- Current
- Browse
- Collections
-
For contributors
- For Authors
- Instructions to authors
- Article processing charge
- e-submission
- For Reviewers
- Instructions for reviewers
- How to become a reviewer
- Best reviewers
- For Readers
- Readership
- Subscription
- Permission guidelines
- About
- Editorial policy
Articles
- Page Path
- HOME > Diabetes Metab J > Volume 43(5); 2019 > Article
-
Original ArticleClinical Diabetes & Therapeutics Efficacy and Safety of Sodium-Glucose Cotransporter-2 Inhibitors in Korean Patients with Type 2 Diabetes Mellitus in Real-World Clinical Practice
-
A Ram Hong1
 , Bo Kyung Koo2,3, Sang Wan Kim2,3, Ka Hee Yi2,3, Min Kyong Moon2,3
, Bo Kyung Koo2,3, Sang Wan Kim2,3, Ka Hee Yi2,3, Min Kyong Moon2,3
-
Diabetes & Metabolism Journal 2019;43(5):590-606.
DOI: https://doi.org/10.4093/dmj.2018.0134
Published online: February 28, 2019
1Department of Internal Medicine, Chonnam National University Medical School, Gwangju, Korea.
2Department of Internal Medicine, Seoul National University College of Medicine, Seoul, Korea.
3Department of Internal Medicine, Seoul Metropolitan Government Seoul National University Boramae Medical Center, Seoul, Korea.
- Corresponding author: Min Kyong Moon. Department of Internal Medicine, Seoul Metropolitan Government Seoul National University Boramae Medical Center, Seoul National University College of Medicine, 20 Boramae-ro 5-gil, Dongjak-gu, Seoul 07061, Korea. mkmoon@snu.ac.kr
Copyright © 2019 Korean Diabetes Association
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/4.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
ABSTRACT
-
Background
- This study aimed to evaluate the efficacy and safety of sodium-glucose cotransporter-2 (SGLT2) inhibitors in Korean patients who had inadequately controlled type 2 diabetes mellitus (T2DM) in real-world clinical practice.
-
Methods
- We included 410 patients who started SGLT2 inhibitors (empagliflozin or dapagliflozin) as add-on therapy or switch therapy between February 2015 and June 2017. The primary efficacy endpoint was a change in glycosylated hemoglobin (HbA1c) from baseline to week 12. The secondary endpoints were patients achieving HbA1c <7.0% and changes in the fasting plasma glucose (FPG), lipid profiles, body weight, and blood pressure (BP).
-
Results
- The mean HbA1c at baseline was 8.5% (8.6% in the add-on group and 8.4% in the switch group). At week 12, the mean adjusted HbA1c decreased by −0.68% in the overall patients (P<0.001), by −0.94% in the add-on group, and by −0.42% in the switch group. Significant reductions in FPG were also observed both in the add-on group and switch group (−30.3 and −19.8 mg/dL, respectively). Serum triglyceride (−16.5 mg/dL), body weight (−2.1 kg), systolic BP (−4.7 mm Hg), and diastolic BP (−1.3 mm Hg) were significantly improved in the overall patients. Approximately 18.3% of the patients achieved HbA1c <7.0% at week 12. A low incidence of hypoglycemia and genital tract infection was observed (6.3% and 2.2%, respectively).
-
Conclusion
- SGLT2 inhibitors can be a suitable option as either add-on or switch therapy for Korean patients with inadequately controlled T2DM.
- The prevalence of type 2 diabetes mellitus (T2DM) is considerably increasing worldwide [1]. In Korea, approximately 5.0 million individuals (14.4%) aged 30 years or older have T2DM [2]. Given the progressive nature of the disease, combinations of various glucose-lowering agents are often needed to achieve glycemic targets [34]. To overcome unmet needs for glycemic control, several new classes of anti-diabetic agents have been recently introduced for better management of T2DM.
- Sodium-glucose cotransporter-2 (SGLT2) inhibitors act by inhibiting renal glucose reabsorption, thereby enhancing glycosuria and reducing blood glucose levels [56]. The efficacy and safety of SGLT2 inhibitors have been reported in several randomized controlled trials (RCTs), which demonstrated improved glycemic control and significant reductions in body weight and blood pressure (BP) with a low risk of hypoglycemia [678]. The recent Empagliflozin Cardiovascular Outcome Event Trial in Type 2 Diabetes Mellitus Patients-Removing Excess Glucose (EMPA-REG) and Canagliflozin Cardiovascular Assessment Study (CANVAS) showed that the SGLT2 inhibitors empagliflozin and canagliflozin have additional benefits of reducing adverse cardiovascular outcomes compared with placebo [910]. According to the position statement published by the American Diabetes Association (ADA) and the European Association for the Study of Diabetes, SGLT2 inhibitors are recommended as second or third-line agents in patients who failed to achieve glycemic target with one or more oral anti-diabetic drugs (OADs) [3]. In addition, because their mechanism of action is independent of insulin secretion or action, SGLT2 inhibitors can be safely combined with insulin therapy and are even effective in patients with long-standing diabetes with β-cell dysfunction [1112].
- Recently, several real-world data from clinical practice settings were reported which showed similar glucose-lowering efficacy of SGLT2 inhibitors as add-on therapy to those from previous RCTs and meta-analyses [131415]. These findings were consistently observed in Korean patients with T2DM [161718]. In actual clinical practice, it may also be considered to change an OAD to other class of OAD if glycemic goal is not achieved, which means switch therapy. Here, we aimed to evaluate the efficacy and safety of SGLT2 inhibitors as add-on therapy and as switch therapy from a real clinical practice perspective in Korean patients with T2DM who exhibited inadequate glycemic control.
