo Sasaki
Journal of Diabetes Investigation.2023; 14(7): 829. CrossRef
- Newly discovered knowledge pertaining to glucagon and its clinical applications
Original Article
- Basic Research
- Differentiation of Microencapsulated Neonatal Porcine Pancreatic Cell Clusters in Vitro Improves Transplant Efficacy in Type 1 Diabetes Mellitus Mice
- Gyeong-Jin Cheon, Heon-Seok Park, Eun-Young Lee, Min Jung Kim, Young-Hye You, Marie Rhee, Ji-Won Kim, Kun-Ho Yoon
- Diabetes Metab J. 2022;46(5):677-688. Published online February 7, 2022
- DOI: https://doi.org/10.4093/dmj.2021.0202
- 4,553 View
- 252 Download
- 2 Web of Science
- 2 Crossref
-
 Abstract
Abstract
 PDF
PDF Supplementary Material
Supplementary Material PubReader
PubReader  ePub
ePub - Background
Neonatal porcine pancreatic cell clusters (NPCCs) have been proposed as an alternative source of β cells for islet transplantation because of their low cost and growth potential after transplantation. However, the delayed glucose lowering effect due to the immaturity of NPCCs and immunologic rejection remain as a barrier to NPCC’s clinical application. Here, we demonstrate accelerated differentiation and immune-tolerant NPCCs by in vitro chemical treatment and microencapsulation.
Methods
NPCCs isolated from 3-day-old piglets were cultured in F-10 media and then microencapsulated with alginate on day 5. Differentiation of NPCCs is facilitated by media supplemented with activin receptor-like kinase 5 inhibitor II, triiodothyronine and exendin-4 for 2 weeks. Marginal number of microencapsulated NPCCs to cure diabetes with and without differentiation were transplanted into diabetic mice and observed for 8 weeks.
Results
The proportion of insulin-positive cells and insulin mRNA levels of NPCCs were significantly increased in vitro in the differentiated group compared with the undifferentiated group. Blood glucose levels decreased eventually after transplantation of microencapsulated NPCCs in diabetic mice and normalized after 7 weeks in the differentiated group. In addition, the differentiated group showed nearly normal glucose tolerance at 8 weeks after transplantation. In contrast, neither blood glucose levels nor glucose tolerance were improved in the undifferentiated group. Retrieved graft in the differentiated group showed greater insulin response to high glucose compared with the undifferentiated group.
Conclusion
in vitro differentiation of microencapsulated immature NPCCs increased the proportion of insulin-positive cells and improved transplant efficacy in diabetic mice without immune rejection. -
Citations
Citations to this article as recorded by- Dual-targeted nano-encapsulation of neonatal porcine islet-like cell clusters with triiodothyronine-loaded bifunctional polymersomes
Sang Hoon Lee, Minse Kim, Eun-Jin Lee, Sun Mi Ahn, Yu-Rim Ahn, Jaewon Choi, Jung-Taek Kang, Hyun-Ouk Kim
Discover Nano.2024;[Epub] CrossRef - Long‐term efficacy of encapsulated xenogeneic islet transplantation: Impact of encapsulation techniques and donor genetic traits
Heon‐Seok Park, Eun Young Lee, Young‐Hye You, Marie Rhee, Jong‐Min Kim, Seong‐Soo Hwang, Poong‐Yeon Lee
Journal of Diabetes Investigation.2024;[Epub] CrossRef
- Dual-targeted nano-encapsulation of neonatal porcine islet-like cell clusters with triiodothyronine-loaded bifunctional polymersomes

 KDA
KDA
 First
First Prev
Prev





