Subclinical Hypothyroidism Is Independently Associated with Microalbuminuria in a Cohort of Prediabetic Egyptian Adults
Article information
Abstract
Background
Recent evidence has suggested an association between subclinical hypothyroidism (SCH) and microalbuminuria in patients with type 2 diabetes. However, whether SCH is related to microalbuminuria among subjects with prediabetes has not been studied. Thus, we evaluated the association between SCH and microalbuminuria in a cohort of prediabetic Egyptian adults.
Methods
A total of 147 prediabetic subjects and 150 healthy controls matched for age and sex were enrolled in this study. Anthropometric measurements, plasma glucose, lipid profile, homeostasis model assessment of insulin resistance (HOMA-IR), thyroid stimulating hormone (TSH), free thyroxine, triiodothyronine levels, and urinary albumin-creatinine ratio (UACR) were assessed.
Results
The prevalence of SCH and microalbuminuria in the prediabetic subjects was higher than that in the healthy controls (16.3% vs. 4%, P<0.001; and 12.9% vs. 5.3%, P=0.02, respectively). Prediabetic subjects with SCH were characterized by significantly higher HOMA-IR, TSH levels, UACR, and prevalence of microalbuminuria than those with euthyroidism. TSH level was associated with total cholesterol (P=0.05), fasting insulin (P=0.01), HOMA-IR (P=0.01), and UACR (P=0.005). UACR was associated with waist circumference (P=0.01), fasting insulin (P=0.05), and HOMA-IR (P=0.02). With multiple logistic regression analysis, SCH was associated with microalbuminuria independent of confounding variables (β=2.59; P=0.01).
Conclusion
Our findings suggest that prediabetic subjects with SCH demonstrate higher prevalence of microalbuminuria than their non-SCH counterparts. SCH is also independently associated with microalbuminuria in prediabetic subjects. Screening and treatment for SCH may be warranted in those patients.
INTRODUCTION
Prediabetes is a high-risk state for diabetes that is typically defined as blood glucose concentrations higher than normal, but lower than diabetes thresholds. According to World Health Organization (WHO), people are at high risk of developing diabetes if they have one of two distinct states: impaired fasting glucose (IFG) defined as a fasting plasma glucose (FPG) concentration of ≥6.1 and <7.0 mmol/L, without impaired glucose tolerance (IGT), and IGT defined as a FPG concentration of <7.0 mmol/L and a 2-hour postload plasma glucose concentration of ≥7.8 and <11.1 mmol/L, measured during a 75 g oral glucose tolerance test [1]. Prevalence of prediabetes is increasing worldwide and experts have projected that more than 470 million people will have prediabetes by 2030 [2]. Around 5% to 10% of people with prediabetes become diabetic every year, with the same proportion converting back to normoglycaemia, although the conversion rate varies with population characteristics and prediabetes definitions [3,4].
The presence of microalbuminuria has become an important tool for therapeutic intervention [5]. Microalbuminuria is an early sign of an increased risk for developing kidney disease in patients with diabetes [6]. Prediabetes has been linked to increased risk of early forms of nephropathy and chronic kidney disease, defined by methods such as urinary albumin excretion rate and estimated glomerular filtration rate [7,8].
Subclinical hypothyroidism (SCH) is a common endocrine disorder and has been reported to range from 4% to 10% in large general population screening surveys [9] and has been found to be 4% to 17% in diabetic patients in previous studies [10-12]. Patients with SCH are usually asymptomatic, but may have cardiac dysfunction, elevated low density lipoprotein and neuropsychiatric symptoms [13]. SCH characterized by elevated thyroid stimulating hormone (TSH) levels with normal free thyroxine (FT4) and triiodothyronine (FT3) concentrations [14].
Both SCH and albuminuria are independent risk factors of cardiovascular disease [15,16]. Recent evidence has suggested an independent association between SCH and microalbuminuria in patients with type 2 diabetes [17]. However, whether SCH is related to microalbuminuria among subjects with prediabetes has not been studied. In this study, we evaluated the possible association between SCH and microalbuminuria among a cohort of Egyptian adults with prediabetes.