INTRODUCTION
- Study population
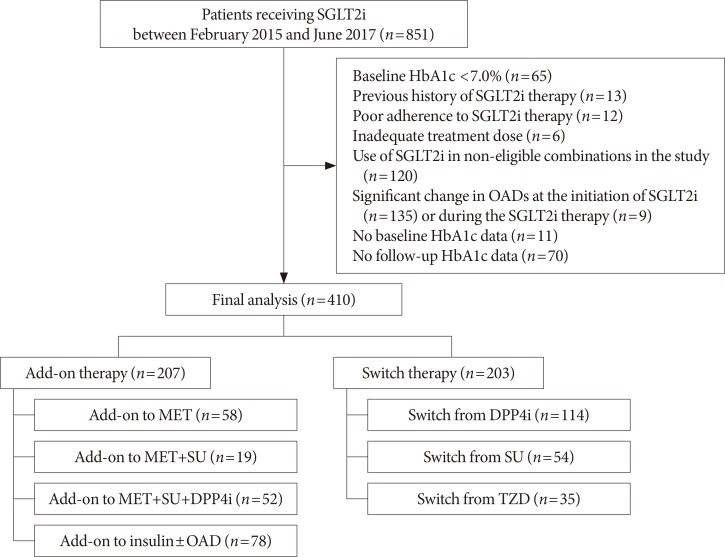
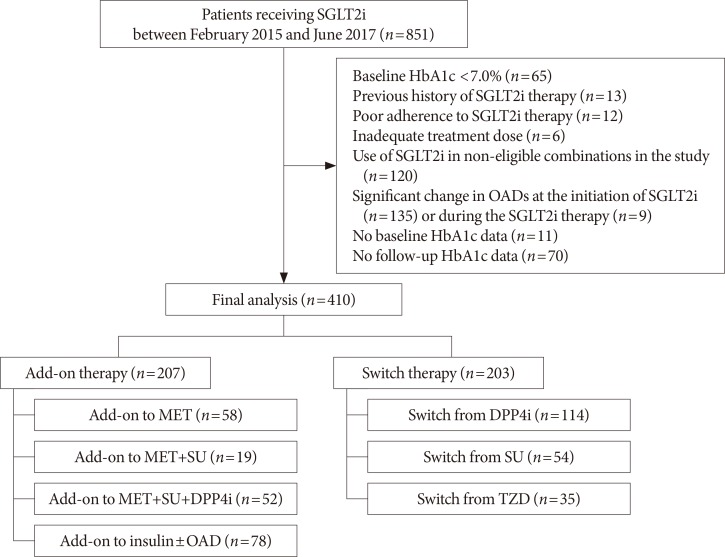
- This was a single-center retrospective study conducted at Seoul Metropolitan Government Seoul National University Boramae Medical Center. The eligible study participants were T2DM patients with inadequate glycemic control (glycosylated hemoglobin [HbA1c] ≥7.0%) who started SGLT2 inhibitors (dapagliflozin or empagliflozin) between February 2015 and June 2017. Given the patients' visit schedule to the outpatient clinic, we regarded the 4 weeks before and after 12 weeks of treatment as an acceptable window for the follow-up HbA1c assessments. Among the 851 initially screened patients, we excluded patients with baseline HbA1c <7.0%, those who had previously used SGLT2 inhibitor, those who took a low dose of SGLT2 inhibitor (dapagliflozin 5 mg) or with poor treatment adherence for primary analysis, had considerable change in OAD class or dose at the initiation of SGLT2 inhibitor or during the treatment, and were not available for baseline or follow-up HbA1c test. We further excluded patients who received SGLT2 inhibitor as monotherapy or received it in combination with other treatments deemed ineligible for the analysis in the present study. The patients were divided into two groups according to their use of SGLT2 inhibitor: the add-on group (i.e., those who used SGLT2 inhibitors in addition to other OADs) and the switch group (i.e., those who switched from other OADs to SGLT2 inhibitors). The add-on group was further stratified by background therapy as add-on to metformin (MET), MET+sulfonylurea (SU), MET+SU+dipeptidyl peptidase 4 (DPP4) inhibitor, and insulin±OAD. Meanwhile, the switch group was further stratified by previously received OADs as switch from DPP4 inhibitor, SU, and thiazolidinedione (TZD). Finally, 410 patients (207 patients in the add-on group and 203 patients in the switch group) were included for analysis. The flow diagram for patient selection is shown in Fig. 1.
- Patient characteristics including age, height, body weight, BP, duration of diabetes, comorbidities, and concomitant medications for hypertension, dyslipidemia, and cardiovascular disease were examined at baseline.
- This study was approved by the Institutional Review Board of Seoul Metropolitan Government Seoul National University Boramae Medical Center (No. 10-2017-27) and was conducted in accordance with the Declaration of Helsinki. The need for informed consent was waived due to the retrospective nature of the study.
- Efficacy and safety assessments
- The primary efficacy endpoint was a change in HbA1c between baseline and week 12 (±4 week). The secondary efficacy endpoints were changes in fasting plasma glucose (FPG), lipid profiles, BP, and body weight after 12 weeks of treatment. The proportion of patients who achieved the glycemic target of HbA1c <7.0% was also evaluated following ADA 2017 recommendation [19]. We further investigated clinical characteristics of patients who have shown good response to SGLT2 inhibitor therapy. We defined good responders as those with HbA1c <7.0% or a decrease in HbA1c of more than 1.0% at 12 weeks after SGLT2 inhibitor therapy [20].
- The safety evaluations included self-monitored hypoglycemia and adverse event of hepatic and renal function based on medical records during the treatment with SGLT2 inhibitors. Hepatic adverse event was defined as over 3-fold increase of alanine aminotransferase (ALT) or aspartate aminotransferase (AST) of the patients' baseline levels. Renal adverse event was considered as a ≥30% decline of estimated glomerular filtration rate (eGFR) from baseline levels as calculated using the Modification of Diet in Renal Disease-equation (mL/min/1.73 m2) [21].
- Laboratory measurements
- HbA1c level was determined using high-performance liquid chromatography (SST; Becton, Dickinson and Company, Franklin Lakes, NJ, USA). FPG was measured in the 12-hour fasted state using the glucose oxidase method (Hitachi 747 chemistry analyzer; Hitachi, Tokyo, Japan). Fasting insulin level was measured using an immunoradiometric assay (DIAsource ImmunoAssays, Nivelles, Belgium). The homeostasis model assessment (HOMA) was used to evaluate pancreatic β-cell function (HOMA-β) and insulin resistance (HOMA-IR) [22]. The fasting total cholesterol, high density lipoprotein cholesterol (HDL-C), low density lipoprotein cholesterol (LDL-C), and triglyceride (TG) levels were measured using an enzymatic colorimetric method (Toshiba Medical System Co. Ltd., Tokyo, Japan). The serum concentrations of AST, ALT, and creatinine were measured using a Hitachi 747.
- Statistical analysis
- All measurements were expressed as the mean±standard deviation (SD) or number (%). Categorical variables were compared using the chi-square test. The changes in HbA1c, FPG, body weight, and lipid profiles between baseline and week 12 within each treatment group were analyzed by via paired t-test. Comparisons across the treatment groups were performed using analysis of covariance after adjusting for their baseline levels. The changes in each parameter were presented as adjusted least square (LS) means and standard error. The predictive factors for SGLT2 inhibitor response were obtained using univariate and multivariate logistic regression analyses. Risk was reported with odds ratios and 95% confidence intervals (CIs). A P<0.05 was considered statistically significant. All statistical analyses were performed using SPSS statistics version 21 for Windows (IBM Corp., Armonk, NY, USA).
METHODS
- Baseline characteristics of study subjects
- The baseline demographic and clinical characteristics of study subjects are presented in Table 1. The mean age was 59.6 years, and the mean duration of diabetes was 12.0 years. Approximately 37.6% of patients were older than 65 years. The mean body weight was 71.6 kg, and the mean body mass index (BMI) was 27.3 kg/m2, with 46.3% of patients classified as obese (BMI ≥25 kg/m2) according to the definition of obesity for Asians [23]. Approximately 37.6% of the patients were treated with insulin in overall patients. The proportion of patients with titration for insulin doses (>10% of the baseline) was 20.1% [24]. The patients' clinical characteristics were comparable between the two groups except for the type of SGLT2 inhibitor therapy, in which a higher proportion of patients were receiving dapagliflozin as add-on therapy than switch therapy (P=0.001).
