- Current
- Browse
- Collections
-
For contributors
- For Authors
- Instructions to authors
- Article processing charge
- e-submission
- For Reviewers
- Instructions for reviewers
- How to become a reviewer
- Best reviewers
- For Readers
- Readership
- Subscription
- Permission guidelines
- About
- Editorial policy
Articles
- Page Path
- HOME > Diabetes Metab J > Volume 37(6); 2013 > Article
-
Original ArticlePathophysiology Subclinical Hypothyroidism Is Independently Associated with Microalbuminuria in a Cohort of Prediabetic Egyptian Adults
- Mervat M. El-Eshmawy1, Hala A. Abd El-Hafez1, Walaa Othman El Shabrawy2, Ibrahim A. Abdel Aal2
-
Diabetes & Metabolism Journal 2013;37(6):450-457.
DOI: https://doi.org/10.4093/dmj.2013.37.6.450
Published online: December 12, 2013
1Department of Internal Medicine, Specialized Medical Hospital, Mansoura University Faculty of Medicine, Mansoura, Egypt.
2Department of Clinical Pathology, Specialized Medical Hospital, Mansoura University Faculty of Medicine, Mansoura, Egypt.
- Corresponding author: Mervat M. El-Eshmawy. Department of Internal Medicine, Specialized Medical Hospital, Mansoura University Faculty of Medicine, Elgomhoria St, Mansoura 35516, Egypt. mervat2040@yahoo.com
Copyright © 2013 Korean Diabetes Association
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
ABSTRACT
-
Background
- Recent evidence has suggested an association between subclinical hypothyroidism (SCH) and microalbuminuria in patients with type 2 diabetes. However, whether SCH is related to microalbuminuria among subjects with prediabetes has not been studied. Thus, we evaluated the association between SCH and microalbuminuria in a cohort of prediabetic Egyptian adults.
-
Methods
- A total of 147 prediabetic subjects and 150 healthy controls matched for age and sex were enrolled in this study. Anthropometric measurements, plasma glucose, lipid profile, homeostasis model assessment of insulin resistance (HOMA-IR), thyroid stimulating hormone (TSH), free thyroxine, triiodothyronine levels, and urinary albumin-creatinine ratio (UACR) were assessed.
-
Results
- The prevalence of SCH and microalbuminuria in the prediabetic subjects was higher than that in the healthy controls (16.3% vs. 4%, P<0.001; and 12.9% vs. 5.3%, P=0.02, respectively). Prediabetic subjects with SCH were characterized by significantly higher HOMA-IR, TSH levels, UACR, and prevalence of microalbuminuria than those with euthyroidism. TSH level was associated with total cholesterol (P=0.05), fasting insulin (P=0.01), HOMA-IR (P=0.01), and UACR (P=0.005). UACR was associated with waist circumference (P=0.01), fasting insulin (P=0.05), and HOMA-IR (P=0.02). With multiple logistic regression analysis, SCH was associated with microalbuminuria independent of confounding variables (β=2.59; P=0.01).
-
Conclusion
- Our findings suggest that prediabetic subjects with SCH demonstrate higher prevalence of microalbuminuria than their non-SCH counterparts. SCH is also independently associated with microalbuminuria in prediabetic subjects. Screening and treatment for SCH may be warranted in those patients.
- Prediabetes is a high-risk state for diabetes that is typically defined as blood glucose concentrations higher than normal, but lower than diabetes thresholds. According to World Health Organization (WHO), people are at high risk of developing diabetes if they have one of two distinct states: impaired fasting glucose (IFG) defined as a fasting plasma glucose (FPG) concentration of ≥6.1 and <7.0 mmol/L, without impaired glucose tolerance (IGT), and IGT defined as a FPG concentration of <7.0 mmol/L and a 2-hour postload plasma glucose concentration of ≥7.8 and <11.1 mmol/L, measured during a 75 g oral glucose tolerance test [1]. Prevalence of prediabetes is increasing worldwide and experts have projected that more than 470 million people will have prediabetes by 2030 [2]. Around 5% to 10% of people with prediabetes become diabetic every year, with the same proportion converting back to normoglycaemia, although the conversion rate varies with population characteristics and prediabetes definitions [3,4].
- The presence of microalbuminuria has become an important tool for therapeutic intervention [5]. Microalbuminuria is an early sign of an increased risk for developing kidney disease in patients with diabetes [6]. Prediabetes has been linked to increased risk of early forms of nephropathy and chronic kidney disease, defined by methods such as urinary albumin excretion rate and estimated glomerular filtration rate [7,8].
- Subclinical hypothyroidism (SCH) is a common endocrine disorder and has been reported to range from 4% to 10% in large general population screening surveys [9] and has been found to be 4% to 17% in diabetic patients in previous studies [10-12]. Patients with SCH are usually asymptomatic, but may have cardiac dysfunction, elevated low density lipoprotein and neuropsychiatric symptoms [13]. SCH characterized by elevated thyroid stimulating hormone (TSH) levels with normal free thyroxine (FT4) and triiodothyronine (FT3) concentrations [14].
- Both SCH and albuminuria are independent risk factors of cardiovascular disease [15,16]. Recent evidence has suggested an independent association between SCH and microalbuminuria in patients with type 2 diabetes [17]. However, whether SCH is related to microalbuminuria among subjects with prediabetes has not been studied. In this study, we evaluated the possible association between SCH and microalbuminuria among a cohort of Egyptian adults with prediabetes.
INTRODUCTION
- The study comprised 147 adults with prediabetes aged 45.6±5.2 years (63 men and 84 women) and 150 age and sex matched healthy controls. Prediabetic subjects were recruited from Endocrinology and Obesity outpatient clinics at Specialized Medical Hospital, Mansoura University Faculty of Medicine, Egypt, during the period from May 2012 to January 2013. Healthy controls were recruited from the same geographic area; Mansoura city; and with the same exclusion criteria for prediabetic subjects. All subjects signed an informed consent to be included in our study. The study was approved by the local ethical committee.
- All participants were subjected to thorough medical history and clinical examination. Anthropometric measurements including height, weight, body mass index (BMI) and waist circumference (WC) were obtained using standardized techniques; height was measured to the nearest 0.5 cm, body weight was measured to the nearest 0.1 kg, BMI was calculated as weight in kilograms divided by height in meters squared (kg/m2), WC was measured at the highest point of the iliac crest. Blood pressure was taken in the sitting position after 10 minutes rest using a random-zero sphygmomanometer. Adults diagnosed with prediabetes based on WHO criteria [1]. SCH was diagnosed by elevated TSH (>4 to 10 µU/mL) levels with normal FT4 and FT3 levels [14]. Urinary albumin-creatinine ratio (UACR) was determined using 24-hour urine samples and defined as milligram of albumin per gram of creatinine (mg/g). Microalbuminuria was defined by a UACR of 30 to 300 mg/g.
- Exclusion criteria were acute or chronic renal failure, glomerulonephritis, overt proteinuria, hypertension or the use of antihypertensive medications, thyroid disorders other than SCH, overt hypothyroidism, hepatic dysfunction, infection, connective tissue disorders, malignancy, pregnancy, women on birth control pills or hormone replacement therapy, and smoking. We also excluded patients taking drugs that could influence thyroid function or proteinuria, e.g., angiotensin-converting enzyme inhibitors and angiotensin II receptor blockers, thiazide diuretics, β-blockers, amiodarone, or lithium.
- Laboratory assay
- FPG and 2-hour postload plasma glucose were estimated using commercially available kit, Cobas (Integra-400) supplied by Roche Diagnostic (Basel, Germany). Fasting serum insulin was assayed by a solid-phase, enzyme-labeled chemiluminescent immunometric assay using immulite analyzer supplied by Siemens (DPC; Cirrus Inc., Los Angeles, CA, USA). Homeostasis model assessment of insulin resistance (HOMA-IR) was calculated with the formula: HOMA-IR=fasting insulin (µU/mL)×fasting glucose (mmol/L)/22.5 [18]. Hemoglobin A1c (HbA1c) was measured as an index of metabolic control on a DCA 2000 analyzer, fast ion exchange resin (Roche Diagnostic). The reference range was 4.4% to 6.4%. Total cholesterol (TC), triglyceride (TG), and high density lipoprotein cholesterol (HDL-C) were assayed by commercially available kits, Cobas (Integra-400) supplied by Roche Diagnostic. Low density lipoprotein cholesterol (LDL-C) was calculated according to Friedewald et al. [19]. FT4, FT3, and TSH were measured by electrochemiluminecent immunoassay, using Elecsys 2010 (Roche Diagnostic). UACR was measured by an immunonephelometric method using Microalbuminuria (AIBT2) kit, Cobas (Integra-400) supplied by Roche Diagnostic.
- Statistical methods
- Data entry and analysis were performed using SPSS statistical package version 17 (SPSS Inc., Chicago, IL, USA). The data were expressed as mean±standard deviation for continuous data, frequency and proportion for categorical data, and median (minimum to maximum) for skewed data. Student t-test and Mann-Whitney were used to compare two groups. A chi-square test was used to compare categorical data. Pearson and Spearman correlations were done to study relation between different items. Multiple logistic regression analysis was performed to evaluate the association of SCH with microalbuminuria. Microalbuminuria, WC, fasting insulin, and HOMA-IR were entered in the regression model. P≤0.05 was considered as significant.
METHODS
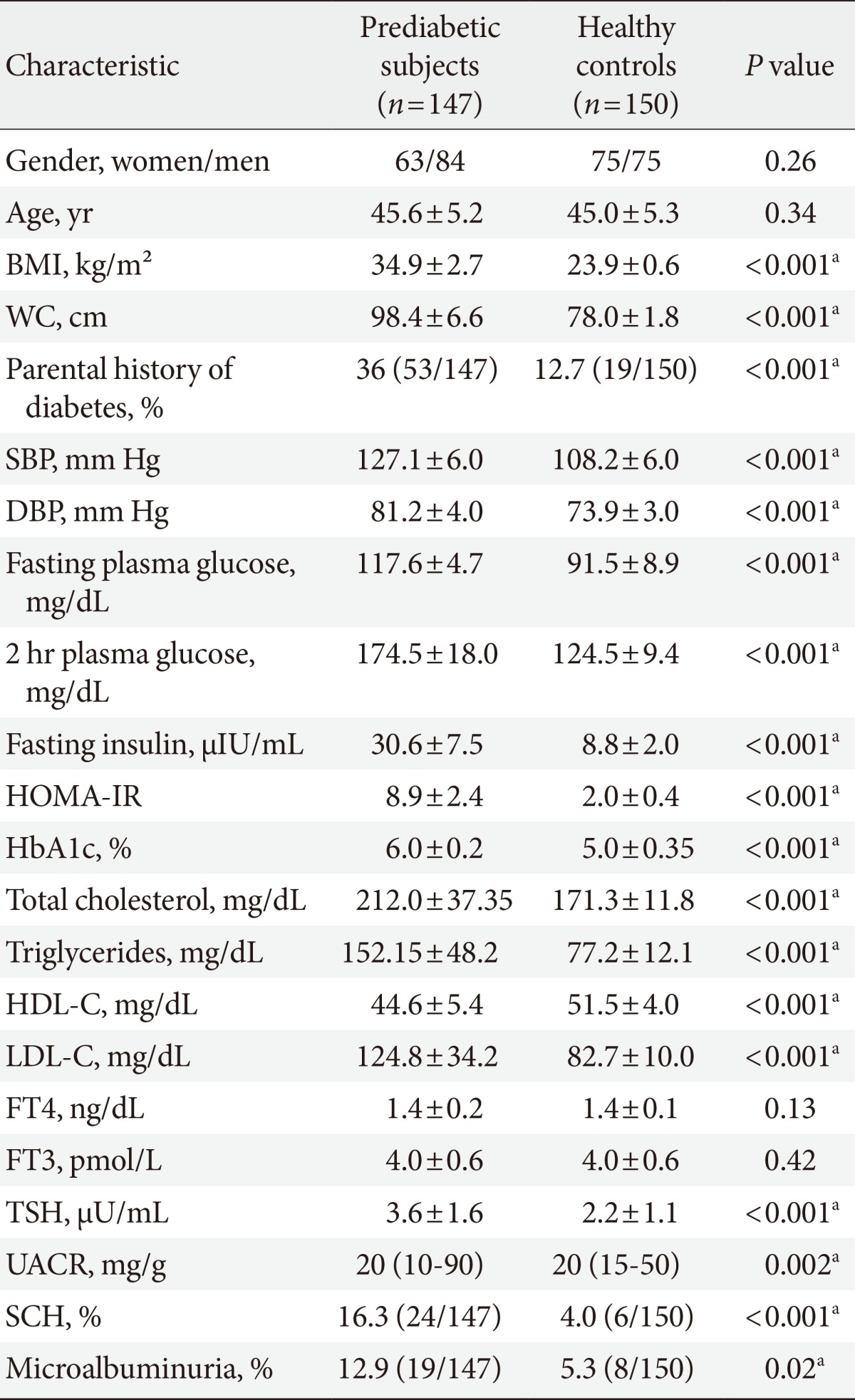
- The baseline characteristics of the 147 prediabetic subjects and healthy controls were described in Table 1. Prediabetic subjects had significantly higher BMI, WC, blood pressure, parental history of diabetes, fasting and 2-hour plasma glucose, HbA1c, fasting insulin and HOMA-IR, TC, TG and LDL-C than healthy controls, however, HDL-C was significantly lower in prediabetic subjects than in healthy controls. TSH and UACR were also significantly higher in prediabetic subjects than in healthy controls (3.59±1.59 vs. 2.23±1.1, P<0.001; and 20 [10 to 90] vs. 20 [15 to 50], P=0.002, respectively). The prevalence of SCH in prediabetic subjects was 16.3% (24/147) compared to 4% (6/150) in healthy controls, P<0.001. SCH was prevalent in women than in men in prediabetic subjects (66.7% [16/24] vs. 33.3% [8/24]; P=0.01). The prevalence of microalbuminuria in prediabetic subjects was 12.9% (19/147) compared to 5.3% (8/150) in healthy controls, P=0.02.
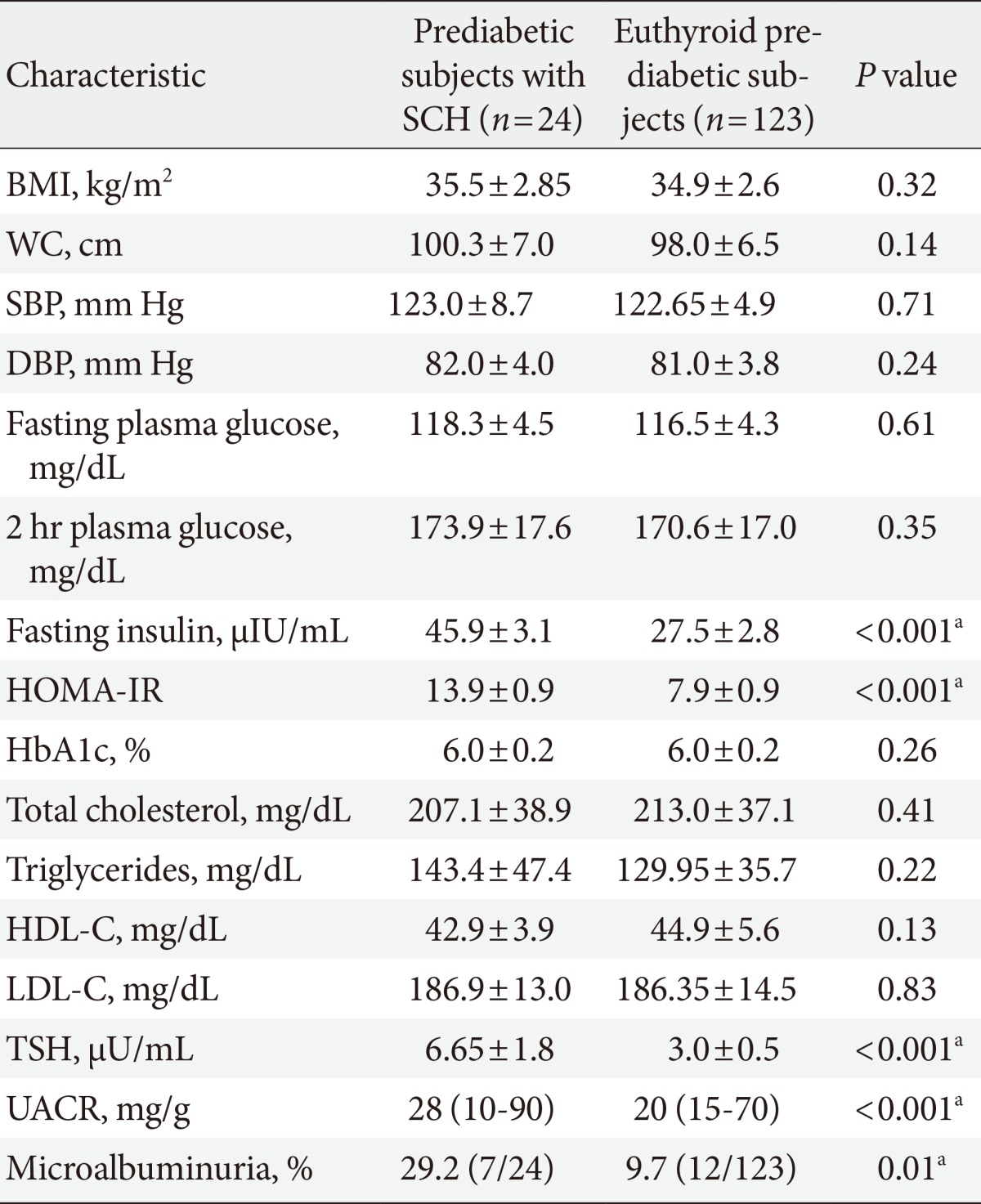
- Prediabetes participants were divided into two groups according to thyroid function: patients with SCH or those with euthyroidism. Prediabetic subjects with SCH were characterized by significantly higher fasting insulin, HOMA-IR, TSH levels (6.65±1.76 mU/mL vs. 2.99±0.48 mU/mL; P<0.001), UACR (28 [10 to 90] vs. 20 [15 to 70]; P<0.001) and prevalence of microalbuminuria (29.2% vs. 9.7%; P=0.01) than those with euthyroidism, whereas BMI, WC, blood pressure, fasting, 2-hour plasma glucose, HbA1c, and lipid profile were not different between the two groups (Table 2).
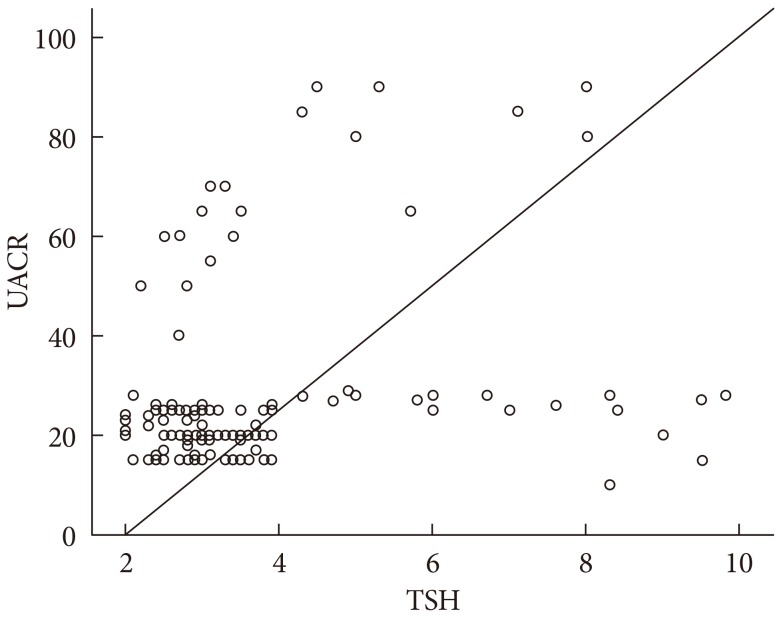
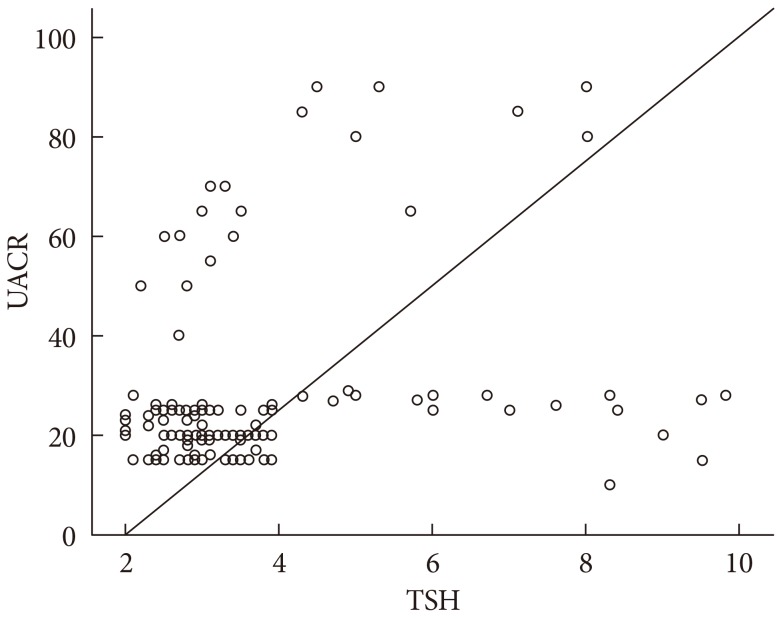
- TSH level was significantly associated with fasting insulin (r=0.18, P=0.01), HOMA-IR (r=0.21, P=0.01), and TC (r=0.16, P=0.05). On the other hand, UACR was significantly associated with WC (r=0.21, P=0.01), fasting insulin (r=0.16, P=0.05), and HOMA-IR (r=0.19, P=0.02) (Table 3). TSH was significantly associated with UACR (r=0.23, P=0.005) (Fig. 1); furthermore, multiple logistic regression analysis demonstrated that SCH was associated with microalbuminuria independent of WC, fasting insulin, and HOMA-IR (β=2.59; odds ratio, 4.62; 95% confidence interval, 1.45 to 14.73; P=0.01) (Table 4). There was insignificant association between UACR and TSH in healthy controls (r=0.07, P=0.4).
RESULTS
- The current study examined, for the first time, the relationship between SCH and microalbuminuria in prediabetic subjects. The main finding was the association between TSH level and UACR; furthermore, SCH was an independent risk factor for microalbuminuria.
- The prediabetic subjects in our study had higher BMI, WC, parental history of diabetes, blood pressure, fasting insulin, HOMA-IR, and lipids than healthy controls. In support, American Diabetes Association [20] reported that overweight/obesity and family history of diabetes are risk factors for prediabetes. On the other hand, blood glucose in the prediabetic range is correlated with many risk factors, including general and central obesity, blood pressure, and TG and lipoprotein concentrations [21].
- In the present study, prediabetic subjects had higher prevalence of microalbuminuria than healthy controls (12.9% vs. 5.3%; P=0.02). This is in agreement with Hwang et al. [22] who found that the prevalence of microalbuminuria among 947 prediabetic subjects was 12.8%. National Health and Nutrition Examination Survey, 1999 to 2006, showed that the prevalence of microalbuminuria increases as glycaemia worsens, i.e., from 6% in normoglycaemia, to 10% in IFG and 29% in undiagnosed diabetes or diagnosed diabetes [8].
- Our results also showed positive correlations between UACR and WC, insulin, and HOMA-IR. In accordance, Chandie Shaw et al. [23] demonstrated that central obesity is an independent risk factor of albuminuria in healthy individuals. In addition, it has been suggested that microalbuminuria is associated with insulin resistance [24,25] and may precede the development of type 2 diabetes [26].
- Haffner et al. [27] previously demonstrated that microalbuminuria may be a feature of the prediabetic state. Moreover, longitudinal studies suggest that prediabetes is a risk factor for chronic kidney disease, but whether this association is attributable to the effects of prediabetes itself, increased incidence of diabetes, or common causes contributing to both hyperglycaemia and kidney pathology is unclear [28,29]. From a mechanistic perspective, subtle damage occurs within the kidney in the prediabetic state whether from chronic exposure to abnormal glycemia that is below the formal threshold for diabetes, elevated intrarenal blood pressure, or oxidative stress [30]. Alternatively, increased UACR could be an early organ-specific manifestation of insulin resistance that heralds the onset of diabetes [31].
- The association between type 2 diabetes and SCH is well recognized with reported prevalence of 4% to 17% [12], however, little is known about the prevalence of SCH in those with prediabetes. In the current study, the prevalence of SCH among prediabetics was 16.3% compared to 4% in healthy controls. We confirmed that SCH was prevalent in women than in men; this is in parallel with previous research studies which demonstrated high prevalence of SCH in type 2 diabetic women [32,33].
- We observed significant association between serum TSH level and TC, fasting insulin, and HOMA-IR. These results parallel those of Chubb et al. [34], who found that small variations in TSH might exert a marked effect on lipid levels in diabetic adults. Furthermore, fasting hyperinsulinemia has been previously reported in patients with SCH [35]. Moreover, Maratou et al. [36] confirmed that patients with SCH have insulin resistance that is comparable with that of the patients with hypothyroidism. In patients with SCH, impairment in insulin-stimulated glucose disposal is caused by impaired translocation of GLUT4 transporters on the cell surface [36].
- In the current study, prediabetic subjects with SCH demonstrated higher UACR and prevalence of microalbuminuria than their non-SCH counterparts. In addition, multiple logistic regression analysis revealed that SCH was independently associated with microalbuminuria in prediabetic subjects. In line, Chen et al. [37] demonstrated that type 2 diabetic patients with SCH are associated with an increased risk of nephropathy after adjustment for other factors. On the other hand, proteinuria had higher TSH levels [38].
- Prediabetes is associated with the simultaneous presence of insulin resistance and β-cell dysfunction; abnormalities that start before glucose changes are detectable. Insulin resistance may be involved in the association between SCH and microalbuminuria. Our study revealed that HOMA-IR was significantly associated with TSH levels and UACR. Moreover, thyroid hormone plays an important role in the development and physiology of the kidney [39]. It has been suggested that the mechanism of SCH associated-kidney dysfunction is related to SCH altering renal hemodynamic by decreasing cardiac output and increasing peripheral vascular resistance [39,40]. Interestingly, recent studies have shown that patients with SCH have endothelial dysfunction characterized by reduced endothelium-dependent vasodilation and impaired nitric oxide availability. Furthermore, this phenomenon is partially in dependent of dyslipidemia and improved by levothyroxine treatment [41]. Endothelial dysfunction is clearly associated with the pathogenesis of not only atherosclerosis but also diabetic nephropathy [42]. Therefore, SCH may have an important role in the development of diabetic nephropathy via vascular endothelial dysfunction.
- Our study has some limitations. First, thyroid function was evaluated at a single time point. Follow-up thyroid function tests are needed to confirm the association between the clinical course of SCH and microalbuminura. Second, the diagnosis of microalbuminura was based on one point measurement. Establishing the diagnosis of microalbuminuria requires at least two of three specimens fall within the microalbuminuric range over a 3- to 6-month period. Third, unconditional logistic regression analysis was performed to evaluate the association of SCH with microalbuminuria. However, the strength of this study is its case control nature which can establish the causal association between SCH and microalbuminura in prediabetic state.
- In conclusion, our findings suggest that prediabetic subjects with SCH demonstrate higher prevalence of microalbuminuria than their non-SCH counterparts. SCH is also independently associated with microalbuminuria in prediabetic subjects. Screening and treatment for SCH may be warranted in those patients.
DISCUSSION
-
Acknowledgements
- The authors thank all sample donors for their contribution to this study and all members of the Endocrinology Unit, Specialized Medical Hospital, Mansoura, Egypt.
ACKNOWLEDGMENTS
- 1. World Health Organization. International Diabetes Foundation. Definition and diagnosis of diabetes mellitus and intermediate hyperglycaemia: report of a WHO/IDF consultation. Geneva: World Health Organization; 2006.
- 2. Tabak AG, Herder C, Rathmann W, Brunner EJ, Kivimaki M. Prediabetes: a high-risk state for diabetes development. Lancet 2012;379:2279-2290. ArticlePubMedPMC
- 3. Forouhi NG, Luan J, Hennings S, Wareham NJ. Incidence of type 2 diabetes in England and its association with baseline impaired fasting glucose: the Ely study 1990-2000. Diabet Med 2007;24:200-207. ArticlePubMed
- 4. Nathan DM, Davidson MB, DeFronzo RA, Heine RJ, Henry RR, Pratley R, Zinman B. American Diabetes Association. Impaired fasting glucose and impaired glucose tolerance: implications for care. Diabetes Care 2007;30:753-759. PubMed
- 5. Agewall S, Wikstrand J, Ljungman S, Fagerberg B. Risk Factor Intervention Study Group. Usefulness of microalbuminuria in predicting cardiovascular mortality in treated hypertensive men with and without diabetes mellitus. Am J Cardiol 1997;80:164-169. ArticlePubMed
- 6. Alzaid AA. Microalbuminuria in patients with NIDDM: an overview. Diabetes Care 1996;19:79-89. ArticlePubMedPDF
- 7. Gabir MM, Hanson RL, Dabelea D, Imperatore G, Roumain J, Bennett PH, Knowler WC. Plasma glucose and prediction of microvascular disease and mortality: evaluation of 1997 American Diabetes Association and 1999 World Health Organization criteria for diagnosis of diabetes. Diabetes Care 2000;23:1113-1118. ArticlePubMedPDF
- 8. Plantinga LC, Crews DC, Coresh J, Miller ER 3rd, Saran R, Yee J, Hedgeman E, Pavkov M, Eberhardt MS, Williams DE, Powe NR. CDC CKD Surveillance Team. Prevalence of chronic kidney disease in US adults with undiagnosed diabetes or prediabetes. Clin J Am Soc Nephrol 2010;5:673-682. ArticlePubMedPMC
- 9. McDermott MT, Ridgway EC. Subclinical hypothyroidism is mild thyroid failure and should be treated. J Clin Endocrinol Metab 2001;86:4585-4590. ArticlePubMed
- 10. Perros P, McCrimmon RJ, Shaw G, Frier BM. Frequency of thyroid dysfunction in diabetic patients: value of annual screening. Diabet Med 1995;12:622-627. ArticlePubMed
- 11. Smithson MJ. Screening for thyroid dysfunction in a community population of diabetic patients. Diabet Med 1998;15:148-150. ArticlePubMed
- 12. Chubb SA, Davis WA, Inman Z, Davis TM. Prevalence and progression of subclinical hypothyroidism in women with type 2 diabetes: the Fremantle Diabetes Study. Clin Endocrinol (Oxf) 2005;62:480-486. ArticlePubMed
- 13. Jones DD, May KE, Geraci SA. Subclinical thyroid disease. Am J Med 2010;123:502-504. ArticlePubMed
- 14. Surks MI, Ortiz E, Daniels GH, Sawin CT, Col NF, Cobin RH, Franklyn JA, Hershman JM, Burman KD, Denke MA, Gorman C, Cooper RS, Weissman NJ. Subclinical thyroid disease: scientific review and guidelines for diagnosis and management. JAMA 2004;291:228-238. ArticlePubMed
- 15. Rodondi N, den Elzen WP, Bauer DC, Cappola AR, Razvi S, Walsh JP, Asvold BO, Iervasi G, Imaizumi M, Collet TH, Bremner A, Maisonneuve P, Sgarbi JA, Khaw KT, Vanderpump MP, Newman AB, Cornuz J, Franklyn JA, Westendorp RG, Vittinghoff E, Gussekloo J. Thyroid Studies Collaboration. Subclinical hypothyroidism and the risk of coronary heart disease and mortality. JAMA 2010;304:1365-1374. ArticlePubMedPMC
- 16. Gerstein HC, Mann JF, Yi Q, Zinman B, Dinneen SF, Hoogwerf B, Halle JP, Young J, Rashkow A, Joyce C, Nawaz S, Yusuf S. HOPE Study Investigators. Albuminuria and risk of cardiovascular events, death, and heart failure in diabetic and nondiabetic individuals. JAMA 2001;286:421-426. ArticlePubMed
- 17. Yasuda T, Kaneto H, Kuroda A, Yamamoto T, Takahara M, Naka T, Miyashita K, Fujisawa K, Sakamoto F, Katakami N, Matsuoka TA, Shimomura I. Subclinical hypothyroidism is independently associated with albuminuria in people with type 2 diabetes. Diabetes Res Clin Pract 2011;94:e75-e77. ArticlePubMed
- 18. Matthews DR, Hosker JP, Rudenski AS, Naylor BA, Treacher DF, Turner RC. Homeostasis model assessment: insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia 1985;28:412-419. ArticlePubMedPDF
- 19. Friedewald WT, Levy RI, Fredrickson DS. Estimation of the concentration of low-density lipoprotein cholesterol in plasma, without use of the preparative ultracentrifuge. Clin Chem 1972;18:499-502. ArticlePubMedPDF
- 20. American Diabetes Association. Standards of medical care in diabetes: 2013. Diabetes Care 2013;36(Suppl 1):S11-S66. ArticlePubMedPDF
- 21. Emerging Risk Factors Collaboration. Sarwar N, Gao P, Seshasai SR, Gobin R, Kaptoge S, Di Angelantonio E, Ingelsson E, Lawlor DA, Selvin E, Stampfer M, Stehouwer CD, Lewington S, Pennells L, Thompson A, Sattar N, White IR, Ray KK, Danesh J. Diabetes mellitus, fasting blood glucose concentration, and risk of vascular disease: a collaborative meta-analysis of 102 prospective studies. Lancet 2010;375:2215-2222. ArticlePubMedPMC
- 22. Hwang ST, Cho YK, Yun JW, Park JH, Kim HJ, Park DI, Sohn CI, Jeon WK, Kim BI, Rhee EJ, Oh KW, Lee WY, Jin W. Impact of non-alcoholic fatty liver disease on microalbuminuria in patients with prediabetes and diabetes. Intern Med J 2010;40:437-442. ArticlePubMed
- 23. Chandie Shaw PK, Berger SP, Mallat M, Frolich M, Dekker FW, Rabelink TJ. Central obesity is an independent risk factor for albuminuria in nondiabetic South Asian subjects. Diabetes Care 2007;30:1840-1844. ArticlePubMedPDF
- 24. Nosadini R, Solini A, Velussi M, Muollo B, Frigato F, Sambataro M, Cipollina MR, De Riva F, Brocco E, Crepaldi G. Impaired insulin-induced glucose uptake by extrahepatic tissue is hallmark of NIDDM patients who have or will develop hypertension and microalbuminuria. Diabetes 1994;43:491-499. ArticlePubMed
- 25. Nosadini R, Cipollina MR, Solini A, Sambataro M, Morocutti A, Doria A, Fioretto P, Brocco E, Muollo B, Frigato F. Close relationship between microalbuminuria and insulin resistance in essential hypertension and non-insulin dependent diabetes mellitus. J Am Soc Nephrol 1992;3(4 Suppl):S56-S63. ArticlePubMed
- 26. Mykkanen L, Haffner SM, Kuusisto J, Pyorala K, Laakso M. Microalbuminuria precedes the development of NIDDM. Diabetes 1994;43:552-557. ArticlePubMed
- 27. Haffner SM, Gonzales C, Valdez RA, Mykkanen L, Hazuda HP, Mitchell BD, Monterrosa A, Stern MP. Is microalbuminuria part of the prediabetic state? The Mexico City Diabetes Study. Diabetologia 1993;36:1002-1006. ArticlePubMedPDF
- 28. Fox CS, Larson MG, Leip EP, Meigs JB, Wilson PW, Levy D. Glycemic status and development of kidney disease: the Framingham Heart Study. Diabetes Care 2005;28:2436-2440. PubMed
- 29. Thomas G, Sehgal AR, Kashyap SR, Srinivas TR, Kirwan JP, Navaneethan SD. Metabolic syndrome and kidney disease: a systematic review and meta-analysis. Clin J Am Soc Nephrol 2011;6:2364-2373. PubMedPMC
- 30. Duncan ER, Walker SJ, Ezzat VA, Wheatcroft SB, Li JM, Shah AM, Kearney MT. Accelerated endothelial dysfunction in mild prediabetic insulin resistance: the early role of reactive oxygen species. Am J Physiol Endocrinol Metab 2007;293:E1311-E1319. ArticlePubMed
- 31. Friedman AN, Marrero D, Ma Y, Ackermann R, Narayan KM, Barrett-Connor E, Watson K, Knowler WC, Horton ES. Diabetes Prevention Program Research Group. Value of urinary albumin-to-creatinine ratio as a predictor of type 2 diabetes in pre-diabetic individuals. Diabetes Care 2008;31:2344-2348. ArticlePubMedPMCPDF
- 32. Canaris GJ, Manowitz NR, Mayor G, Ridgway EC. The Colorado thyroid disease prevalence study. Arch Intern Med 2000;160:526-534. ArticlePubMed
- 33. Hollowell JG, Staehling NW, Flanders WD, Hannon WH, Gunter EW, Spencer CA, Braverman LE. Serum TSH, T(4), and thyroid antibodies in the United States population (1988 to 1994): National Health and Nutrition Examination Survey (NHANES III). J Clin Endocrinol Metab 2002;87:489-499. ArticlePubMed
- 34. Chubb SA, Davis WA, Davis TM. Interactions among thyroid function, insulin sensitivity, and serum lipid concentrations: the Fremantle diabetes study. J Clin Endocrinol Metab 2005;90:5317-5320. ArticlePubMed
- 35. Tuzcu A, Bahceci M, Gokalp D, Tuzun Y, Gunes K. Subclinical hypothyroidism may be associated with elevated high-sensitive c-reactive protein (low grade inflammation) and fasting hyperinsulinemia. Endocr J 2005;52:89-94. ArticlePubMed
- 36. Maratou E, Hadjidakis DJ, Kollias A, Tsegka K, Peppa M, Alevizaki M, Mitrou P, Lambadiari V, Boutati E, Nikzas D, Tountas N, Economopoulos T, Raptis SA, Dimitriadis G. Studies of insulin resistance in patients with clinical and subclinical hypothyroidism. Eur J Endocrinol 2009;160:785-790. ArticlePubMed
- 37. Chen HS, Wu TE, Jap TS, Lu RA, Wang ML, Chen RL, Lin HD. Subclinical hypothyroidism is a risk factor for nephropathy and cardiovascular diseases in type 2 diabetic patients. Diabet Med 2007;24:1336-1344. ArticlePubMed
- 38. Gilles R, den Heijer M, Ross AH, Sweep FC, Hermus AR, Wetzels JF. Thyroid function in patients with proteinuria. Neth J Med 2008;66:483-485. PubMed
- 39. Iglesias P, Diez JJ. Thyroid dysfunction and kidney disease. Eur J Endocrinol 2009;160:503-515. ArticlePubMed
- 40. Vargas F, Moreno JM, Rodriguez-Gomez I, Wangensteen R, Osuna A, Alvarez-Guerra M, Garcia-Estan J. Vascular and renal function in experimental thyroid disorders. Eur J Endocrinol 2006;154:197-212. ArticlePubMed
- 41. Biondi B, Galderisi M, Pagano L, Sidiropulos M, Pulcrano M, A DE, Ippolito S, Rossi A, de Divitiis O, Lombardi G. Endothelial-mediated coronary flow reserve in patients with mild thyroid hormone deficiency. Eur J Endocrinol 2009;161:323-329. ArticlePubMed
- 42. Schalkwijk CG, Stehouwer CD. Vascular complications in diabetes mellitus: the role of endothelial dysfunction. Clin Sci (Lond) 2005;109:143-159. ArticlePubMedPDF
REFERENCES
Values are presented as mean±standard deviation, number (%), or median (minimum-maximum).
BMI, body mass index; WC, waist circumference; SBP, systolic blood pressure; DBP, diastolic blood pressure; HOMA-IR, homeostasis model assessment of insulin resistance; HbA1c, hemoglobin A1c; HDL-C, high density lipoprotein cholesterol; LDL-C, low density lipoprotein cholesterol; FT4, free thyroxine; FT3, triiodothyronine; TSH, thyroid stimulating hormone; UACR, urinary albumin-creatinine ratio; SCH, subclinical hypothyroidism.
aP is significant if ≤0.05.
Values are presented as mean±standard deviation, number (%), or median (minimum-maximum).
SCH, subclinical hypothyroidism; BMI, body mass index; WC, waist circumference; SBP, systolic blood pressure; DBP, diastolic blood pressure; HOMA-IR, homeostasis model assessment of insulin resistance; HbA1c, hemoglobin A1c; HDL-C, high density lipoprotein cholesterol; LDL-C, low density lipoprotein cholesterol; TSH, thyroid stimulating hormone; UACR, urinary albumin-creatinine ratio.
aP is significant if ≤0.05.
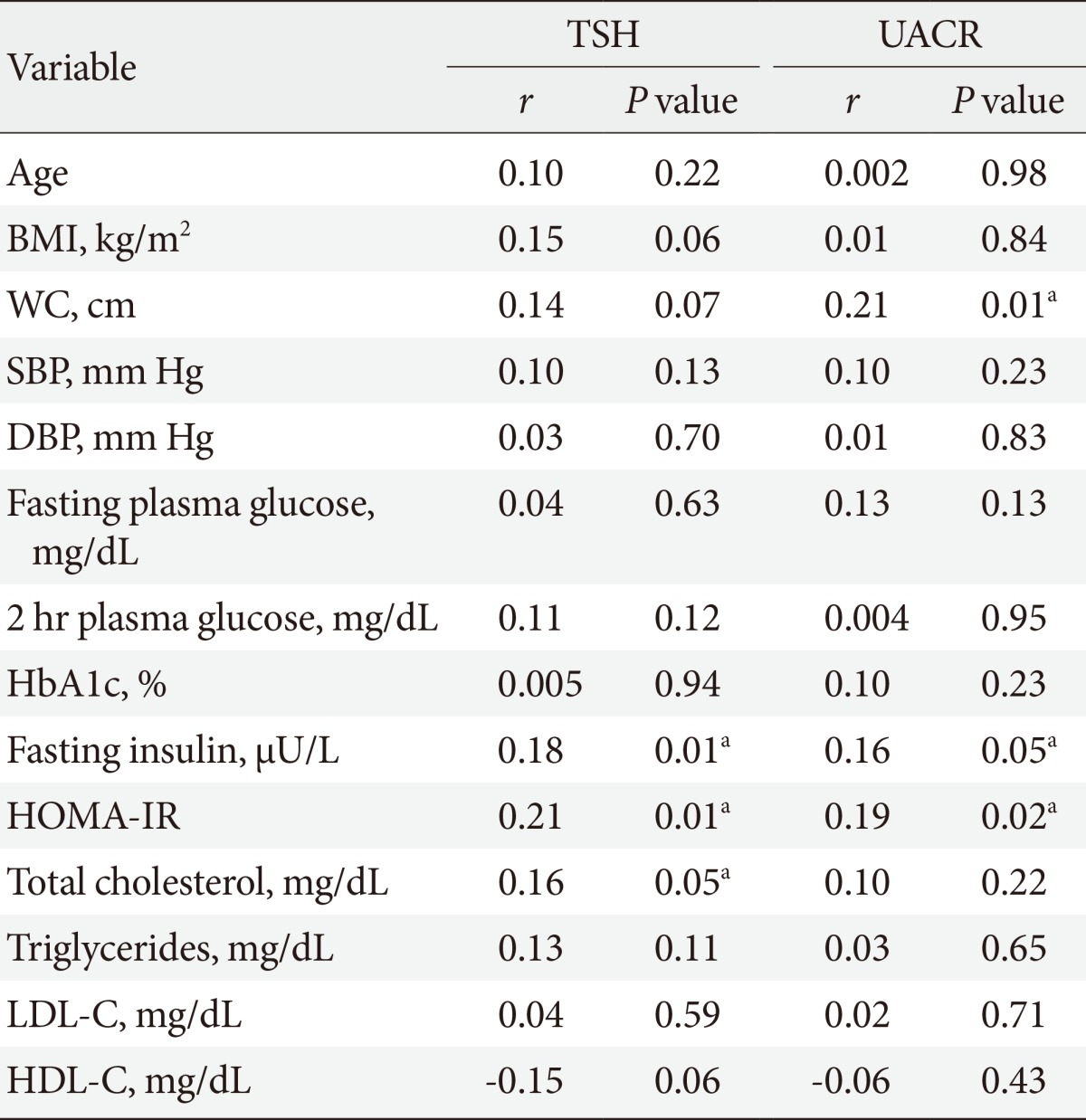
TSH, thyroid stimulating hormone; UACR, urinary albumin-creatinine ratio; BMI, body mass index; WC, waist circumference; SBP, systolic blood pressure; DBP, diastolic blood pressure; HbA1c, hemoglobin A1c; HOMA-IR, homeostasis model assessment of insulin resistance; LDL-C, low density lipoprotein cholesterol; HDL-C, high density lipoprotein cholesterol.
aP is significant if ≤0.05.
Figure & Data
References
Citations

- The prevalence of subclinical hypothyroidism in a pre-diabetes population and an analysis of related factors
Xingyu Chang, Yaqi Wang, Yi Liu, Yanyu Shen, Jiaqing Feng, Qianqian Liu, Chenjun Jiang, Jing Yu, Xulei Tang, Gaojing Jing, Qianglong Niu, Songbo Fu
Annals of Medicine.2023; 55(1): 643. CrossRef - High Thyroid-Stimulating Hormone and Low Free Triiodothyronine Levels Are Associated with Chronic Kidney Disease in Three Population-Based Studies from Germany
Till Ittermann, Sabrina von Rheinbaben, Marcello R. P. Markus, Marcus Dörr, Antje Steveling, Matthias Nauck, Alexander Teumer, Maik Gollasch, Dominik Spira, Maximilian König, Ilja Demuth, Elisabeth Steinhagen-Thiessen, Henry Völzke, Sylvia Stracke
Journal of Clinical Medicine.2023; 12(17): 5763. CrossRef - Free triiodothyronine predicts the risk of developing diabetic kidney disease
Weihong Li, Zhi Yang, Shengjian Li, Shanshan Jiang, Wan Hu, Zhenying Wan, Ping Tu, Peng Duan
BMC Nephrology.2023;[Epub] CrossRef - The Correlation between Thyroid Hormone Levels and the Kidney Disease Progression Risk in Patients with Type 2 Diabetes
Zhi Yang, Peng Duan, Weihong Li, Ronghui Nie, Xiaoyang Lou, Lina Wang, Kexia Wu, Jiang Liu, Ping Tu, Xiaoyang Lai
Diabetes, Metabolic Syndrome and Obesity: Targets and Therapy.2022; Volume 15: 59. CrossRef - Thyroid hormone status in patients with impaired kidney function
Juliane Peters, Stefanos Roumeliotis, Peter R. Mertens, Vassilios Liakopoulos
International Urology and Nephrology.2021; 53(11): 2349. CrossRef - Subclinical hypothyroidism is associated with albuminuria in Japanese nondiabetic subjects
Akiko Toda, Shigeko Hara, Hiroshi Tsuji, Yasuji Arase
Endocrine.2020; 68(3): 592. CrossRef - Hypothyroidism and Kidney Function: A Mendelian Randomization Study
Christina Ellervik, Samia Mora, Paul M. Ridker, Daniel I. Chasman
Thyroid.2020; 30(3): 365. CrossRef - Relationship between serum thyrotropin and urine albumin excretion in euthyroid subjects with diabetes
Gautam Das, Peter N Taylor, Hussam Abusahmin, Amer Ali, Brian P Tennant, John Geen, Onyebuchi Okosieme
Annals of Clinical Biochemistry: International Journal of Laboratory Medicine.2019; 56(1): 155. CrossRef - The longitudinal effect of subclinical hypothyroidism on urine microalbumin-to-urine creatinine ratio in patients with type 2 diabetes mellitus
Juan Xie, Xiaoqing Wang, Yiyuan Zhang, Hailun Li, Yong Xu, Donghui Zheng
BMC Endocrine Disorders.2019;[Epub] CrossRef - The effect of l-thyroxine substitution on oxidative stress in early-stage diabetic nephropathy patients with subclinical hypothyroidism: a randomized double-blind and placebo-controlled study
Yong Chen, Ganlin Wu, Meirong Xu
International Urology and Nephrology.2018; 50(1): 97. CrossRef - Association between subclinical hypothyroidism and diabetic nephropathy in type 2 diabetes
Ayman Abd-Elrahman Mohamed Nsr-Allah, Azza H. Abd-El Fatah, Mahmoud Azab, Maggie M. Fawzi
The Egyptian Journal of Internal Medicine.2018; 30(3): 160. CrossRef - Role of Microalbuminuria in Predicting Cardiovascular Mortality in Individuals With Subclinical Hypothyroidism
Tushar A. Tuliani, Maithili Shenoy, Kevin Belgrave, Abhishek Deshmukh, Sadip Pant, Anthony Hilliard, Luis Afonso
The American Journal of the Medical Sciences.2017; 354(3): 285. CrossRef - Inositol(s) in thyroid function, growth and autoimmunity
Salvatore Benvenga, Alessandro Antonelli
Reviews in Endocrine and Metabolic Disorders.2016; 17(4): 471. CrossRef - Hypothyroïdie infra-clinique et risque d’évènements cardiovasculaires chez la femme diabétique de type 2
M. Gourine, K. Bentadj, S. Mostefa-Kara, A. Cherrak, S. Halimi, M. Belhadj
Médecine des Maladies Métaboliques.2016; 10(5): 466. CrossRef - Can levothyroxine treatment reduce urinary albumin excretion rate in patients with early type 2 diabetic nephropathy and subclinical hypothyroidism? A randomized double-blind and placebo-controlled study
Peng Liu, Ruidong Liu, Xia Chen, Yingying Chen, Debao Wang, Fengmei Zhang, Yangang Wang
Current Medical Research and Opinion.2015; 31(12): 2233. CrossRef - Free Triiodothyronine Levels Are Associated with Diabetic Nephropathy in Euthyroid Patients with Type 2 Diabetes
Jingcheng Wu, Xiaohua Li, Yang Tao, Yufei Wang, Yongde Peng
International Journal of Endocrinology.2015; 2015: 1. CrossRef - Letter: Subclinical Hypothyroidism Is Independently Associated with Microalbuminuria in a Cohort of Prediabetic Egyptian Adults (Diabetes Metab J2013;37:450-7)
In-Kyung Jeong
Diabetes & Metabolism Journal.2014; 38(1): 83. CrossRef - Free Triiodothyronine Concentrations Are Inversely Associated with Microalbuminuria
Yulin Zhou, Lei Ye, Tiange Wang, Jie Hong, Yufang Bi, Jie Zhang, Baihui Xu, Jichao Sun, Xiaolin Huang, Min Xu
International Journal of Endocrinology.2014; 2014: 1. CrossRef - Thyroid hormone and heart failure: from myocardial protection to systemic regulation
Laura Sabatino, Giorgio Iervasi, Alessandro Pingitore
Expert Review of Cardiovascular Therapy.2014; 12(10): 1227. CrossRef

 KDA
KDA
 PubReader
PubReader Cite
Cite