METHODS
The study comprised 147 adults with prediabetes aged 45.6±5.2 years (63 men and 84 women) and 150 age and sex matched healthy controls. Prediabetic subjects were recruited from Endocrinology and Obesity outpatient clinics at Specialized Medical Hospital, Mansoura University Faculty of Medicine, Egypt, during the period from May 2012 to January 2013. Healthy controls were recruited from the same geographic area; Mansoura city; and with the same exclusion criteria for prediabetic subjects. All subjects signed an informed consent to be included in our study. The study was approved by the local ethical committee.
All participants were subjected to thorough medical history and clinical examination. Anthropometric measurements including height, weight, body mass index (BMI) and waist circumference (WC) were obtained using standardized techniques; height was measured to the nearest 0.5 cm, body weight was measured to the nearest 0.1 kg, BMI was calculated as weight in kilograms divided by height in meters squared (kg/m2), WC was measured at the highest point of the iliac crest. Blood pressure was taken in the sitting position after 10 minutes rest using a random-zero sphygmomanometer. Adults diagnosed with prediabetes based on WHO criteria [1]. SCH was diagnosed by elevated TSH (>4 to 10 µU/mL) levels with normal FT4 and FT3 levels [14]. Urinary albumin-creatinine ratio (UACR) was determined using 24-hour urine samples and defined as milligram of albumin per gram of creatinine (mg/g). Microalbuminuria was defined by a UACR of 30 to 300 mg/g.
Exclusion criteria were acute or chronic renal failure, glomerulonephritis, overt proteinuria, hypertension or the use of antihypertensive medications, thyroid disorders other than SCH, overt hypothyroidism, hepatic dysfunction, infection, connective tissue disorders, malignancy, pregnancy, women on birth control pills or hormone replacement therapy, and smoking. We also excluded patients taking drugs that could influence thyroid function or proteinuria, e.g., angiotensin-converting enzyme inhibitors and angiotensin II receptor blockers, thiazide diuretics, β-blockers, amiodarone, or lithium.
Laboratory assay
FPG and 2-hour postload plasma glucose were estimated using commercially available kit, Cobas (Integra-400) supplied by Roche Diagnostic (Basel, Germany). Fasting serum insulin was assayed by a solid-phase, enzyme-labeled chemiluminescent immunometric assay using immulite analyzer supplied by Siemens (DPC; Cirrus Inc., Los Angeles, CA, USA). Homeostasis model assessment of insulin resistance (HOMA-IR) was calculated with the formula: HOMA-IR=fasting insulin (µU/mL)×fasting glucose (mmol/L)/22.5 [18]. Hemoglobin A1c (HbA1c) was measured as an index of metabolic control on a DCA 2000 analyzer, fast ion exchange resin (Roche Diagnostic). The reference range was 4.4% to 6.4%. Total cholesterol (TC), triglyceride (TG), and high density lipoprotein cholesterol (HDL-C) were assayed by commercially available kits, Cobas (Integra-400) supplied by Roche Diagnostic. Low density lipoprotein cholesterol (LDL-C) was calculated according to Friedewald et al. [19]. FT4, FT3, and TSH were measured by electrochemiluminecent immunoassay, using Elecsys 2010 (Roche Diagnostic). UACR was measured by an immunonephelometric method using Microalbuminuria (AIBT2) kit, Cobas (Integra-400) supplied by Roche Diagnostic.
Statistical methods
Data entry and analysis were performed using SPSS statistical package version 17 (SPSS Inc., Chicago, IL, USA). The data were expressed as mean±standard deviation for continuous data, frequency and proportion for categorical data, and median (minimum to maximum) for skewed data. Student t-test and Mann-Whitney were used to compare two groups. A chi-square test was used to compare categorical data. Pearson and Spearman correlations were done to study relation between different items. Multiple logistic regression analysis was performed to evaluate the association of SCH with microalbuminuria. Microalbuminuria, WC, fasting insulin, and HOMA-IR were entered in the regression model. P≤0.05 was considered as significant.
RESULTS
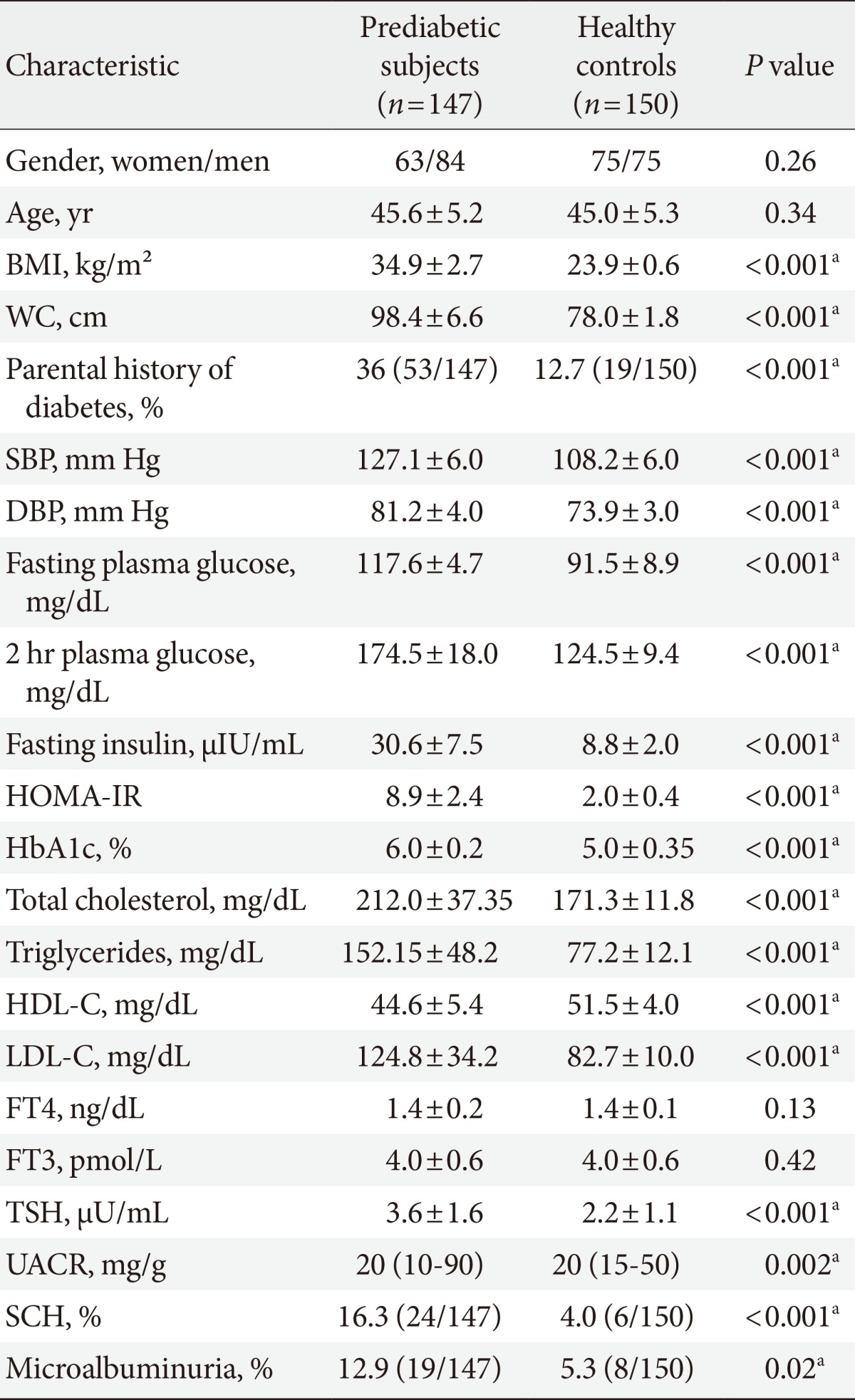
The baseline characteristics of the 147 prediabetic subjects and healthy controls were described in Table 1. Prediabetic subjects had significantly higher BMI, WC, blood pressure, parental history of diabetes, fasting and 2-hour plasma glucose, HbA1c, fasting insulin and HOMA-IR, TC, TG and LDL-C than healthy controls, however, HDL-C was significantly lower in prediabetic subjects than in healthy controls. TSH and UACR were also significantly higher in prediabetic subjects than in healthy controls (3.59±1.59 vs. 2.23±1.1, P<0.001; and 20 [10 to 90] vs. 20 [15 to 50], P=0.002, respectively). The prevalence of SCH in prediabetic subjects was 16.3% (24/147) compared to 4% (6/150) in healthy controls, P<0.001. SCH was prevalent in women than in men in prediabetic subjects (66.7% [16/24] vs. 33.3% [8/24]; P=0.01). The prevalence of microalbuminuria in prediabetic subjects was 12.9% (19/147) compared to 5.3% (8/150) in healthy controls, P=0.02.
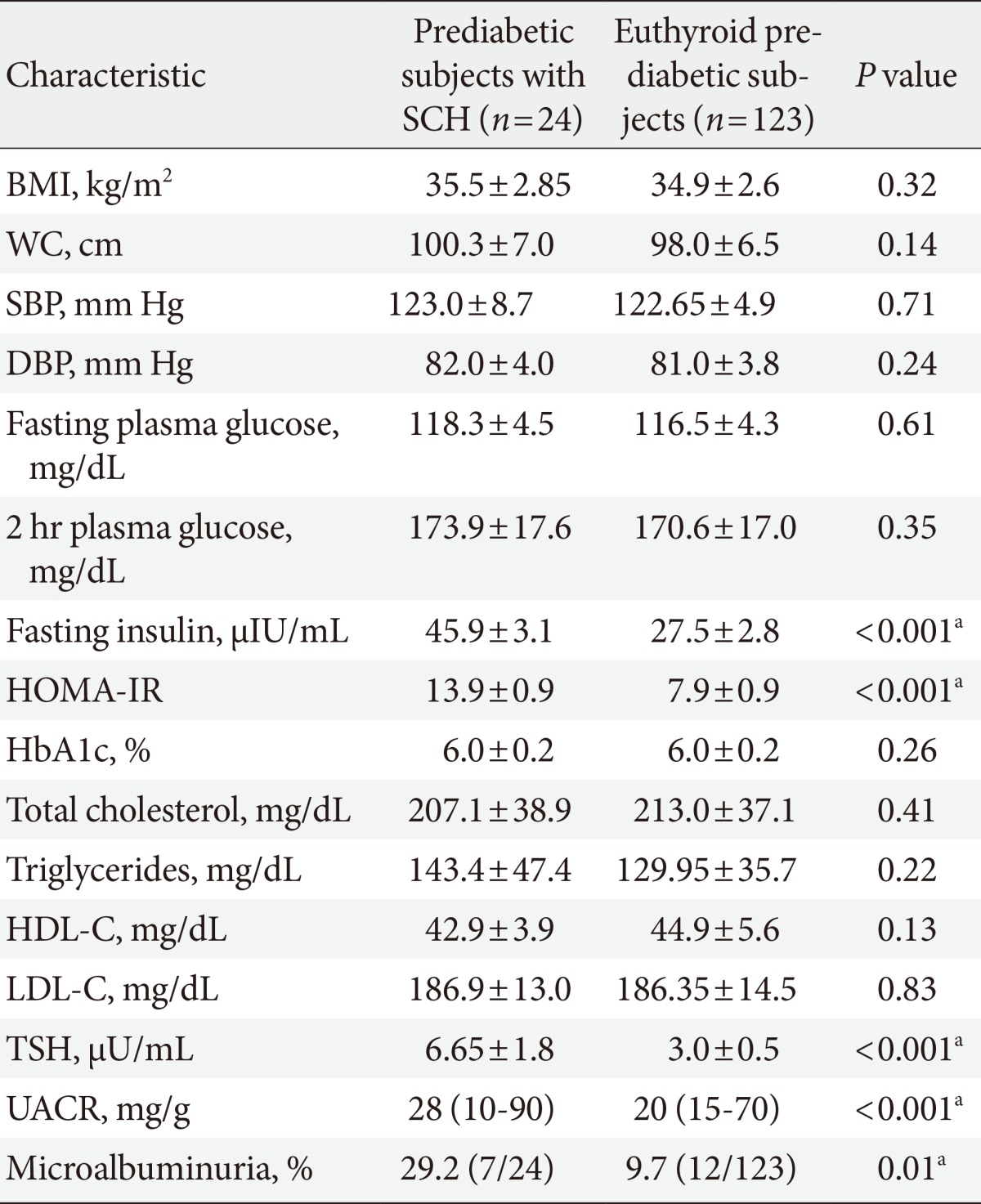
Prediabetes participants were divided into two groups according to thyroid function: patients with SCH or those with euthyroidism. Prediabetic subjects with SCH were characterized by significantly higher fasting insulin, HOMA-IR, TSH levels (6.65±1.76 mU/mL vs. 2.99±0.48 mU/mL; P<0.001), UACR (28 [10 to 90] vs. 20 [15 to 70]; P<0.001) and prevalence of microalbuminuria (29.2% vs. 9.7%; P=0.01) than those with euthyroidism, whereas BMI, WC, blood pressure, fasting, 2-hour plasma glucose, HbA1c, and lipid profile were not different between the two groups (Table 2).
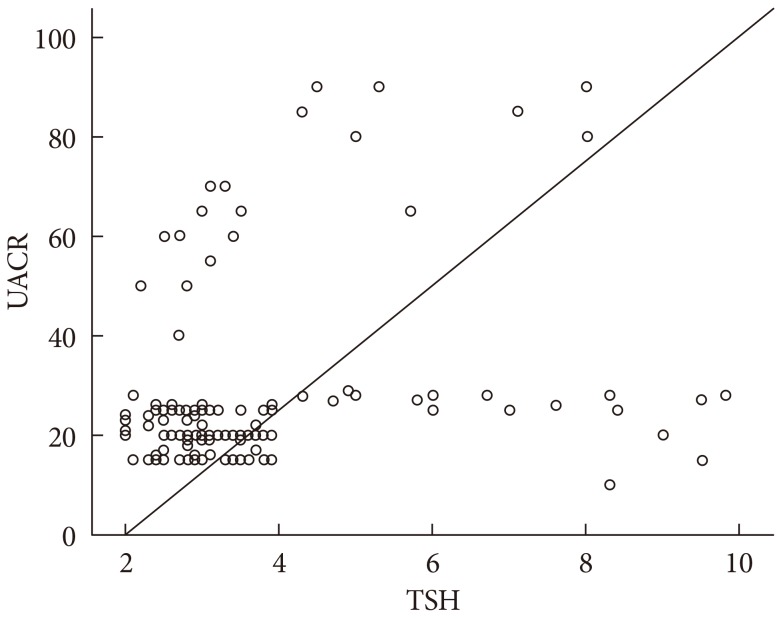
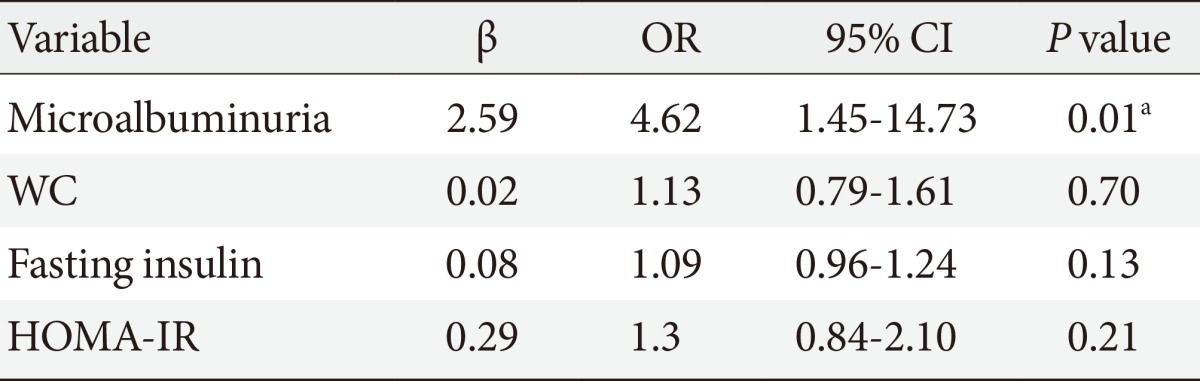
TSH level was significantly associated with fasting insulin (r=0.18, P=0.01), HOMA-IR (r=0.21, P=0.01), and TC (r=0.16, P=0.05). On the other hand, UACR was significantly associated with WC (r=0.21, P=0.01), fasting insulin (r=0.16, P=0.05), and HOMA-IR (r=0.19, P=0.02) (Table 3). TSH was significantly associated with UACR (r=0.23, P=0.005) (Fig. 1); furthermore, multiple logistic regression analysis demonstrated that SCH was associated with microalbuminuria independent of WC, fasting insulin, and HOMA-IR (β=2.59; odds ratio, 4.62; 95% confidence interval, 1.45 to 14.73; P=0.01) (Table 4). There was insignificant association between UACR and TSH in healthy controls (r=0.07, P=0.4).
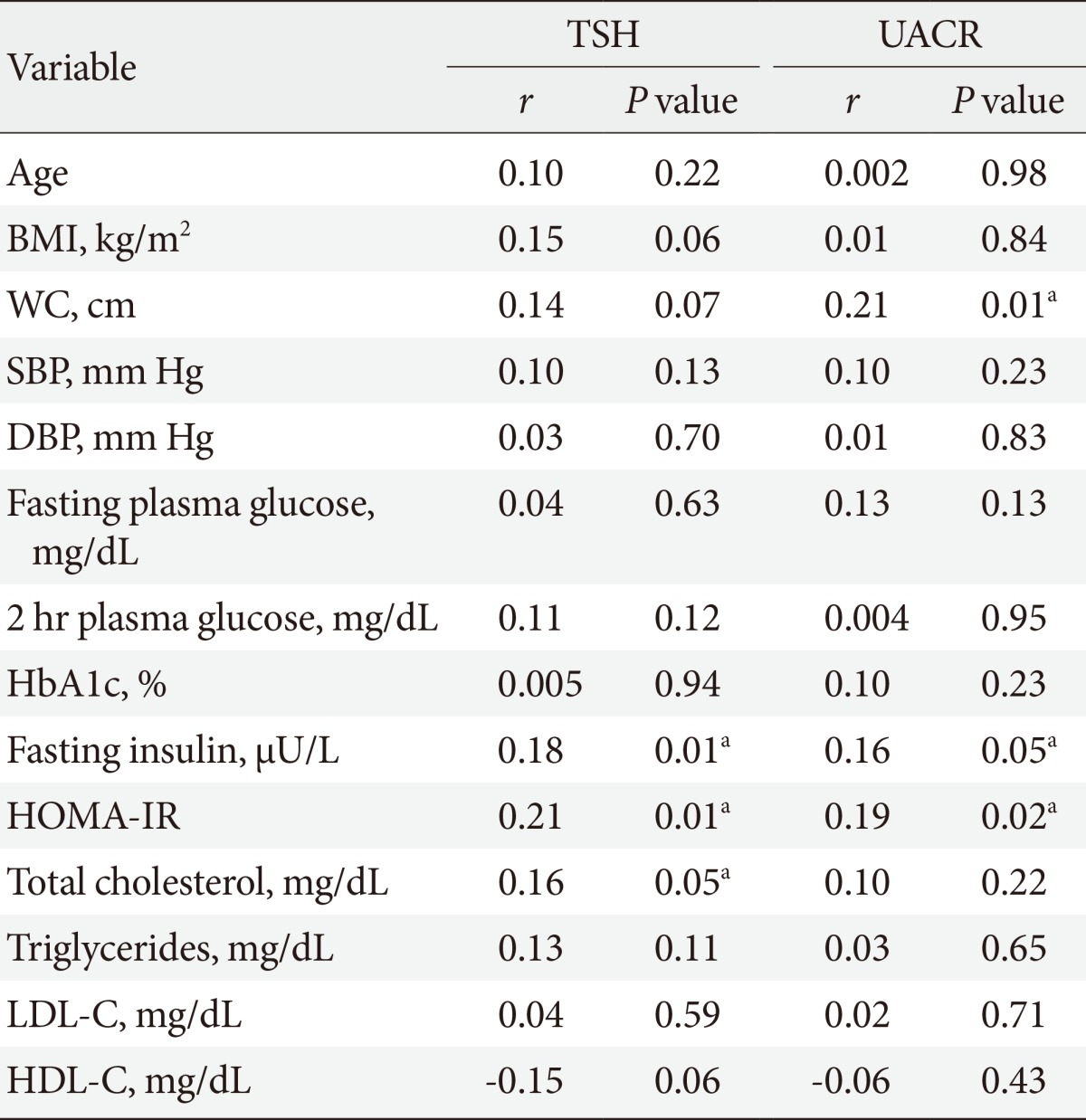
Correlation between thyroid stimulating hormone and urinary albumin-creatinine ratio with other parameters in prediabetic subjects
DISCUSSION
The current study examined, for the first time, the relationship between SCH and microalbuminuria in prediabetic subjects. The main finding was the association between TSH level and UACR; furthermore, SCH was an independent risk factor for microalbuminuria.
The prediabetic subjects in our study had higher BMI, WC, parental history of diabetes, blood pressure, fasting insulin, HOMA-IR, and lipids than healthy controls. In support, American Diabetes Association [20] reported that overweight/obesity and family history of diabetes are risk factors for prediabetes. On the other hand, blood glucose in the prediabetic range is correlated with many risk factors, including general and central obesity, blood pressure, and TG and lipoprotein concentrations [21].
In the present study, prediabetic subjects had higher prevalence of microalbuminuria than healthy controls (12.9% vs. 5.3%; P=0.02). This is in agreement with Hwang et al. [22] who found that the prevalence of microalbuminuria among 947 prediabetic subjects was 12.8%. National Health and Nutrition Examination Survey, 1999 to 2006, showed that the prevalence of microalbuminuria increases as glycaemia worsens, i.e., from 6% in normoglycaemia, to 10% in IFG and 29% in undiagnosed diabetes or diagnosed diabetes [8].
Our results also showed positive correlations between UACR and WC, insulin, and HOMA-IR. In accordance, Chandie Shaw et al. [23] demonstrated that central obesity is an independent risk factor of albuminuria in healthy individuals. In addition, it has been suggested that microalbuminuria is associated with insulin resistance [24,25] and may precede the development of type 2 diabetes [26].
Haffner et al. [27] previously demonstrated that microalbuminuria may be a feature of the prediabetic state. Moreover, longitudinal studies suggest that prediabetes is a risk factor for chronic kidney disease, but whether this association is attributable to the effects of prediabetes itself, increased incidence of diabetes, or common causes contributing to both hyperglycaemia and kidney pathology is unclear [28,29]. From a mechanistic perspective, subtle damage occurs within the kidney in the prediabetic state whether from chronic exposure to abnormal glycemia that is below the formal threshold for diabetes, elevated intrarenal blood pressure, or oxidative stress [30]. Alternatively, increased UACR could be an early organ-specific manifestation of insulin resistance that heralds the onset of diabetes [31].
The association between type 2 diabetes and SCH is well recognized with reported prevalence of 4% to 17% [12], however, little is known about the prevalence of SCH in those with prediabetes. In the current study, the prevalence of SCH among prediabetics was 16.3% compared to 4% in healthy controls. We confirmed that SCH was prevalent in women than in men; this is in parallel with previous research studies which demonstrated high prevalence of SCH in type 2 diabetic women [32,33].
We observed significant association between serum TSH level and TC, fasting insulin, and HOMA-IR. These results parallel those of Chubb et al. [34], who found that small variations in TSH might exert a marked effect on lipid levels in diabetic adults. Furthermore, fasting hyperinsulinemia has been previously reported in patients with SCH [35]. Moreover, Maratou et al. [36] confirmed that patients with SCH have insulin resistance that is comparable with that of the patients with hypothyroidism. In patients with SCH, impairment in insulin-stimulated glucose disposal is caused by impaired translocation of GLUT4 transporters on the cell surface [36].
In the current study, prediabetic subjects with SCH demonstrated higher UACR and prevalence of microalbuminuria than their non-SCH counterparts. In addition, multiple logistic regression analysis revealed that SCH was independently associated with microalbuminuria in prediabetic subjects. In line, Chen et al. [37] demonstrated that type 2 diabetic patients with SCH are associated with an increased risk of nephropathy after adjustment for other factors. On the other hand, proteinuria had higher TSH levels [38].
Prediabetes is associated with the simultaneous presence of insulin resistance and β-cell dysfunction; abnormalities that start before glucose changes are detectable. Insulin resistance may be involved in the association between SCH and microalbuminuria. Our study revealed that HOMA-IR was significantly associated with TSH levels and UACR. Moreover, thyroid hormone plays an important role in the development and physiology of the kidney [39]. It has been suggested that the mechanism of SCH associated-kidney dysfunction is related to SCH altering renal hemodynamic by decreasing cardiac output and increasing peripheral vascular resistance [39,40]. Interestingly, recent studies have shown that patients with SCH have endothelial dysfunction characterized by reduced endothelium-dependent vasodilation and impaired nitric oxide availability. Furthermore, this phenomenon is partially in dependent of dyslipidemia and improved by levothyroxine treatment [41]. Endothelial dysfunction is clearly associated with the pathogenesis of not only atherosclerosis but also diabetic nephropathy [42]. Therefore, SCH may have an important role in the development of diabetic nephropathy via vascular endothelial dysfunction.
Our study has some limitations. First, thyroid function was evaluated at a single time point. Follow-up thyroid function tests are needed to confirm the association between the clinical course of SCH and microalbuminura. Second, the diagnosis of microalbuminura was based on one point measurement. Establishing the diagnosis of microalbuminuria requires at least two of three specimens fall within the microalbuminuric range over a 3- to 6-month period. Third, unconditional logistic regression analysis was performed to evaluate the association of SCH with microalbuminuria. However, the strength of this study is its case control nature which can establish the causal association between SCH and microalbuminura in prediabetic state.
In conclusion, our findings suggest that prediabetic subjects with SCH demonstrate higher prevalence of microalbuminuria than their non-SCH counterparts. SCH is also independently associated with microalbuminuria in prediabetic subjects. Screening and treatment for SCH may be warranted in those patients.
ACKNOWLEDGMENTS
The authors thank all sample donors for their contribution to this study and all members of the Endocrinology Unit, Specialized Medical Hospital, Mansoura, Egypt.
Notes
No potential conflict of interest relevant to this article was reported.