- At baseline, the average HbA1c was 8.6% in the add-on group and 8.4% in the switch group. The mean concentration of FPG was significantly higher in the add-on group than that in the switch group (164.8 mg/dL vs. 148.4 mg/dL, P<0.001). Meanwhile, BP and lipid profiles did not differ between the two groups. In addition, there were also no significant differences in the baseline AST, ALT, serum creatinine, and eGFR levels. The add-on and switch therapy groups were also similar in terms of the prevalence of hypertension, dyslipidemia, or cardiovascular disease and use of concurrent medications including statins, fenofibrates, omega-3 fatty acids, angiotensin-converting enzyme inhibitors (or angiotensin II receptor blocker), or anti-platelet agents.
- Efficacy
- At 12 weeks, SGLT2 inhibitors exhibited a significant reduction in HbA1c levels of −0.68% (95% CI, −0.78 to −0.58) in the overall patients (P<0.001). The mean adjusted HbA1c decreased by −0.94% in the add-on group, and by −0.42% in the switch group (both P<0.001). The between-treatment difference in the LS mean change was −0.52% (95% CI, −0.68 to −0.37; P<0.001) (Table 2). There was a significant reduction of −25.1 mg/dL (95% CI, −29.5 to −20.8) in the FPG level in the overall patients (P<0.001). The add-on group and switch group exhibited −30.3 and −19.8 mg/dL of reduction in FPG levels, respectively (both P<0.001). The between-treatment difference in the LS mean change was −10.5 mg/dL (95% CI, −16.6 to −4.4; P=0.001). In the overall patients, there were also considerable reductions in serum TG level, body weight, systolic BP, and diastolic BP of −16.5 mg/dL (95% CI, −24.2 to −8.7), −2.1 kg (95% CI, −2.4 to −1.7), −4.7 mm Hg (95% CI, −6.1 to −3.2), and −1.3 mm Hg (95% CI, −2.4 to −0.2), respectively. Serum HDL-C, LDL-C, and eGFR levels did not differ between baseline and week 12.
- We further compared changes in cardiometabolic parameters in each treatment group according to the background glucose-lowering therapy. In the add-on group, the glucose-lowering effect did not differ between add-on to MET, MET+SU, and MET+SU+DPP4 inhibitor (LS mean change of −1.20%, −1.16%, and −1.06%, respectively), whereas, insulin add-on showed a smaller reduction of HbA1c than that in other regimens (LS mean change of −0.71%; between-treatment difference from add-on to MET of −0.49% [95% CI, −0.84 to −0.15; P=0.001]). The magnitude of HbA1c reduction did not differ among previous OADs in the switch group (Table 3). We observed an increase in serum TG level for switching from TZD compared with switching from DPP4 inhibitor (P=0.002). There were no between-treatment differences in FPG, LDL-C, body weight, systolic BP, and diastolic BP in both treatment groups according to the background therapy (Supplementary Tables 1 and 2).
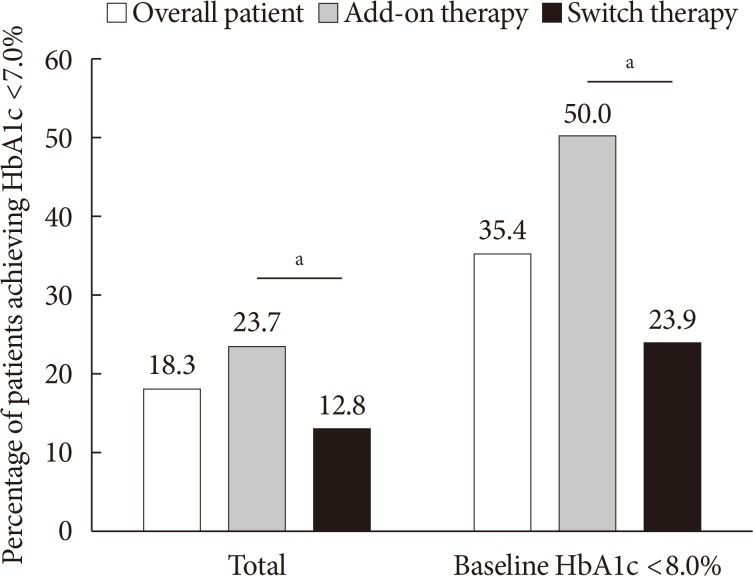
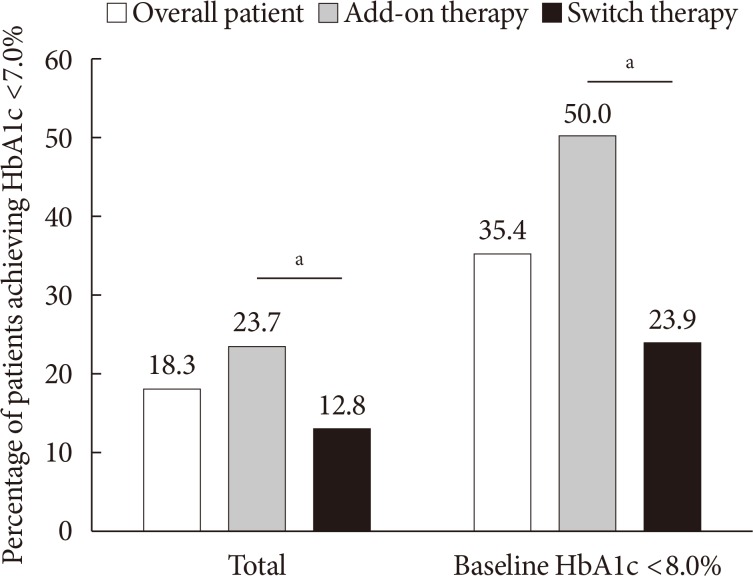
- The proportion of patients who achieved HbA1c <7.0% was 18.3% in overall patients, 23.7% in the add-on group, and 12.8% in the switch group (P=0.004). Among the patients with baseline HbA1c levels <8.0% (n=158), 50.0% in the add-on group and 23.9% in the switch group achieved HbA1c levels <7.0% at week 12 (P=0.001) (Fig. 2). In the add-on group, 46.6%, 47.4%, 11.5%, and 9.0% of the patients in the add-on to MET, MET+SU, MET+SU+DPP4 inhibitor, and insulin±OAD subgroups, respectively, exhibited HbA1c <7.0% at week 12 (P<0.001). In the switch group, 15.8%, 7.4%, and 11.4% of the patients in the DPP4 inhibitor, SU, and TZD subgroups, respectively, exhibited HbA1c <7.0% (P=0.305).
- In overall, 171 patients (41.7%) experienced HbA1c <7.0% or a decrease in HbA1c >1.0% and were categorized as good responders. Higher baseline glycemic parameters (HbA1c and FPG) and higher HOMA-IR were associated with a SGLT2 inhibitor response. Patients receiving insulin therapy showed a poor response to SGLT2 inhibitors (Table 4). To identify predictive factors for good responders, we performed logistic regression analyses. Using univariate analysis, higher baseline HbA1c and FPG levels, and no insulin therapy were associated with a good response to SGLT2 inhibitors. Even after multivariate analysis, higher baseline HbA1c and no insulin therapy had significant associations with good responders (Supplementary Table 3). In subgroup analysis, higher baseline HbA1c and no insulin therapy were significantly associated with good responders in the add-on group, whereas only higher HbA1c level remained significant in the switch group using multivariate analysis (Supplementary Tables 4 and 5).
- Safety
- In the present study, two patients showed treatment-emergent ≥3-fold increase in ALT or ALT compared with baseline levels. However, the actual AST and ALT levels were within normal limits, indicating no clinically relevant hepatic adverse event. Four patients showed more than 30% decrease in eGFR levels after initiation of SGLT2 inhibitor. All four patients had baseline eGFR levels of ≥60 mL/min/1.73 m2, and only one patient had eGFR <60 mL/min/1.73 m2 after starting SGLT2 inhibitor treatment. No patient required renal replacement therapy during the treatment period.
- Overall, hypoglycemia and genital tract infection was observed in 26 (6.3%) and nine patients (2.2%), respectively. The frequency of hypoglycemia was higher in the patients receiving insulin therapy than those receiving OADs (10.4% vs. 3.9%, P=0.009). Diabetic ketoacidosis did not occur during the treatment period.
RESULTS
- In this real-world study, we demonstrated that SGLT2 inhibitors significantly improved glycemic control in Korean patients with inadequately controlled T2DM (change in HbA1c of −0.68% in the overall study population; −0.94% in the add-on group; and −0.42% in the switch group). Treatment with SGLT2 inhibitors also exhibited significant improvements in FPG, TG, body weight, systolic BP, and diastolic BP. Our findings are consistent with previous RCTs that support the efficacy of SGLT2 inhibitors in patients with T2DM in a real-world setting.
- Because previous RCT data were limited to analyses for add-on therapy, we separately examined the efficacy SGLT2 inhibitors as add-on and switch therapy. SGLT2 inhibitors when used in combination with OADs or insulin were shown to be significantly effective in reducing HbA1c, and this finding was comparable to the results from RCTs [7] and those from the real-world setting [2526]. The glucose-lowering efficacy of SGLT2 inhibitors did not differ between its combination with MET, MET+SU, and MET+SU+DPP4; however, the efficacy was greater than that in the combination with insulin. Meanwhile, the adjusted mean change of HbA1c with add-on to MET (−1.20%) was greater in the current study than those from RCTs (−0.94% to −0.56%) [2728293031]. The efficacy of SGLT2 inhibitors as an add-on to MET+SU in HbA1c reduction (−1.16% vs. −1.06% to −0.82%) was better in the current study than that reported in RCTs [323334]. Meanwhile, the observed reductions of HbA1c in the other add-on subgroups (Table 3) were comparable to those obtained from RCTs, and these findings seem to be attributed to higher baseline HbA1c level in our study than those in previous studies.
- The additive glucose-lowering effect of SGLT2 inhibitors to insulin was greater than those reported in a meta-analysis (HbA1c reduction, −0.71% vs. −0.56%) [12]. In our study, insulin doses were self-titrated for the individual patient during the treatment period. Along with the insulin-independent mechanism of action, SLGT2 inhibitors may also improve insulin sensitivity and thereby preserve β-cell function [113536]. It is difficult to determine why our study showed better glucose-lowering efficacy than previous reports. However, it may be partly attributed to the different pathophysiology of diabetes between Caucasian and Asians patients, including Koreans. Compared with Caucasian patients, Asian patients with T2DM have reduced β-cell function and higher insulin sensitivity [3738]. There might be also some differences in the pharmacokinetic concentration and pharmacodynamic response of SGLT2 inhibitors between Asians and Caucasian T2DM populations caused by body size difference, similar to those observed in DPP4 inhibitors [3940]. Therefore, differences in ethnic group and characteristics of the study population seem to influence the findings of our study. In addition, our study participants may have greater β-cell deterioration due to the long disease duration (>10 years), which may result in higher baseline HbA1c than those in the previous RCTs. This could further contribute to a greater reduction to SGLT2 inhibitors in the present study.
- In addition to HbA1c, we observed improvements in FPG (−25.1 mg/dL), body weight (−2.1 kg), systolic BP (−4.7 mm Hg), and diastolic BP (−1.3 mm Hg). These findings were comparable to meta-analysis data of RCTs that showed significant reduction in FPG of 19.8 to 34.2 mg/dL, body weight of 1.6 to 2.5 kg, systolic BP of 2.8 to 4.9 mm Hg, and diastolic BP of 1.5 to 2.0 mm Hg [7]. Further, SGLT2 inhibitors significantly improved serum TG levels (−16.5 mg/dL) in the current study, which is consistent with the findings of RCTs, whereas, HDL-C and LDL-C did not differ between baseline and 12 weeks after treatment. Because majority of patients were already receiving statin therapy, treatment with SGLT2 inhibitors may not result in meaningful changes in lipid profiles. However, given the limited number of our study participants, further studies are needed to investigate this issue. Although we could not evaluate the incidence of major adverse cardiovascular events due to the short treatment period, the observed significant improvements in cardiometabolic parameters suggest a potential microvascular and macrovascular benefit with SGLT2 inhibitors.
- Due to retrospective nature of this study, we examined relative reduction in HbA1c after switching from several OADs to SGLT2 inhibitors, instead of direct comparison of glucose-lowering efficacy between SGLT2 inhibitors and other OADs. Switching to SGLT2 inhibitors demonstrated significant reduction in HbA1c of −0.42% regardless of previous OAD classes, including DPP4 inhibitor, SU, and TZD. A total of 122 patients (60.1%) showed reduction in HbA1c with SGLT2 inhibitors, and 105 (51.7%) showed more than 0.3% reduction in HbA1c in the switch group. FPG and serum TG were also substantially reduced after 12-week treatment with SLGT2 inhibitors. Furthermore, switch therapy to SGLT2 inhibitors showed additional benefits for reduction of body weight and systolic BP. Because all switch and add-on regimens showed significant glycemic improvement with SGLT2 inhibitor, we considered the possibility of selection bias in our study participants; if patients who stopped SGLT2 inhibitors early due to poor treatment response were excluded from the analysis, the glycemic efficacy may be overestimated. Hence, we thoroughly reviewed 12 patients who were excluded in the final analysis due to poor adherence to SGLT2 inhibitors (Fig. 1). There was only one patient who stopped the medication because of poor glycemic response, and she took high-dose glucocorticoid during the period when she was receiving dapagliflozin. Considering the degree of HbA1c reduction after switching to SGLT2 inhibitors, it is possible that altering OAD itself may improve the patients' lifestyle in a positive direction, which may further enhance glycemic and cardiometabolic parameters. Collectively, our findings suggest that switching to SGLT2 inhibitors may be an attractive regimen in patients with inadequately controlled T2DM in the real-clinical practice.
- We analyzed the clinical predictors of a good response to SGLT2 inhibitors. Higher glycemic parameters and no treatment with insulin therapy were linked with a SGLT2 inhibitor response. A previous study showed that younger age, male sex, higher glycemic parameter, lower BMI, short duration of T2DM, and higher eGFR were linked to a good response to dapagliflozin in Korean patients [16]. This study defined the good responder as a ≥10% reduction in HbA1c value after 12 weeks of dapagliflozin, and add-on and switch group were not separately analyzed.
- In the current study, the overall incidence of self-reported hypoglycemia and genital tract infections was similar or slightly lower than those reported in RCTs (6.3% in hypoglycemia; 2.2% in genital tract infection) [3241424344454647]. When we considered the cases who discontinued SGLT2 inhibitor early due to these adverse events—mild vaginitis (n=1) and hypoglycemia (n=1)—their overall incidences were still low. All reported cases of hypoglycemia and genital symptoms were mild in our study. No clinically relevant adverse hepatic or renal event and diabetic ketoacidosis were observed during the treatment period. This further supports the safety profiles of SGLT2 inhibitors shown by RCTs in a real-world setting.
- There are several limitations of our study to be considered. First, because this was a retrospective study, there was substantial heterogeneity across the background glucose-lowering therapy for individual patients. In addition, we could not systematically collect information about adverse events. In the present study, the occurrence of adverse events including hypoglycemia and genital tract infection were determined by self-report from patients. Hence, there may be missing data and misdiagnosis for the safety profiles related to SGLT2 inhibitors. For example, in the case of genital infection, it is possible that confusion with urinary tract infection has occurred. Second, because a substantial proportion of patients was receiving dapagliflozin (10 mg), direct comparisons of glucose-lowering efficacy and drug-related safety profiles according to the dose and type of SGLT2 inhibitor were not possible. Third, because canagliflozin is not yet commercially available in South Korea, and dapagliflozin and empagliflozin are the only available SGLT2 inhibitors in our institution, we collected data for these two drugs. Fourth, this study was conducted over a relatively short 12-week period. Thus, further studies are needed to investigate long-term effect and tolerability of SGLT2 inhibitors in real clinical practice.
- Despite these limitations, the present study is valuable to investigate the real-world efficacy and safety profiles of SGLT2 inhibitors in Korean patients and to comparatively assess them based on the various combination regimens of SGLT2 inhibitors. There have been few real-world studies of SGLT2 inhibitors reported in Koreans. Previous two studies evaluated the efficacy and safety of dapagliflozin or empagliflozin-based quadruple therapy in Koreans [1718]. Another Korean study examined the efficacy of dapagliflozin and clinical characteristics of good responders to dapagliflozin [16]. Our study is the first Korean study that analyzed the efficacy and safety of dapagliflozin and empagliflozin as add-on or switch therapy in various combinations of background glucose-lowering therapy.
- In conclusion, SGLT2 inhibitors as add-on or switch therapy resulted in a greater reduction in HbA1c in Korean patients with inadequately controlled T2DM in a real-world setting. SGLT2 inhibitors also demonstrated substantial reductions in body weight and BP and improvement of lipid profiles. These findings indicate that SGLT2 inhibitors as add-on or switch regimen may be applicable in real clinical practice, particularly in patients with inadequately controlled T2DM under treatment with dual or triple drug combinations.
DISCUSSION
-
Acknowledgements
- None
ACKNOWLEDGMENTS
-
CONFLICTS OF INTEREST: No potential conflict of interest relevant to this article was reported.
-
AUTHOR CONTRIBUTIONS:
NOTES
SUPPLEMENTARY MATERIALS
Supplementary Table 1
Supplementary Table 2
Supplementary Table 3
Supplementary Table 4
Supplementary Table 5
- 1. NCD Risk Factor Collaboration (NCD-RisC). Worldwide trends in diabetes since 1980: a pooled analysis of 751 population-based studies with 4.4 million participants. Lancet 2016;387:1513-1530. PubMedPMC
- 2. Korean Diabetes Association. Diabetes fact sheet in Korea 2018 cited 2019 Jan 12. http://www.diabetes.or.kr/bbs/skin/dianews/download.php?code=admin&number=1694.
- 3. Inzucchi SE, Bergenstal RM, Buse JB, Diamant M, Ferrannini E, Nauck M, Peters AL, Tsapas A, Wender R, Matthews DR. Management of hyperglycaemia in type 2 diabetes, 2015: a patient-centred approach. Update to a position statement of the American Diabetes Association and the European Association for the Study of Diabetes. Diabetologia 2015;58:429-442. ArticlePubMedPDF
- 4. American Diabetes Association. 8. Pharmacologic approaches to glycemic treatment. Diabetes Care 2017;40(Suppl 1):S64-S74. ArticlePubMedPDF
- 5. Abdul-Ghani MA, DeFronzo RA. Lowering plasma glucose concentration by inhibiting renal sodium-glucose cotransport. J Intern Med 2014;276:352-363. ArticlePubMedPMCPDF
- 6. Ferrannini E, Solini A. SGLT2 inhibition in diabetes mellitus: rationale and clinical prospects. Nat Rev Endocrinol 2012;8:495-502. ArticlePubMedPDF
- 7. Zaccardi F, Webb DR, Htike ZZ, Youssef D, Khunti K, Davies MJ. Efficacy and safety of sodium-glucose co-transporter-2 inhibitors in type 2 diabetes mellitus: systematic review and network meta-analysis. Diabetes Obes Metab 2016;18:783-794. ArticlePubMed
- 8. Vasilakou D, Karagiannis T, Athanasiadou E, Mainou M, Liakos A, Bekiari E, Sarigianni M, Matthews DR, Tsapas A. Sodium-glucose cotransporter 2 inhibitors for type 2 diabetes: a systematic review and meta-analysis. Ann Intern Med 2013;159:262-274. ArticlePubMed
- 9. Zinman B, Wanner C, Lachin JM, Fitchett D, Bluhmki E, Hantel S, Mattheus M, Devins T, Johansen OE, Woerle HJ, Broedl UC, Inzucchi SE. EMPA-REG OUTCOME Investigators. Empagliflozin, cardiovascular outcomes, and mortality in type 2 diabetes. N Engl J Med 2015;373:2117-2128. ArticlePubMed
- 10. Neal B, Perkovic V, Mahaffey KW, de Zeeuw D, Fulcher G, Erondu N, Shaw W, Law G, Desai M, Matthews DR. CANVAS Program Collaborative Group. Canagliflozin and cardiovascular and renal events in type 2 diabetes. N Engl J Med 2017;377:644-657. ArticlePubMed
- 11. Singh AK, Singh R. Sodium-glucose co-transporter-2 inhibitors as add-on therapy to insulin: rationale and evidences. Expert Rev Clin Pharmacol 2016;9:409-418. ArticlePubMed
- 12. Tang H, Cui W, Li D, Wang T, Zhang J, Zhai S, Song Y. Sodium-glucose co-transporter 2 inhibitors in addition to insulin therapy for management of type 2 diabetes mellitus: a meta-analysis of randomized controlled trials. Diabetes Obes Metab 2017;19:142-147. ArticlePubMedPDF
- 13. Tamez-Perez HE, Delgadillo-Esteban E, Soni-Duque D, Hernandez-Coria MI, Tamez-Pena AL. SGLT2 inhibitors as add on therapy in type 2 diabetes: a real world study. J Diabetes Metab Disord 2017;16:27. ArticlePubMedPMCPDF
- 14. Brown RE, Gupta N, Aronson R. Effect of dapagliflozin on glycemic control, weight, and blood pressure in patients with type 2 diabetes attending a specialist endocrinology practice in Canada: a retrospective cohort analysis. Diabetes Technol Ther 2017;19:685-691. ArticlePubMed
- 15. Blonde L, Patel C, Bookhart B, Pfeifer M, Chen YW, Wu B. A real-world analysis of glycemic control among patients with type 2 diabetes treated with canagliflozin versus dapagliflozin. Curr Med Res Opin 2018;34:1143-1152. ArticlePubMed
- 16. Han E, Kim A, Lee SJ, Kim JY, Kim JH, Lee WJ, Lee BW. Characteristics of dapagliflozin responders: a longitudinal, prospective, nationwide dapagliflozin surveillance study in Korea. Diabetes Ther 2018;9:1689-1701. ArticlePubMedPMCPDF
- 17. Ku EJ, Lee DH, Jeon HJ, Oh TK. Effectiveness and safety of empagliflozin-based quadruple therapy compared with insulin glargine-based therapy in patients with inadequately controlled type 2 diabetes: an observational study in clinical practice. Diabetes Obes Metab 2019;21:173-177. ArticlePubMedPDF
- 18. Jeon HJ, Ku EJ, Oh TK. Dapagliflozin improves blood glucose in diabetes on triple oral hypoglycemic agents having inadequate glucose control. Diabetes Res Clin Pract 2018;142:188-194. ArticlePubMed
- 19. Standards of medical care in diabetes: 2017. Summary of revisions. Diabetes Care 2017;40(Suppl 1):S4-S5. ArticlePubMedPDF
- 20. Horie I, Haraguchi A, Sako A, Akeshima J, Niri T, Shigeno R, Ito A, Nozaki A, Natsuda S, Akazawa S, Mori Y, Ando T, Kawakami A, Abiru N. Predictive factors of efficacy of combination therapy with basal insulin and liraglutide in type 2 diabetes when switched from longstanding basal-bolus insulin: association between the responses of β- and α-cells to GLP-1 stimulation and the glycaemic control at 6 months after switching therapy. Diabetes Res Clin Pract 2018;144:161-170. ArticlePubMed
- 21. Levey AS, Bosch JP, Lewis JB, Greene T, Rogers N, Roth D. A more accurate method to estimate glomerular filtration rate from serum creatinine: a new prediction equation. Modification of Diet in Renal Disease Study Group. Ann Intern Med 1999;130:461-470. ArticlePubMed
- 22. Matthews DR, Hosker JP, Rudenski AS, Naylor BA, Treacher DF, Turner RC. Homeostasis model assessment: insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia 1985;28:412-419. ArticlePubMedPDF
- 23. World Health Organization. The Asia-Pacific perspective: redefining obesity and its treatment cited 2019 Jan 12. Available from: http://www.wpro.who.int/nutrition/documents/docs/Redefiningobesity.pdf.
- 24. Wilding JP, Woo V, Rohwedder K, Sugg J, Parikh S. Dapagliflozin 006 Study Group. Dapagliflozin in patients with type 2 diabetes receiving high doses of insulin: efficacy and safety over 2 years. Diabetes Obes Metab 2014;16:124-136. ArticlePubMed
- 25. Chow W, Miyasato G, Kokkotos FK, Bailey RA, Buysman EK, Henk HJ. Real-world canagliflozin utilization: glycemic control among patients with type 2 diabetes mellitus: a multi-database synthesis. Clin Ther 2016;38:2071-2082. ArticlePubMed
- 26. Scheerer MF, Rist R, Proske O, Meng A, Kostev K. Changes in HbA1c, body weight, and systolic blood pressure in type 2 diabetes patients initiating dapagliflozin therapy: a primary care database study. Diabetes Metab Syndr Obes 2016;9:337-345. ArticlePubMedPMCPDF
- 27. Lavalle-Gonzalez FJ, Januszewicz A, Davidson J, Tong C, Qiu R, Canovatchel W, Meininger G. Efficacy and safety of canagliflozin compared with placebo and sitagliptin in patients with type 2 diabetes on background metformin monotherapy: a randomised trial. Diabetologia 2013;56:2582-2592. ArticlePubMedPMCPDF
- 28. Rosenstock J, Seman LJ, Jelaska A, Hantel S, Pinnetti S, Hach T, Woerle HJ. Efficacy and safety of empagliflozin, a sodium glucose cotransporter 2 (SGLT2) inhibitor, as add-on to metformin in type 2 diabetes with mild hyperglycaemia. Diabetes Obes Metab 2013;15:1154-1160. ArticlePubMed
- 29. Haring HU, Merker L, Seewaldt-Becker E, Weimer M, Meinicke T, Broedl UC, Woerle HJ. EMPA-REG MET Trial Investigators. Empagliflozin as add-on to metformin in patients with type 2 diabetes: a 24-week, randomized, double-blind, placebo-controlled trial. Diabetes Care 2014;37:1650-1659. ArticlePubMedPDF
- 30. DeFronzo RA, Lewin A, Patel S, Liu D, Kaste R, Woerle HJ, Broedl UC. Erratum. Combination of empagliflozin and linagliptin as second-line therapy in subjects with type 2 diabetes inadequately controlled on metformin. Diabetes Care 2015;38:384-393. 1173PubMed
- 31. Ross S, Thamer C, Cescutti J, Meinicke T, Woerle HJ, Broedl UC. Efficacy and safety of empagliflozin twice daily versus once daily in patients with type 2 diabetes inadequately controlled on metformin: a 16-week, randomized, placebo-controlled trial. Diabetes Obes Metab 2015;17:699-702. ArticlePubMed
- 32. Haring HU, Merker L, Seewaldt-Becker E, Weimer M, Meinicke T, Woerle HJ, Broedl UC. EMPA-REG METSU Trial Investigators. Empagliflozin as add-on to metformin plus sulfonylurea in patients with type 2 diabetes: a 24-week, randomized, double-blind, placebo-controlled trial. Diabetes Care 2013;36:3396-3404. PubMedPMC
- 33. Matthaei S, Bowering K, Rohwedder K, Grohl A, Parikh S. Study 05 Group. Dapagliflozin improves glycemic control and reduces body weight as add-on therapy to metformin plus sulfonylurea: a 24-week randomized, double-blind clinical trial. Diabetes Care 2015;38:365-372. ArticlePubMedPDF
- 34. Wilding JP, Charpentier G, Hollander P, Gonzalez-Galvez G, Mathieu C, Vercruysse F, Usiskin K, Law G, Black S, Canovatchel W, Meininger G. Efficacy and safety of canagliflozin in patients with type 2 diabetes mellitus inadequately controlled with metformin and sulphonylurea: a randomised trial. Int J Clin Pract 2013;67:1267-1282. ArticlePubMedPDF
- 35. Kern M, Kloting N, Mark M, Mayoux E, Klein T, Bluher M. The SGLT2 inhibitor empagliflozin improves insulin sensitivity in db/db mice both as monotherapy and in combination with linagliptin. Metabolism 2016;65:114-123. ArticlePubMed
- 36. Ahn CH, Oh TJ, Kwak SH, Cho YM. Sodium-glucose cotransporter-2 inhibition improves incretin sensitivity of pancreatic β-cells in people with type 2 diabetes. Diabetes Obes Metab 2018;20:370-377. ArticlePubMedPDF
- 37. Ma RC, Chan JC. Type 2 diabetes in East Asians: similarities and differences with populations in Europe and the United States. Ann N Y Acad Sci 2013;1281:64-91. ArticlePubMedPMCPDF
- 38. Yoon KH, Lee JH, Kim JW, Cho JH, Choi YH, Ko SH, Zimmet P, Son HY. Epidemic obesity and type 2 diabetes in Asia. Lancet 2006;368:1681-1688. ArticlePubMed
- 39. Graefe-Mody U, Retlich S, Friedrich C. Clinical pharmacokinetics and pharmacodynamics of linagliptin. Clin Pharmacokinet 2012;51:411-427. ArticlePubMed
- 40. Hu P, Yin Q, Deckert F, Jiang J, Liu D, Kjems L, Dole WP, He YL. Pharmacokinetics and pharmacodynamics of vildagliptin in healthy Chinese volunteers. J Clin Pharmacol 2009;49:39-49. ArticlePubMed
- 41. Jabbour S, Seufert J, Scheen A, Bailey CJ, Karup C, Langkilde AM. Dapagliflozin in patients with type 2 diabetes mellitus: a pooled analysis of safety data from phase IIb/III clinical trials. Diabetes Obes Metab 2018;20:620-628. ArticlePubMedPDF
- 42. Ferrannini E, Seman L, Seewaldt-Becker E, Hantel S, Pinnetti S, Woerle HJ. A phase IIb, randomized, placebo-controlled study of the SGLT2 inhibitor empagliflozin in patients with type 2 diabetes. Diabetes Obes Metab 2013;15:721-728. ArticlePubMed
- 43. Søfteland E, Meier JJ, Vangen B, Toorawa R, Maldonado-Lutomirsky M, Broedl UC. Empagliflozin as add-on therapy in patients with type 2 diabetes inadequately controlled with linagliptin and metformin: a 24-week randomized, double-blind, parallel-group trial. Diabetes Care 2017;40:201-209. ArticlePubMedPDF
- 44. Roden M, Weng J, Eilbracht J, Delafont B, Kim G, Woerle HJ, Broedl UC. EMPA-REG MONO trial investigators. Empagliflozin monotherapy with sitagliptin as an active comparator in patients with type 2 diabetes: a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Diabetes Endocrinol 2013;1:208-219. ArticlePubMed
- 45. Wilding JP, Norwood P, T'joen C, Bastien A, List JF, Fiedorek FT. A study of dapagliflozin in patients with type 2 diabetes receiving high doses of insulin plus insulin sensitizers: applicability of a novel insulin-independent treatment. Diabetes Care 2009;32:1656-1662. PubMedPMC
- 46. Neal B, Perkovic V, de Zeeuw D, Mahaffey KW, Fulcher G, Ways K, Desai M, Shaw W, Capuano G, Alba M, Jiang J, Vercruysse F, Meininger G, Matthews D. CANVAS Trial Collaborative Group. Efficacy and safety of canagliflozin, an inhibitor of sodium-glucose cotransporter 2, when used in conjunction with insulin therapy in patients with type 2 diabetes. Diabetes Care 2015;38:403-411. ArticlePubMedPDF
- 47. Rosenstock J, Jelaska A, Frappin G, Salsali A, Kim G, Woerle HJ, Broedl UC. EMPA-REG MDI Trial Investigators. Improved glucose control with weight loss, lower insulin doses, and no increased hypoglycemia with empagliflozin added to titrated multiple daily injections of insulin in obese inadequately controlled type 2 diabetes. Diabetes Care 2014;37:1815-1823. ArticlePubMedPDF
REFERENCES
Flow diagram of study subjects. SGLT2i, sodium-glucose cotransporter-2 inhibitor; HbA1c, glycosylated hemoglobin; OAD, oral anti-diabetic drug; MET, metformin; SU, sulfonylurea; DPP4i, dipeptidyl peptidase 4 inhibitor; TZD, thiazolidinedione.
Percentage of patients who achieved glycosylated hemoglobin (HbA1c) <7.0% in overall patients and those with baseline HbA1c <8.0%. aP<0.05 between add-on therapy and switch therapy.
Baseline demographic and clinical characteristics
Values are presented as mean±standard deviation or number (%). Hypertension is defined as a systolic blood pressure ≥140 mm Hg, or diastolic blood pressure ≥90 mm Hg, or taking anti-hypertensive medication. Dyslipidemia is defined as a total cholesterol ≥240 mg/dL or taking lipid-lowering agents [2].
SGLT2, sodium-glucose cotransporter-2; HbA1c, glycosylated hemoglobin; HDL-C, high density lipoprotein cholesterol; LDL-C, low density lipoprotein cholesterol; HOMA-β, homeostasis model assessment of β-cell function; HOMA-IR, homeostasis model assessment of insulin resistance; eGFR, estimated glomerular filtration rate; ACEi, ACE inhibitor; ARB, angiotensin II receptor blocker.
Changes in measurements after 12 weeks of treatment with sodium-glucose cotransporter-2 inhibitors
Values are presented as mean±standard error.
LS, least square; CI, confidence interval; HbA1c, glycosylated hemoglobin; FPG, fasting plasma glucose; TG, triglyceride; HDL-C, high density lipoprotein cholesterol; LDL-C, low density lipoprotein cholesterol; SBP, systolic blood pressure; DBP, diastolic blood pressure; eGFR, estimated glomerular filtration rate.
aDifference in the LS mean change, which was calculated as add-on therapy minus switch therapy, bP<0.05 from baseline in overall patients and each treatment group, cP<0.05 for the between-treatment group difference.
Changes in glycosylated hemoglobin after 12 weeks of treatment with sodium-glucose cotransporter-2 inhibitors as add-on therapy and switch therapy
Values are presented as mean±standard error.
LS, least square; CI, confidence interval; MET, metformin; SU, sulfonylurea; DPP4i, dipeptidyl peptidase 4 inhibitor; TZD, thiazolidinedione.
aDifference in the LS mean change, which was calculated as compared to adding MET in add-on group and switching from DPP4i in switch group, bP<0.05 from baseline in overall patients and each treatment subgroup, cP<0.05 for the between-treatment group difference.
Comparison of clinical characteristics between the good responders and the poor responders in overall patients
Figure & Data
References
Citations

- Effectiveness and Safety of Sodium-Glucose Cotransporter 2 Inhibitors Added to Dual or Triple Treatment in Patients with Type 2 Diabetes Mellitus
Yesol Hong, Yoomin Jeon, Yoona Choi, Tae Kyu Chung, Howard Lee
Diabetes Therapy.2024; 15(2): 487. CrossRef - Real-world assessment of effectiveness and safety profile of remogliflozin etabonate in management of type 2 diabetes mellitus
Bipin Sethi, Subhankar Chowdhury, Supratik Bhattacharya, Sagar Katare, Sachin Suryawanshi, Hanmant Barkate
International Journal of Diabetes in Developing Countries.2023; 43(2): 214. CrossRef - Effects of dapagliflozin compared with glimepiride on body composition in Asian patients with type 2 diabetes inadequately controlled with metformin: The BEYOND study
Hyeong Kyu Park, Kyoung‐Ah Kim, Kyung‐Wan Min, Tae‐Seo Sohn, In Kyung Jeong, Chul Woo Ahn, Nan‐Hee Kim, Ie Byung Park, Ho Chan Cho, Choon Hee Chung, Sung Hee Choi, Kang Seo Park, Seoung‐Oh Yang, Kwan Woo Lee
Diabetes, Obesity and Metabolism.2023; 25(9): 2743. CrossRef - Efficacy and Safety of Evogliptin Add-on Therapy to Dapagliflozin/Metformin Combinations in Patients with Poorly Controlled Type 2 Diabetes Mellitus: A 24-Week Multicenter Randomized Placebo-Controlled Parallel-Design Phase-3 Trial with a 28-Week Extensio
Jun Sung Moon, Il Rae Park, Hae Jin Kim, Choon Hee Chung, Kyu Chang Won, Kyung Ah Han, Cheol-Young Park, Jong Chul Won, Dong Jun Kim, Gwan Pyo Koh, Eun Sook Kim, Jae Myung Yu, Eun-Gyoung Hong, Chang Beom Lee, Kun-Ho Yoon
Diabetes & Metabolism Journal.2023; 47(6): 808. CrossRef - Real-world Data of Glycemic Control in a Suburban Population in Northern India during the COVID-19 Pandemic
Jaydip V. Revale, Preeti J. Revale
International Journal of Diabetes and Technology.2023; 2(2): 60. CrossRef - Sodium glucose co-transporter-2 inhibitor, Empagliflozin, is associated with significant reduction in weight, body mass index, fasting glucose, and A1c levels in Type 2 diabetic patients with established coronary heart disease: the SUPER GATE study
Satilmis Bilgin, Ozge Kurtkulagi, Tuba Taslamacioglu Duman, Burcin Meryem Atak Tel, Gizem Kahveci, Murat Kiran, Eray Erge, Gulali Aktas
Irish Journal of Medical Science (1971 -).2022; 191(4): 1647. CrossRef - Efficacy and Safety of Empagliflozin as Add-On Therapy in Patients of Type-2 Diabetes Mellitus
Nauman Wazir, Shafqat Ur Rehman
Journal of Gandhara Medical and Dental Science.2022; 9(1): 24. CrossRef - Five comparative cohorts to assess the risk of genital tract infections associated with sodium‐glucose cotransporter‐2 inhibitors initiation in type 2 diabetes mellitus
Wajd Alkabbani, Arsène Zongo, Jasjeet K. Minhas‐Sandhu, Dean T. Eurich, Baiju R. Shah, Mhd. Wasem Alsabbagh, John‐Michael Gamble
Diabetic Medicine.2022;[Epub] CrossRef - Effect of Dapagliflozin in Combination with Lobeglitazone and Metformin in Korean Patients with Type 2 Diabetes in Real-World Clinical Practice
Da Hea Seo, Young Ju Suh, Yongin Cho, Seong Hee Ahn, Seongha Seo, Seongbin Hong, Yong-ho Lee, Young Ju Choi, Eunjig Lee, So Hun Kim
Yonsei Medical Journal.2022; 63(9): 825. CrossRef - Using real-world data for supporting regulatory decision making: Comparison of cardiovascular and safety outcomes of an empagliflozin randomized clinical trial versus real-world data
Ha Young Jang, In-Wha Kim, Jung Mi Oh
Frontiers in Pharmacology.2022;[Epub] CrossRef - Study comparing the efficacy and renal safety for patients with diabetes switching from dapagliflozin to empagliflozin
Ai-Yu Yang, Hung-Chun Chen
International Journal of Clinical Pharmacy.2021; 43(4): 1015. CrossRef - Empagliflozin Regulates the AdipoR1/p-AMPK/p-ACC Pathway to Alleviate Lipid Deposition in Diabetic Nephropathy
Zhiqin Zhang, Lihua Ni, Lian Zhang, Dongqing Zha, Chun Hu, Lingli Zhang, Huiling Feng, Xiaobao Wei, Xiaoyan Wu
Diabetes, Metabolic Syndrome and Obesity: Targets and Therapy.2021; Volume 14: 227. CrossRef - Efficacy and Safety of Luseogliflozin in Patients with Type 2 Diabetes Complicated by Hepatic Dysfunction: A Single-Site, Single-Arm, Open-Label, Exploratory Trial
Hiroaki Seino
Diabetes Therapy.2021; 12(3): 863. CrossRef - Sodium–Glucose Cotransporter 2 Inhibitors and Risk of Retinal Vein Occlusion Among Patients With Type 2 Diabetes: A Propensity Score–Matched Cohort Study
Min-Kyung Lee, Bongsung Kim, Kyungdo Han, Jae-Hyuk Lee, Minhee Kim, Mee Kyoung Kim, Ki-Hyun Baek, Ki-Ho Song, Hyuk-Sang Kwon, