
- Current
- Browse
- Collections
-
For contributors
- For Authors
- Instructions to authors
- Article processing charge
- e-submission
- For Reviewers
- Instructions for reviewers
- How to become a reviewer
- Best reviewers
- For Readers
- Readership
- Subscription
- Permission guidelines
- About
- Editorial policy
Search
- Page Path
- HOME > Search
Original Articles
- Type 1 Diabetes
- A New Tool to Identify Pediatric Patients with Atypical Diabetes Associated with Gene Polymorphisms
- Sophie Welsch, Antoine Harvengt, Paola Gallo, Manon Martin, Dominique Beckers, Thierry Mouraux, Nicole Seret, Marie-Christine Lebrethon, Raphaël Helaers, Pascal Brouillard, Miikka Vikkula, Philippe A. Lysy
- Received May 26, 2023 Accepted November 25, 2023 Published online March 22, 2024
- DOI: https://doi.org/10.4093/dmj.2023.0166 [Epub ahead of print]
- 880 View
- 56 Download
-
 Abstract
Abstract
 PDF
PDF Supplementary Material
Supplementary Material PubReader
PubReader  ePub
ePub - Background
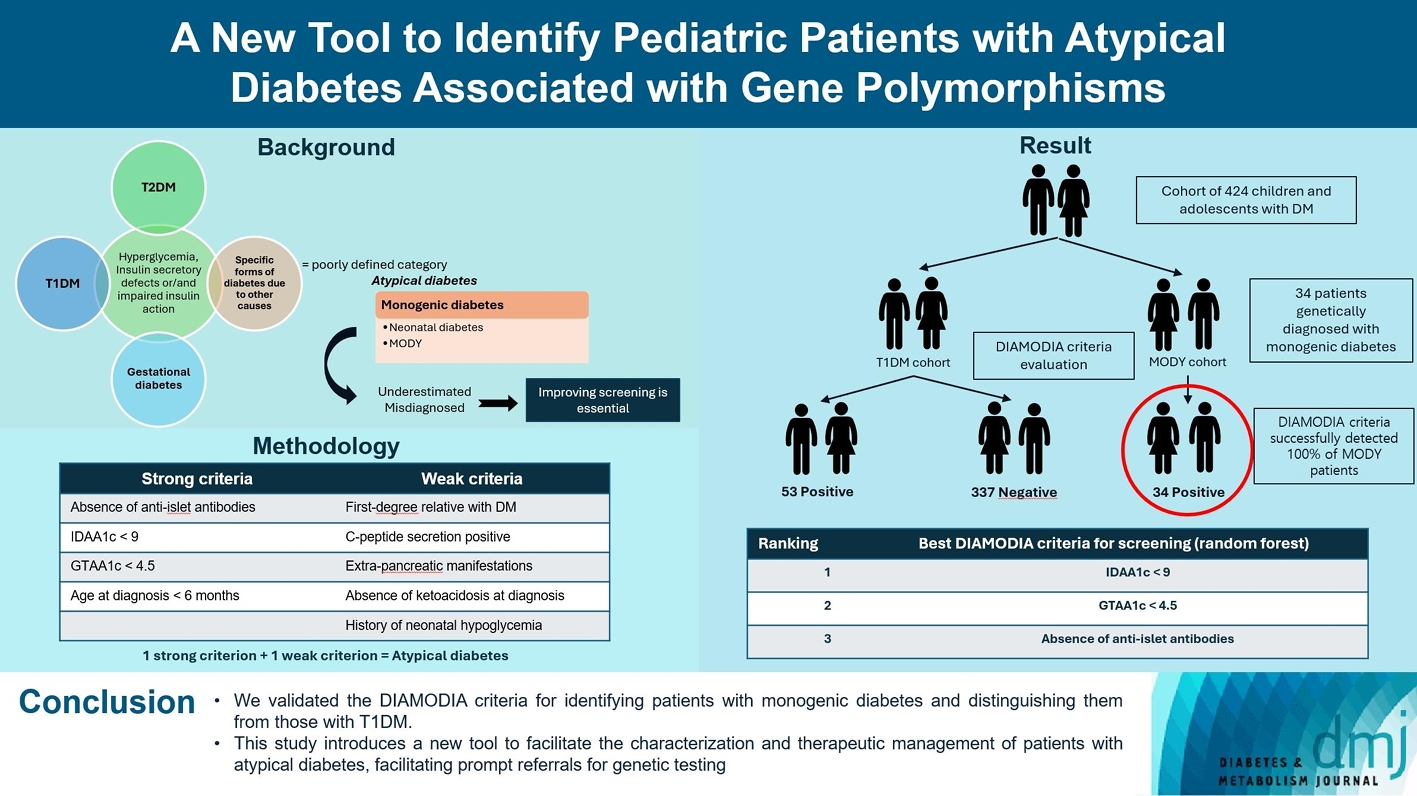
Recent diabetes subclassifications have improved the differentiation between patients with type 1 diabetes mellitus (T1DM) and type 2 diabetes mellitus despite several overlapping features, yet without considering genetic forms of diabetes. We sought to facilitate the identification of monogenic diabetes by creating a new tool that we validated in a pediatric maturity-onset diabetes of the young (MODY) cohort.
Methods
We first created the DIAgnose MOnogenic DIAbetes (DIAMODIA) criteria based on the pre-existing, but incomplete, MODY calculator. This new score is composed of four strong and five weak criteria, with patients having to display at least one weak and one strong criterion.
Results
The effectiveness of the DIAMODIA criteria was evaluated in two patient cohorts, the first consisting of patients with confirmed MODY diabetes (n=34) and the second of patients with T1DM (n=390). These DIAMODIA criteria successfully detected 100% of MODY patients. Multiple correspondence analysis performed on the MODY and T1DM cohorts enabled us to differentiate MODY patients from T1DM. The three most relevant variables to distinguish a MODY from T1DM profile were: lower insulin-dose adjusted A1c score ≤9, glycemic target-adjusted A1c score ≤4.5, and absence of three anti-islet cell autoantibodies.
Conclusion
We validated the DIAMODIA criteria, as it effectively identified all monogenic diabetes patients (MODY cohort) and succeeded to differentiate T1DM from MODY patients. The creation of this new and effective tool is likely to facilitate the characterization and therapeutic management of patients with atypical diabetes, and promptly referring them for genetic testing which would markedly improve clinical care and counseling, as well.
- Type 1 Diabetes
- Optimal Coefficient of Variance Threshold to Minimize Hypoglycemia Risk in Individuals with Well-Controlled Type 1 Diabetes Mellitus
- Jee Hee Yoo, Seung Hee Yang, Sang-Man Jin, Jae Hyeon Kim
- Received March 14, 2023 Accepted August 12, 2023 Published online March 4, 2024
- DOI: https://doi.org/10.4093/dmj.2023.0083 [Epub ahead of print]

- 566 View
- 23 Download
-
 Abstract
Abstract
 PDF
PDF Supplementary Material
Supplementary Material PubReader
PubReader  ePub
ePub - Background
This study investigated the optimal coefficient of variance (%CV) for preventing hypoglycemia based on real-time continuous glucose monitoring (rt-CGM) data in people with type 1 diabetes mellitus (T1DM) already achieving their mean glucose (MG) target.
Methods
Data from 172 subjects who underwent rt-CGM for at least 90 days and for whom 439 90-day glycemic profiles were available were analyzed. Receiver operator characteristic analysis was conducted to determine the cut-off value of %CV to achieve time below range (%TBR)<54 mg/dL <1 and =0.
Results
Overall mean glycosylated hemoglobin was 6.8% and median %TBR<54 mg/dL was 0.2%. MG was significantly higher and %CV significantly lower in profiles achieving %TBR<54 mg/dL <1 compared to %TBR<54 mg/dL ≥1 (all P<0.001). The cut-off value of %CV for achieving %TBR<54 mg/dL <1 was 37.5%, 37.3%, and 31.0%, in the whole population, MG >135 mg/dL, and ≤135 mg/dL, respectively. The cut-off value for %TBR<54 mg/dL=0% was 29.2% in MG ≤135 mg/dL. In profiles with MG ≤135 mg/dL, 94.2% of profiles with a %CV <31 achieved the target of %TBR<54 mg/dL <1, and 97.3% with a %CV <29.2 achieved the target of %TBR<54 mg/ dL=0%. When MG was >135 mg/dL, 99.4% of profiles with a %CV <37.3 achieved %TBR<54 mg/dL <1.
Conclusion
In well-controlled T1DM with MG ≤135 mg/dL, we suggest a %CV <31% to achieve the %TBR<54 mg/dL <1 target. Furthermore, we suggest a %CV <29.2% to achieve the target of %TBR<54 mg/dL =0 for people at high risk of hypoglycemia.
- Complications
- Does the Relationship of the Autonomic Symptoms Questionnaire COMPASS 31 with Cardiovascular Autonomic Tests Differ between Type 1 and Type 2 Diabetes Mellitus?
- Ilenia D’Ippolito, Marika Menduni, Cinzia D’Amato, Aikaterini Andreadi, Davide Lauro, Vincenza Spallone
- Received August 28, 2023 Accepted November 22, 2023 Published online February 26, 2024
- DOI: https://doi.org/10.4093/dmj.2023.0301 [Epub ahead of print]
- 645 View
- 47 Download
-
 Abstract
Abstract
 PDF
PDF Supplementary Material
Supplementary Material PubReader
PubReader  ePub
ePub - Background
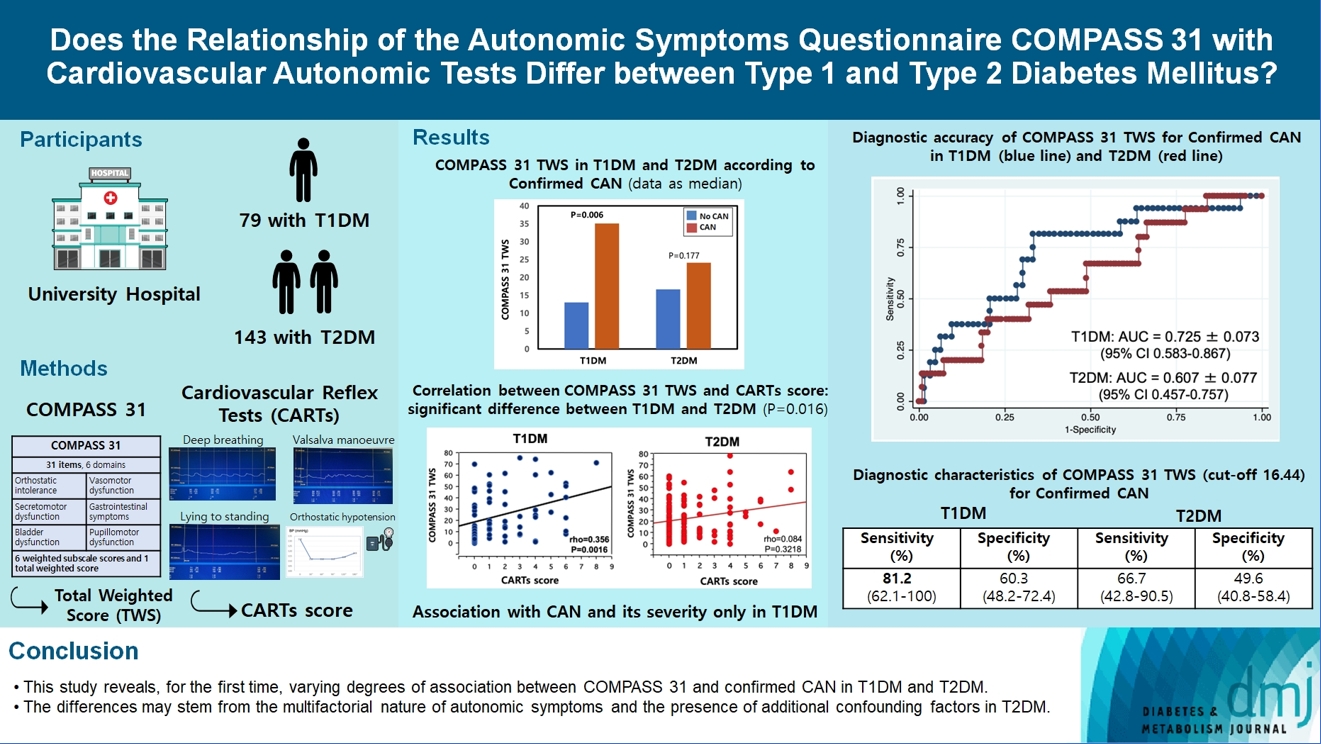
The aim was to investigate if autonomic symptoms questionnaire Composite Autonomic Symptom Score (COMPASS) 31 has different association with cardiovascular autonomic neuropathy (CAN) and diagnostic performance between type 1 diabetes mellitus (T1DM) and type 2 diabetes mellitus (T2DM).
Methods
Seventy-nine participants with T1DM and 140 with T2DM completed COMPASS 31 before cardiovascular reflex tests (CARTs) for CAN, and assessment of symptoms, signs, vibration, and thermal perception thresholds for diabetic polyneuropathy (DPN) diagnosis.
Results
COMPASS 31 total weighted score (TWS) was similar in the two groups, but significantly associated with confirmed CAN only in T1DM (P=0.0056) and not T2DM group (P=0.1768) and correlated with CARTs score more strongly in T1DM (rho=0.356, P=0.0016) than in T2DM group (rho=0.084, P=0.3218) (P=0.016). Only in T1DM and not T2DM group, the area under the receiver operating characteristic curve (AUC) reached a fair diagnostic accuracy (>0.7) for confirmed CAN (0.73±0.07 vs. 0.61±0.08) and DPN (0.75±0.06 vs. 0.68±0.05), although without a significant difference. COMPASS 31 TWS (cut-off 16.44) reached acceptable diagnostic performance in T1DM, with sensitivity for confirmed CAN 81.2% and sensitivity and specificity for DPN 76.3% and 78%, compared to T2DM group (all <70%). AUC for DPN of orthostatic intolerance domain was higher in T1DM compared to T2DM group (0.73±0.05 vs. 0.58±0.04, P=0.027).
Conclusion
COMPASS 31 is more weakly related to CAN in T2DM than in T1DM, with a fair diagnostic accuracy for confirmed CAN only in T1DM. This difference supports a multifactorial origin of symptoms and should be considered when using COMPASS 31.
- Basic Research
- Extracellular Vimentin Alters Energy Metabolism And Induces Adipocyte Hypertrophy
- Ji-Hae Park, Soyeon Kwon, Young Mi Park
- Diabetes Metab J. 2024;48(2):215-230. Published online September 26, 2023
- DOI: https://doi.org/10.4093/dmj.2022.0332

- 2,563 View
- 217 Download
-
 Abstract
Abstract
 PDF
PDF Supplementary Material
Supplementary Material PubReader
PubReader  ePub
ePub - Background
Previous studies have reported that oxidative stress contributes to obesity characterized by adipocyte hypertrophy. However, mechanism has not been studied extensively. In the current study, we evaluated role of extracellular vimentin secreted by oxidized low-density lipoprotein (oxLDL) in energy metabolism in adipocytes.
Methods
We treated 3T3-L1-derived adipocytes with oxLDL and measured vimentin which was secreted in the media. We evaluated changes in uptake of glucose and free fatty acid, expression of molecules functioning in energy metabolism, synthesis of adenosine triphosphate (ATP) and lactate, markers for endoplasmic reticulum (ER) stress and autophagy in adipocytes treated with recombinant vimentin.
Results
Adipocytes secreted vimentin in response to oxLDL. Microscopic evaluation revealed that vimentin treatment induced increase in adipocyte size and increase in sizes of intracellular lipid droplets with increased intracellular triglyceride. Adipocytes treated with vimentin showed increased uptake of glucose and free fatty acid with increased expression of plasma membrane glucose transporter type 1 (GLUT1), GLUT4, and CD36. Vimentin treatment increased transcription of GLUT1 and hypoxia-inducible factor 1α (Hif-1α) but decreased GLUT4 transcription. Adipose triglyceride lipase (ATGL), peroxisome proliferator-activated receptor γ (PPARγ), sterol regulatory element-binding protein 1 (SREBP1), diacylglycerol O-acyltransferase 1 (DGAT1) and 2 were decreased by vimentin treatment. Markers for ER stress were increased and autophagy was impaired in vimentin-treated adipocytes. No change was observed in synthesis of ATP and lactate in the adipocytes treated with vimentin.
Conclusion
We concluded that extracellular vimentin regulates expression of molecules in energy metabolism and promotes adipocyte hypertrophy. Our results show that vimentin functions in the interplay between oxidative stress and metabolism, suggesting a mechanism by which adipocyte hypertrophy is induced in oxidative stress.
Review
- Pathophysiology
- Immune-Checkpoint Inhibitors-Induced Type 1 Diabetes Mellitus: From Its Molecular Mechanisms to Clinical Practice
- Yun Kyung Cho, Chang Hee Jung
- Diabetes Metab J. 2023;47(6):757-766. Published online July 24, 2023
- DOI: https://doi.org/10.4093/dmj.2023.0072
- 2,925 View
- 333 Download
- 2 Crossref
-
 Abstract
Abstract
 PDF
PDF PubReader
PubReader  ePub
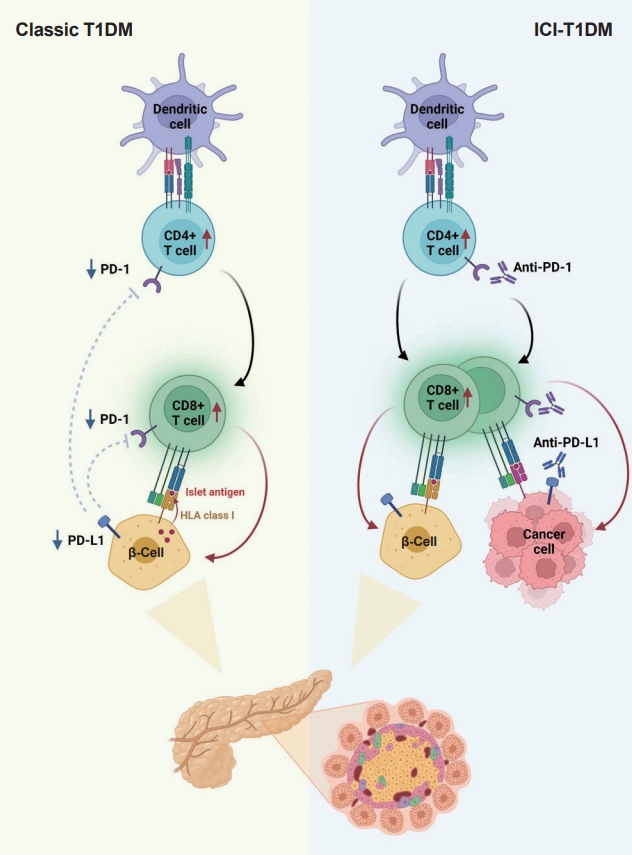
ePub - With the increasing use of immune-checkpoint inhibitors (ICIs), such as anti-cytotoxic T lymphocyte-associated antigen 4 (CTLA-4) and anti-programmed cell death-1 (PD-1), for the treatment of malignancies, cases of ICI-induced type 1 diabetes mellitus (ICI-T1DM) have been reported globally. This review focuses on the features and pathogenesis of this disease. T1DM is an immune-related adverse event that occurs following the administration of anti-PD-1 or anti-programmed death ligand-1 (PDL1) alone or in combination with anti-CTLA-4. More than half of the reported cases presented as abrupt-onset diabetic ketoacidosis. The primary mechanism of ICI-T1DM is T-cell stimulation, which results from the loss of interaction between PD-1 and PD-L1 in pancreatic islet. The similarities and differences between ICI-T1DM and classical T1DM may provide insights into this disease entity. ICI-T1DM is a rare but often life-threatening medical emergency that healthcare professionals and patients need to be aware of. Early detection of and screening for this disease is imperative. At present, the only known treatment for ICI-T1DM is insulin injection. Further research into the mechanisms and risk factors associated with ICI-T1DM development may contribute to a better understanding of this disease entity and the identification of possible preventive strategies.
-
Citations
Citations to this article as recorded by- Research Advances of Immune Checkpoint Inhibitors Related Endocrine Adverse Events
晶晶 王
Advances in Clinical Medicine.2024; 14(02): 2706. CrossRef - Immune checkpoint inhibitor‑associated diabetes mellitus in patients with HCC: Report of three cases and literature review
Gaocheng Wang, Jingjing Wang, Shuilin Dong, Zhanguo Zhang, Wanguang Zhang, Jianping Zhao
Experimental and Therapeutic Medicine.2024;[Epub] CrossRef
- Research Advances of Immune Checkpoint Inhibitors Related Endocrine Adverse Events
Original Articles
- Lifestyle
- Clinical Effects of a Home Care Pilot Program for Patients with Type 1 Diabetes Mellitus: A Retrospective Cohort Study
- Sejeong Lee, KyungYi Kim, Ji Eun Kim, Yura Hyun, Minyoung Lee, Myung-Il Hahm, Sang Gyu Lee, Eun Seok Kang
- Diabetes Metab J. 2023;47(5):693-702. Published online June 22, 2023
- DOI: https://doi.org/10.4093/dmj.2022.0170
- 1,981 View
- 138 Download
- 1 Web of Science
- 1 Crossref
-
 Abstract
Abstract
 PDF
PDF Supplementary Material
Supplementary Material PubReader
PubReader  ePub
ePub - Background
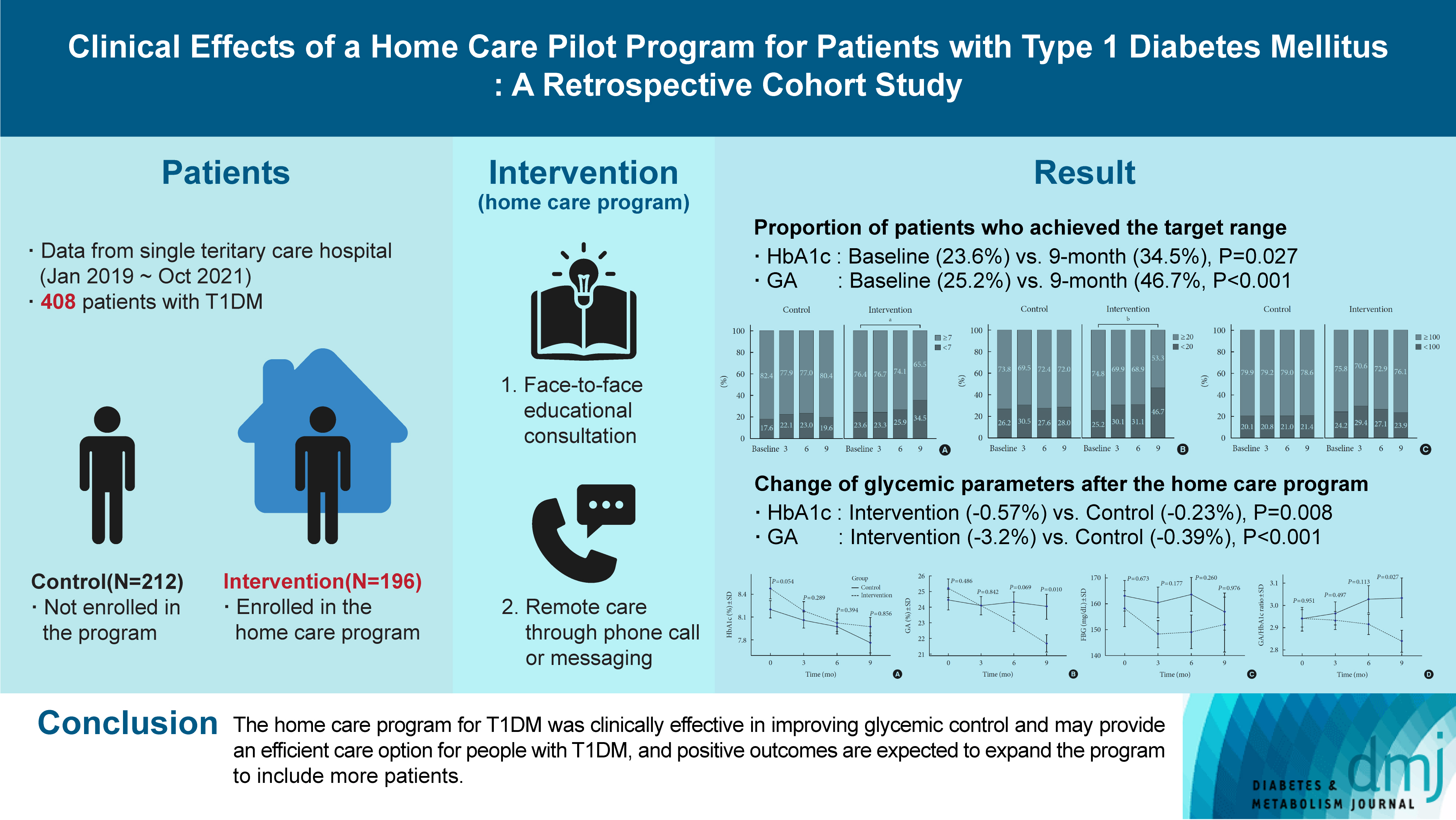
Given the importance of continuous self-care for people with type 1 diabetes mellitus (T1DM), the Ministry of Health and Welfare of Korea launched a pilot program for chronic disease management. Herein, we applied a home care pilot program to people with T1DM to investigate its effects.
Methods
This retrospective cohort study was conducted at a single tertiary hospital (January 2019 to October 2021). A multidisciplinary team comprising doctors, nurses, and clinical nutritionists provided specialized education and periodically assessed patients’ health status through phone calls or text messages. A linear mixed model adjusting for age, sex, and body mass index was used to analyze the glycemic control changes before and after implementing the program between the intervention and control groups.
Results
Among 408 people with T1DM, 196 were enrolled in the intervention group and 212 in the control group. The reduction in glycosylated hemoglobin (HbA1c) after the program was significantly greater in the intervention group than in the control group (estimated marginal mean, –0.57% vs. –0.23%, P=0.008); the same trend was confirmed for glycoalbumin (GA) (–3.2% vs. –0.39%, P<0.001). More patients achieved the target values of HbA1c (<7.0%) and GA (<20%) in the intervention group than in the control group at the 9-month follow-up (34.5% vs. 19.6% and 46.7% vs. 28.0%, respectively).
Conclusion
The home care program for T1DM was clinically effective in improving glycemic control and may provide an efficient care option for people with T1DM, and positive outcomes are expected to expand the program to include more patients. -
Citations
Citations to this article as recorded by- Glycemic outcomes and patient satisfaction and self-management improves in transition from standard to virtual multidisciplinary care
Noga Minsky, Liat Arnon Klug, Tatyana Kolobov, Elizabeth Tarshish, Yuval Shalev Many, Aviva Lipsitz, Amna Jabarin, Nicole Morozov, Dania Halperin, Moshe Shalom, Rachel Nissanholtz-Gannot, Genya Aharon-Hananel, Amir Tirosh, Orly Tamir
Diabetes Research and Clinical Practice.2024; 209: 111587. CrossRef
- Glycemic outcomes and patient satisfaction and self-management improves in transition from standard to virtual multidisciplinary care
- Technology/Device
- Glycemia according to the Use of Continuous Glucose Monitoring among Adults with Type 1 Diabetes Mellitus in Korea: A Real-World Study
- You-Bin Lee, Minjee Kim, Jae Hyeon Kim
- Diabetes Metab J. 2023;47(3):405-414. Published online March 6, 2023
- DOI: https://doi.org/10.4093/dmj.2022.0032

- 3,355 View
- 122 Download
- 3 Web of Science
- 4 Crossref
-
 Abstract
Abstract
 PDF
PDF Supplementary Material
Supplementary Material PubReader
PubReader  ePub
ePub - Background
We explored the association between continuous glucose monitoring (CGM) use and glycemia among adults with type 1 diabetes mellitus (T1DM) and determined the status of CGM metrics among adults with T1DM using CGM in the real-world.
Methods
For this propensity-matched cross-sectional study, individuals with T1DM who visited the outpatient clinic of the Endocrinology Department of Samsung Medical Center between March 2018 and February 2020 were screened. Among them, 111 CGM users (for ≥9 months) were matched based on propensity score considering age, sex, and diabetes duration in a 1:2 ratio with 203 CGM never-users. The association between CGM use and glycemic measures was explored. In a subpopulation of CGM users who had been using official applications (not “do-it-yourself” software) such that Ambulatory Glucose Profile data for ≥1 month were available (n=87), standardized CGM metrics were summarized.
Results
Linear regression analyses identified CGM use as a determining factor for log-transformed glycosylated hemoglobin. The fully-adjusted odds ratio (OR) and 95% confidence interval (CI) for uncontrolled glycosylated hemoglobin (>8%) were 0.365 (95% CI, 0.190 to 0.703) in CGM users compared to never-users. The fully-adjusted OR for controlled glycosylated hemoglobin (<7%) was 1.861 (95% CI, 1.119 to 3.096) in CGM users compared to never-users. Among individuals who had been using official applications for CGM, time in range (TIR) values within recent 30- and 90-day periods were 62.45%±16.63% and 63.08%±15.32%, respectively.
Conclusion
CGM use was associated with glycemic control status among Korean adults with T1DM in the real-world, although CGM metrics including TIR might require further improvement among CGM users. -
Citations
Citations to this article as recorded by- Real-World Continuous Glucose Monitoring Data from a Population with Type 1 Diabetes in South Korea: Nationwide Single-System Analysis
Ji Yoon Kim, Sang-Man Jin, Sarah B. Andrade, Boyang Chen, Jae Hyeon Kim
Diabetes Technology & Therapeutics.2024;[Epub] CrossRef - Accuracy and Safety of the 15-Day CareSens Air Continuous Glucose Monitoring System
Kyung-Soo Kim, Seung-Hwan Lee, Won Sang Yoo, Cheol-Young Park
Diabetes Technology & Therapeutics.2024; 26(4): 222. CrossRef - Navigating the Seas of Glycemic Control: The Role of Continuous Glucose Monitoring in Type 1 Diabetes Mellitus
Jun Sung Moon
Diabetes & Metabolism Journal.2023; 47(3): 345. CrossRef - Smart Insulin Pen: Managing Insulin Therapy for People with Diabetes in the Digital Era
Jee Hee Yoo, Jae Hyeon Kim
The Journal of Korean Diabetes.2023; 24(4): 190. CrossRef
- Real-World Continuous Glucose Monitoring Data from a Population with Type 1 Diabetes in South Korea: Nationwide Single-System Analysis
- Guideline/Fact Sheet
- Insulin Fact Sheet in Type 1 and 2 Diabetes Mellitus and Trends of Antidiabetic Medication Use in Insulin Users with Type 2 Diabetes Mellitus: 2002 to 2019
- Jiyun Park, Gyuri Kim, Bong-Sung Kim, Kyung-Do Han, So Yoon Kwon, So Hee Park, You-Bin Lee, Sang-Man Jin, Jae Hyeon Kim
- Diabetes Metab J. 2023;47(2):211-219. Published online February 7, 2023
- DOI: https://doi.org/10.4093/dmj.2022.0346

- 3,655 View
- 263 Download
- 3 Web of Science
- 4 Crossref
-
 Abstract
Abstract
 PDF
PDF Supplementary Material
Supplementary Material PubReader
PubReader  ePub
ePub - Background
This study investigated the trends of insulin use among Korean patients with type 1 diabetes mellitus (T1DM) and type 2 diabetes mellitus (T2DM). Changes in prescription of antidiabetic medications in T2DM patients taking insulin therapy were evaluated.
Methods
We analyzed data from the National Health Insurance Service database in Korea to evaluate the prevalence of insulin users and trends of insulin use in T1DM and T2DM patients from January 2002 to December 2019. We also investigated numbers and types of antidiabetic medications in insulin users with T2DM.
Results
The overall total number of insulin users increased from 2002 to 2019, reaching 348,254 for T2DM and 20,287 for T1DM in 2019 compared with 109,974 for T2DM and 34,972 for T1DM in 2002. The proportion of patients using basal analogs and short acting analogs have increased and those using human insulin, premixed insulin, or biphasic human insulin have decreased (rapid acting analogs: 71.85% and 24.12% in T1DM and T2DM, respectively, in 2019; basal analogs: 76.75% and 75.09% in T1DM and T2DM, respectively, in 2019). The use of other antidiabetic medication in addition to insulin increased for T2DM, especially in dual therapy, reaching up to 52.35% in 2019 compared with 16.72% in 2002.
Conclusion
The proportion of the patients using basal or rapid acting analogs increased among all insulin users in both T1DM and T2DM patients. Among patients with T2DM, the proportion of patients using antidiabetic medications in addition to insulin was significantly increased compared to those who used insulin alone. -
Citations
Citations to this article as recorded by- Real-World Continuous Glucose Monitoring Data from a Population with Type 1 Diabetes in South Korea: Nationwide Single-System Analysis
Ji Yoon Kim, Sang-Man Jin, Sarah B. Andrade, Boyang Chen, Jae Hyeon Kim
Diabetes Technology & Therapeutics.2024;[Epub] CrossRef - Continuous glucose monitoring for glycaemic control and cardiovascular risk reduction in patients with type 2 diabetes not on insulin therapy: A clinical trial
Joseph Reed, Tony Dong, Elke Eaton, Janice Friswold, Jodie Porges, Sadeer G. Al‐Kindi, Sanjay Rajagopalan, Ian J. Neeland
Diabetes, Obesity and Metabolism.2024;[Epub] CrossRef - Evaluation of pharmacokinetic interactions between lobeglitazone, empagliflozin, and metformin in healthy subjects
Heeyoung Kim, Choon Ok Kim, Hyeonsoo Park, Min Soo Park, Dasohm Kim, Taegon Hong, Yesong Shin, Byung Hak Jin
Translational and Clinical Pharmacology.2023; 31(1): 59. CrossRef - Smart Insulin Pen: Managing Insulin Therapy for People with Diabetes in the Digital Era
Jee Hee Yoo, Jae Hyeon Kim
The Journal of Korean Diabetes.2023; 24(4): 190. CrossRef
- Real-World Continuous Glucose Monitoring Data from a Population with Type 1 Diabetes in South Korea: Nationwide Single-System Analysis
Review
- Technology/Device
- Advances in Continuous Glucose Monitoring and Integrated Devices for Management of Diabetes with Insulin-Based Therapy: Improvement in Glycemic Control
- Jee Hee Yoo, Jae Hyeon Kim
- Diabetes Metab J. 2023;47(1):27-41. Published online January 12, 2023
- DOI: https://doi.org/10.4093/dmj.2022.0271

- 6,473 View
- 389 Download
- 12 Web of Science
- 16 Crossref
-
 Abstract
Abstract
 PDF
PDF PubReader
PubReader  ePub
ePub - Continuous glucose monitoring (CGM) technology has evolved over the past decade with the integration of various devices including insulin pumps, connected insulin pens (CIPs), automated insulin delivery (AID) systems, and virtual platforms. CGM has shown consistent benefits in glycemic outcomes in type 1 diabetes mellitus (T1DM) and type 2 diabetes mellitus (T2DM) treated with insulin. Moreover, the combined effect of CGM and education have been shown to improve glycemic outcomes more than CGM alone. Now a CIP is the expected future technology that does not need to be worn all day like insulin pumps and helps to calculate insulin doses with a built-in bolus calculator. Although only a few clinical trials have assessed the effectiveness of CIPs, they consistently show benefits in glycemic outcomes by reducing missed doses of insulin and improving problematic adherence. AID systems and virtual platforms made it possible to achieve target glycosylated hemoglobin in diabetes while minimizing hypoglycemia, which has always been challenging in T1DM. Now fully automatic AID systems and tools for diabetes decisions based on artificial intelligence are in development. These advances in technology could reduce the burden associated with insulin treatment for diabetes.
-
Citations
Citations to this article as recorded by- Accuracy and Safety of the 15-Day CareSens Air Continuous Glucose Monitoring System
Kyung-Soo Kim, Seung-Hwan Lee, Won Sang Yoo, Cheol-Young Park
Diabetes Technology & Therapeutics.2024; 26(4): 222. CrossRef - Real-World Continuous Glucose Monitoring Data from a Population with Type 1 Diabetes in South Korea: Nationwide Single-System Analysis
Ji Yoon Kim, Sang-Man Jin, Sarah Andrade, Boyang Chen, Jae Hyeon Kim
Diabetes Technology & Therapeutics.2024;[Epub] CrossRef - Recent advances in the precision control strategy of artificial pancreas
Wuyi Ming, Xudong Guo, Guojun Zhang, Yinxia Liu, Yongxin Wang, Hongmei Zhang, Haofang Liang, Yuan Yang
Medical & Biological Engineering & Computing.2024;[Epub] CrossRef - Digital Health in Diabetes and Cardiovascular Disease
Dorothy Avoke, Abdallah Elshafeey, Robert Weinstein, Chang H. Kim, Seth S. Martin
Endocrine Research.2024; : 1. CrossRef - Continuous glucose monitoring with structured education in adults with type 2 diabetes managed by multiple daily insulin injections: a multicentre randomised controlled trial
Ji Yoon Kim, Sang-Man Jin, Kang Hee Sim, Bo-Yeon Kim, Jae Hyoung Cho, Jun Sung Moon, Soo Lim, Eun Seok Kang, Cheol-Young Park, Sin Gon Kim, Jae Hyeon Kim
Diabetologia.2024;[Epub] CrossRef - Glycemic Outcomes During Early Use of the MiniMed™ 780G Advanced Hybrid Closed-Loop System with Guardian™ 4 Sensor
Toni L. Cordero, Zheng Dai, Arcelia Arrieta, Fang Niu, Melissa Vella, John Shin, Andrew S. Rhinehart, Jennifer McVean, Scott W. Lee, Robert H. Slover, Gregory P. Forlenza, Dorothy I. Shulman, Rodica Pop-Busui, James R. Thrasher, Mark S. Kipnes, Mark P. Ch
Diabetes Technology & Therapeutics.2023; 25(9): 652. CrossRef - Navigating the Seas of Glycemic Control: The Role of Continuous Glucose Monitoring in Type 1 Diabetes Mellitus
Jun Sung Moon
Diabetes & Metabolism Journal.2023; 47(3): 345. CrossRef - APSec1.0: Innovative Security Protocol Design with Formal Security Analysis for the Artificial Pancreas System
Jiyoon Kim, Jongmin Oh, Daehyeon Son, Hoseok Kwon, Philip Virgil Astillo, Ilsun You
Sensors.2023; 23(12): 5501. CrossRef - Advances and Development of Electronic Neural Interfaces
Xue Jiaxiang, Liu Zhixin
Journal of Computing and Natural Science.2023; : 147. CrossRef - Continuous Glucose Monitoring (CGM) and Metabolic Control in a Cohort of Patients with Type 1 Diabetes and Coeliac Disease
Flavia Amaro, Maria Alessandra Saltarelli, Marina Primavera, Marina Cerruto, Stefano Tumini
Endocrines.2023; 4(3): 595. CrossRef - Comparison of Glycemia Risk Index with Time in Range for Assessing Glycemic Quality
Ji Yoon Kim, Jee Hee Yoo, Jae Hyeon Kim
Diabetes Technology & Therapeutics.2023; 25(12): 883. CrossRef - The Benefits Of Continuous Glucose Monitoring In Pregnancy
Jee Hee Yoo, Jae Hyeon Kim
Endocrinology and Metabolism.2023; 38(5): 472. CrossRef - The Growing Challenge of Diabetes Management in an Aging Society
Seung-Hwan Lee
Diabetes & Metabolism Journal.2023; 47(5): 630. CrossRef - Recent advances in artificial intelligence-assisted endocrinology and diabetes
Ioannis T. Oikonomakos, Ranjit M. Anjana, Viswanathan Mohan, Charlotte Steenblock, Stefan R. Bornstein
Exploration of Endocrine and Metabolic Disease.2023; 1(1): 16. CrossRef - An Observational Pilot Study of a Tailored Environmental Monitoring and Alert System for Improved Management of Chronic Respiratory Diseases
Mohammed Alotaibi, Fady Alnajjar, Badr A Alsayed, Tareq Alhmiedat, Ashraf M Marei, Anas Bushnag, Luqman Ali
Journal of Multidisciplinary Healthcare.2023; Volume 16: 3799. CrossRef - Smart Insulin Pen: Managing Insulin Therapy for People with Diabetes in the Digital Era
Jee Hee Yoo, Jae Hyeon Kim
The Journal of Korean Diabetes.2023; 24(4): 190. CrossRef
- Accuracy and Safety of the 15-Day CareSens Air Continuous Glucose Monitoring System
Original Articles
- Type 1 Diabetes
- Performance of Fast-Acting Aspart Insulin as Compared to Aspart Insulin in Insulin Pump for Managing Type 1 Diabetes Mellitus: A Meta-Analysis
- Deep Dutta, Ritin Mohindra, Kunal Mahajan, Meha Sharma
- Diabetes Metab J. 2023;47(1):72-81. Published online June 24, 2022
- DOI: https://doi.org/10.4093/dmj.2022.0035
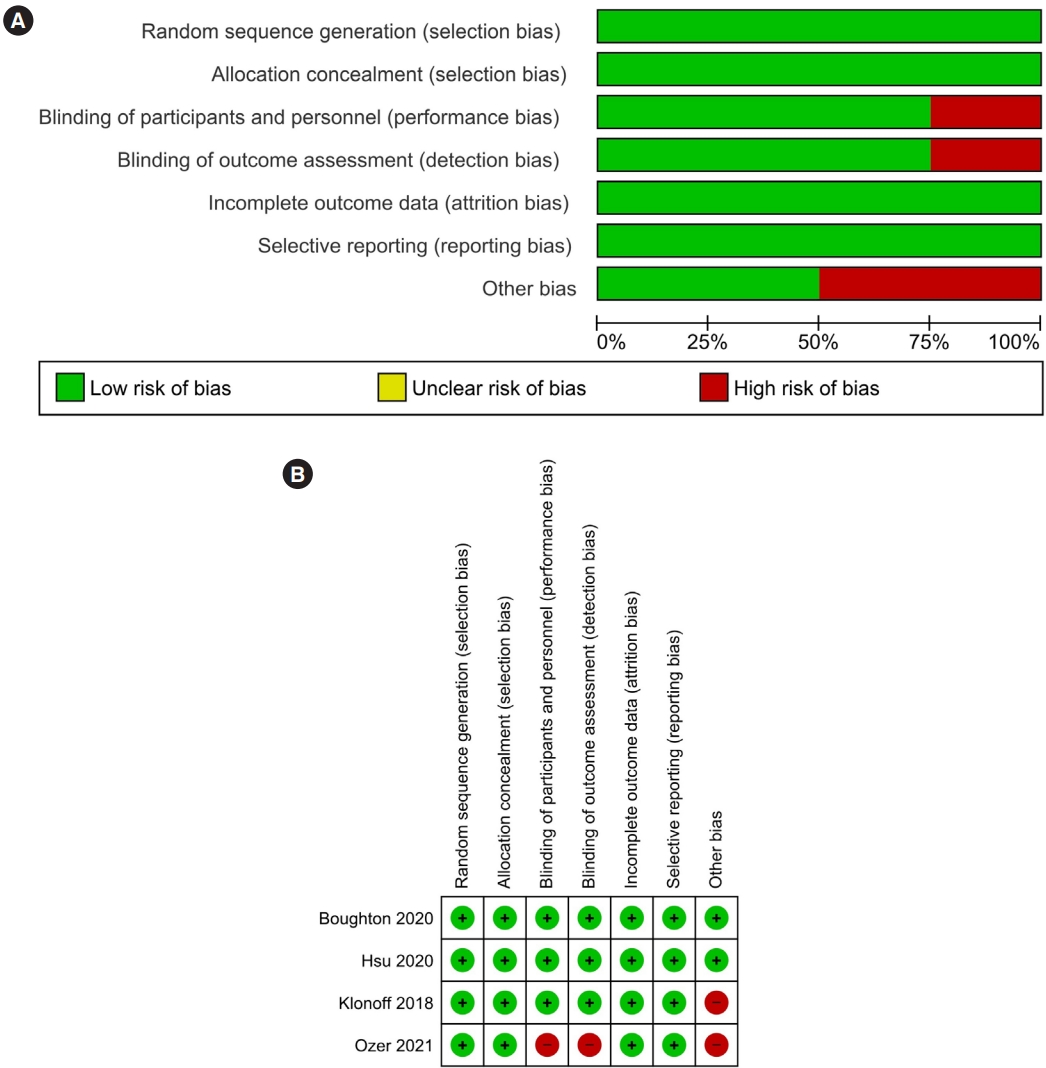
- 4,978 View
- 250 Download
- 1 Web of Science
- 2 Crossref
-
 Abstract
Abstract
 PDF
PDF Supplementary Material
Supplementary Material PubReader
PubReader  ePub
ePub - Background
No meta-analysis has analysed efficacy and safety of fast-acting aspart insulin (FIAsp) with insulin pump in type 1 diabetes mellitus (T1DM).
Methods
Electronic databases were searched for randomised controlled trials (RCTs) involving T1DM patients on insulin pump receiving FIAsp in intervention arm, and placebo/active comparator insulin in control arm. Primary outcome was to evaluate changes in 1- and 2-hour post-prandial glucose (1hPPG and 2hPPG). Secondary outcomes were to evaluate alterations in percentage time with blood glucose <3.9 mmol/L (hypoglycaemia), time in range (TIR) blood glucose 3.9 to 10 mmol/L, insulin requirements and adverse events.
Results
Data from four RCTs involving 640 patients was analysed. FIAsp use in insulin pump was associated with significantly greater lowering of 1hPPG (mean difference [MD], –1.35 mmol/L; 95% confidence interval [CI], –1.72 to –0.98; P<0.01; I2=63%) and 2hPPG (MD, –1.19 mmol/L; 95% CI, –1.38 to –1.00; P<0.01; I2=0%) as compared to controls. TIR was comparable among groups (MD, 1.06%; 95% CI, –3.84 to 5.96; P=0.67; I2=70%). Duration of blood glucose <3.9 mmol/L was lower in FIAsp group, approaching significance (MD, –0.91%; 95% CI, –1.84 to 0.03; P=0.06; I2=0%). Total hypoglycaemic episodes (risk ratio [RR], 1.35; 95% CI, 0.55 to 3.31; P=0.51; I2=0%), severe hypoglycaemia (RR, 2.26; 95% CI, 0.77 to 6.66; P=0.14), infusion site reactions (RR, 1.35; 95% CI, 0.63 to 2.93; P=0.77; I2=0%), and treatment-emergent adverse events (RR, 1.13; 95% CI, 0.80 to 1.60; P=0.50; I2=0%) were comparable.
Conclusion
FIAsp use in insulin pump is associated with better post-prandial glycaemic control with no increased hypoglycaemia or glycaemic variability. -
Citations
Citations to this article as recorded by- Burden and Coping Strategies of Hypoglycemia in People with Diabetes
Aris Liakos, Thomas Karagiannis, Ioannis Avgerinos, Apostolos Tsapas, Eleni Bekiari
Current Diabetes Reviews.2024;[Epub] CrossRef - Efficacy and Safety of Ultra-rapid Lispro Insulin in Managing Type-1 and Type-2 Diabetes: A Systematic Review and Meta-Analysis
Deep Dutta, Lakshmi Nagendra, Saptarshi Bhattacharya, Meha Sharma
Indian Journal of Endocrinology and Metabolism.2023; 27(6): 467. CrossRef
- Burden and Coping Strategies of Hypoglycemia in People with Diabetes
- Type 1 Diabetes
- Identification of Key Genes and Pathways in Peripheral Blood Mononuclear Cells of Type 1 Diabetes Mellitus by Integrated Bioinformatics Analysis
- Xing Li, Mingyu Liao, Jiangheng Guan, Ling Zhou, Rufei Shen, Min Long, Jiaqing Shao
- Diabetes Metab J. 2022;46(3):451-463. Published online April 1, 2022
- DOI: https://doi.org/10.4093/dmj.2021.0018

- 7,038 View
- 292 Download
- 6 Web of Science
- 9 Crossref
-
 Abstract
Abstract
 PDF
PDF Supplementary Material
Supplementary Material PubReader
PubReader  ePub
ePub - Background
The onset and progression of type 1 diabetes mellitus (T1DM) is closely related to autoimmunity. Effective monitoring of the immune system and developing targeted therapies are frontier fields in T1DM treatment. Currently, the most available tissue that reflects the immune system is peripheral blood mononuclear cells (PBMCs). Thus, the aim of this study was to identify key PBMC biomarkers of T1DM.
Methods
Common differentially expressed genes (DEGs) were screened from the Gene Expression Omnibus (GEO) datasets GSE9006, GSE72377, and GSE55098, and PBMC mRNA expression in T1DM patients was compared with that in healthy participants by GEO2R. Gene Ontology, Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway and protein-protein interaction (PPI) network analyses of DEGs were performed using the Cytoscape, DAVID, and STRING databases. The vital hub genes were validated by reverse transcription-polymerase chain reaction using clinical samples. The disease-gene-drug interaction network was built using the Comparative Toxicogenomics Database (CTD) and Drug Gene Interaction Database (DGIdb).
Results
We found that various biological functions or pathways related to the immune system and glucose metabolism changed in PBMCs from T1DM patients. In the PPI network, the DEGs of module 1 were significantly enriched in processes including inflammatory and immune responses and in pathways of proteoglycans in cancer. Moreover, we focused on four vital hub genes, namely, chitinase-3-like protein 1 (CHI3L1), C-X-C motif chemokine ligand 1 (CXCL1), matrix metallopeptidase 9 (MMP9), and granzyme B (GZMB), and confirmed them in clinical PBMC samples. Furthermore, the disease-gene-drug interaction network revealed the potential of key genes as reference markers in T1DM.
Conclusion
These results provide new insight into T1DM pathogenesis and novel biomarkers that could be widely representative reference indicators or potential therapeutic targets for clinical applications. -
Citations
Citations to this article as recorded by- Single-cell and transcriptomic analyses reveal the influence of diabetes on ovarian cancer
Zhihao Zhao, Qilin Wang, Fang Zhao, Junnan Ma, Xue Sui, Hyok Chol Choe, Peng Chen, Xue Gao, Lin Zhang
BMC Genomics.2024;[Epub] CrossRef - Bioinformatics analysis identifies TGF-β signaling pathway-associated molecular subtypes and gene signature in diabetic foot
Guanggang Du, Jie Chen, Xuezhu Zhu, Zongdong Zhu
iScience.2024; 27(3): 109094. CrossRef - Identification of Comorbidities, Genomic Associations, and Molecular Mechanisms for COVID-19 Using Bioinformatics Approaches
Shudeb Babu Sen Omit, Salma Akhter, Humayan Kabir Rana, A. R. M. Mahamudul Hasan Rana, Nitun Kumar Podder, Mahmudul Islam Rakib, Ashadun Nobi, Ali Imran
BioMed Research International.2023; 2023: 1. CrossRef - Advanced Delivery Strategies for Immunotherapy in Type I Diabetes Mellitus
Mingshu Huang, Weixing Chen, Min Wang, Yisheng Huang, Hongyu Liu, Yue Ming, Yuanxin Chen, Zhengming Tang, Bo Jia
BioDrugs.2023; 37(3): 331. CrossRef - Identification of the key genes of tuberculosis and construction of a diagnostic model via weighted gene co-expression network analysis
Baiying Li, Lifang Sun, Yaping Sun, Libo Zhen, Qi Qi, Ting Mo, Huijie Wang, Meihua Qiu, Qingshan Cai
Journal of Infection and Chemotherapy.2023; 29(11): 1046. CrossRef - Probing biological network in concurrent carcinomas and Type-2 diabetes for potential biomarker screening: An advanced computational paradigm
Abdullah Al Marzan, Shatila Shahi, Md Sakil Arman, Md Zafrul Hasan, Ajit Ghosh
Advances in Biomarker Sciences and Technology.2023; 5: 89. CrossRef - Transcriptional analysis of human peripheral blood mononuclear cells stimulated by Mycobacterium tuberculosis antigen
Jing Wei, Fangzheng Guo, Yamin Song, Kun Xu, Feiyang Lin, Kangsheng Li, Baiqing Li, Zhongqing Qian, Xiaojing Wang, Hongtao Wang, Tao Xu
Frontiers in Cellular and Infection Microbiology.2023;[Epub] CrossRef - Combining bioinformatics and machine learning algorithms to identify and analyze shared biomarkers and pathways in COVID-19 convalescence and diabetes mellitus
Jinru Shen, Yaolou Wang, Xijin Deng, Si Ri Gu Leng Sana
Frontiers in Endocrinology.2023;[Epub] CrossRef - Transcriptome analysis of peripheral blood mononuclear cells in patients with type 1 diabetes mellitus
Zhaoxiang Wang, Li Zhang, Fengyan Tang, Zhongming Yang, Mengzhu Wang, Jue Jia, Dong Wang, Ling Yang, Shao Zhong, Guoyue Yuan
Endocrine.2022; 78(2): 270. CrossRef
- Single-cell and transcriptomic analyses reveal the influence of diabetes on ovarian cancer
Short Communication
- Others
- Comparison of Insulin-Treated Patients with Ambiguous Diabetes Type with Definite Type 1 and Type 2 Diabetes Mellitus Subjects: A Clinical Perspective
- Insa Laspe, Juris J. Meier, Michael A. Nauck
- Diabetes Metab J. 2023;47(1):140-146. Published online March 22, 2022
- DOI: https://doi.org/10.4093/dmj.2021.0322

- 65,535 View
- 184 Download
-
 Abstract
Abstract
 PDF
PDF Supplementary Material
Supplementary Material PubReader
PubReader  ePub
ePub - In clinical practice, the distinction between type 1 diabetes mellitus (T1DM) and type 2 diabetes mellitus (T2DM) can be challenging, leaving patients with “ambiguous” diabetes type. Insulin-treated patients (n=115) previously diagnosed with T2DM had to be re-classified based on clinical phenotype and laboratory results, and were operationally defined as having an ambiguous diabetes type. They were compared against patients with definite T1DM and T2DM regarding 12 clinical and laboratory features typically different between diabetes types. Characteristics of patients with ambiguous diabetes type, representing approximately 6% of all patients with T1DM or T2DM seen at our specialized clinic, fell in between those of patients with definite T1DM and T2DM, both regarding individual features and with respect to a novel classification based on multi-variable regression analysis (P<0.0001). In conclusion, a substantial proportion of diabetes patients in a tertiary care centre presented with an “ambiguous” diabetes type. Their clinical characteristics fall in between those of definite T1DM or T2DM patients.
Original Article
- Type 1 Diabetes
- Abnormal Responses in Cognitive Impulsivity Circuits Are Associated with Glycosylated Hemoglobin Trajectories in Type 1 Diabetes Mellitus and Impaired Metabolic Control
- Helena Jorge, Isabel C. Duarte, Sandra Paiva, Ana Paula Relvas, Miguel Castelo-Branco
- Diabetes Metab J. 2022;46(6):866-878. Published online March 22, 2022
- DOI: https://doi.org/10.4093/dmj.2021.0307
- 4,456 View
- 173 Download
- 3 Web of Science
- 3 Crossref
-
 Abstract
Abstract
 PDF
PDF Supplementary Material
Supplementary Material PubReader
PubReader  ePub
ePub - Background
Risky health decisions and impulse control profiles may impact on metabolic control in type 1 diabetes mellitus (T1DM). We hypothesize that the neural correlates of cognitive impulsivity and decision-making in T1DM relate to metabolic control trajectories.
Methods
We combined functional magnetic resonance imaging (fMRI), measures of metabolic trajectories (glycosylated hemoglobin [HbA1c] over multiple time points) and behavioral assessment using a cognitive impulsivity paradigm, the Balloon Analogue Risk Task (BART), in 50 participants (25 T1DM and 25 controls).
Results
Behavioral results showed that T1DM participants followed a rigid conservative risk strategy along the iterative game. Imaging group comparisons showed that patients showed larger activation of reward related, limbic regions (nucleus accumbens, amygdala) and insula (interoceptive saliency network) in initial game stages. Upon game completion differences emerged in relation to error monitoring (anterior cingulate cortex [ACC]) and inhibitory control (inferior frontal gyrus). Importantly, activity in the saliency network (ACC and insula), which monitors interoceptive states, was related with metabolic trajectories, which was also found for limbic/reward networks. Parietal and posterior cingulate regions activated both in controls and patients with adaptive decision-making, and positively associated with metabolic trajectories.
Conclusion
We found triple converging evidence when comparing metabolic trajectories, patients versus controls or risk averse (non-learners) versus patients who learned by trial and error. Dopaminergic reward and saliency (interoceptive and error monitoring) circuits show a tight link with impaired metabolic trajectories and cognitive impulsivity in T1DM. Activity in parietal and posterior cingulate are associated with adaptive trajectories. This link between reward-saliency-inhibition circuits suggests novel strategies for patient management. -
Citations
Citations to this article as recorded by- The usefulness of an intervention with a serious video game as a complementary approach to cognitive behavioural therapy in eating disorders: A pilot randomized clinical trial for impulsivity management
Cristina Vintró‐Alcaraz, Núria Mallorquí‐Bagué, María Lozano‐Madrid, Giulia Testa, Roser Granero, Isabel Sánchez, Janet Treasure, Susana Jiménez‐Murcia, Fernando Fernández‐Aranda
European Eating Disorders Review.2023; 31(6): 781. CrossRef - Adaptations of the balloon analog risk task for neuroimaging settings: a systematic review
Charline Compagne, Juliana Teti Mayer, Damien Gabriel, Alexandre Comte, Eloi Magnin, Djamila Bennabi, Thomas Tannou
Frontiers in Neuroscience.2023;[Epub] CrossRef - Trust-based health decision-making recruits the neural interoceptive saliency network which relates to temporal trajectories of Hemoglobin A1C in Diabetes Type 1
Helena Jorge, Isabel C. Duarte, Miguel Melo, Ana Paula Relvas, Miguel Castelo-Branco
Brain Imaging and Behavior.2023; 18(1): 171. CrossRef
- The usefulness of an intervention with a serious video game as a complementary approach to cognitive behavioural therapy in eating disorders: A pilot randomized clinical trial for impulsivity management
Short Communications
- Pathophysiology
- Glutamic Acid Decarboxylase and Tyrosine Phosphatase-Related Islet Antigen-2 Positivity among Children and Adolescents with Diabetes in Korea
- Ka Young Kim, Min Seung Kim, Yun Jeong Lee, Young Ah Lee, Seong Yong Lee, Choong Ho Shin, Jae Hyun Kim
- Diabetes Metab J. 2022;46(6):948-952. Published online March 8, 2022
- DOI: https://doi.org/10.4093/dmj.2021.0332
- 4,598 View
- 183 Download
- 3 Web of Science
- 2 Crossref
-
 Abstract
Abstract
 PDF
PDF Supplementary Material
Supplementary Material PubReader
PubReader  ePub
ePub - Autoantibodies against glutamic acid decarboxylase (GADA), tyrosine phosphatase-related islet antigen 2 (IA2A), insulin (INSA), and islet cells (ICA) are critical for determining the type of diabetes and management strategy in new-onset diabetes mellitus (NODM), but there have been few reports of all diabetes-associated autoantibody (DAA) in Korea. We retrospectively analyzed 193 patients with NODM aged 0 to 18 years who were followed at two tertiary centers in Korea (2017 to 2021). Patients with type 1 diabetes mellitus (T1DM) and type 2 diabetes mellitus (T2DM) were 93 (48.2%) and 100 (51.8%), respectively. In T1DM patients, the DAA positivity rate was 94.6%; prevalence of GADA, IA2A, INSA, and ICA was 71.0%, 71.0%, 31.2%, and 10.8%, respectively; and IA2A added 10.7% point autoantibody positivity (83.9% for GADA+INSA+ICA and 94.6% for GADA+INSA+ICA+IA2A). Among the patients with T2DM, 12 (12.0%) were positive for DAA, and all were positive for INSA. These findings suggest that DAA at diagnosis, especially GADA and IA2A, is useful for classifying diabetes in Korean children and adolescents.
-
Citations
Citations to this article as recorded by- Immune-Checkpoint Inhibitors-Induced Type 1 Diabetes Mellitus: From Its Molecular Mechanisms to Clinical Practice
Yun Kyung Cho, Chang Hee Jung
Diabetes & Metabolism Journal.2023; 47(6): 757. CrossRef - Diagnostic and Therapeutic Strategies of Type 2 Diabetes Mellitus in Youth
Hwa Young Kim, Jae Hyun Kim
The Ewha Medical Journal.2022;[Epub] CrossRef
- Immune-Checkpoint Inhibitors-Induced Type 1 Diabetes Mellitus: From Its Molecular Mechanisms to Clinical Practice
- Technology/Device
- A 4-Week, Two-Center, Open-Label, Single-Arm Study to Evaluate the Safety and Efficacy of EOPatch in Well-Controlled Type 1 Diabetes Mellitus
- Jiyun Park, Nammi Park, Sangjin Han, You-Bin Lee, Gyuri Kim, Sang-Man Jin, Woo Je Lee, Jae Hyeon Kim
- Diabetes Metab J. 2022;46(6):941-947. Published online March 8, 2022
- DOI: https://doi.org/10.4093/dmj.2021.0299
- 5,246 View
- 269 Download
- 3 Web of Science
- 3 Crossref
-
 Abstract
Abstract
 PDF
PDF Supplementary Material
Supplementary Material PubReader
PubReader  ePub
ePub - This study evaluated the safety and efficacy of tubeless patch pump called EOPatch in patients with well-controlled type 1 diabetes mellitus (T1DM). This 4-week, two-center, open-label, single-arm study enrolled 10 adult patients diagnosed with T1DM with glycosylated hemoglobin less than 7.5%. The co-primary end points were patch pump usage time for one attachment and number of serious adverse events related to the patch pump. The secondary end points were total amount of insulin injected per patch and changes in glycemic parameters including continuous glucose monitoring data compared to those at study entry. The median usage time per patch was 84.00 hours (interquartile range, 64.50 to 92.50). Serious adverse events did not occur during the trial. Four weeks later, time in range 70 to 180 mg/dL was significantly improved (70.71%±17.14 % vs. 82.96%±9.14%, P=0.01). The times spent below range (<54 mg/dL) and above range (>180 mg/dL) also improved (All P<0.05). Four-week treatment with a tubeless patch pump was safe and led to clinical improvement in glycemic control.
-
Citations
Citations to this article as recorded by- Multilayer track‐etched membrane‐based electroosmotic pump for drug delivery
Qian Yang, Zebo Zhang, Junshu Lin, Boyu Zhu, Rongying Yu, Xinru Li, Bin Su, Bo Zhao
ELECTROPHORESIS.2024; 45(5-6): 433. CrossRef - Comparison between a tubeless, on-body automated insulin delivery system and a tubeless, on-body sensor-augmented pump in type 1 diabetes: a multicentre randomised controlled trial
Ji Yoon Kim, Sang-Man Jin, Eun Seok Kang, Soo Heon Kwak, Yeoree Yang, Jee Hee Yoo, Jae Hyun Bae, Jun Sung Moon, Chang Hee Jung, Ji Cheol Bae, Sunghwan Suh, Sun Joon Moon, Sun Ok Song, Suk Chon, Jae Hyeon Kim
Diabetologia.2024;[Epub] CrossRef - A true continuous healthcare system for type 1 diabetes
Jiyong Kim, Salman Khan, Eun Kyu Kim, Hye-Jun Kil, Bo Min Kang, Hyo Geon Lee, Jin-Woo Park, Jun Young Yoon, Woochul Kim
Nano Energy.2023; 113: 108553. CrossRef
- Multilayer track‐etched membrane‐based electroosmotic pump for drug delivery

 KDA
KDA
 First
First Prev
Prev





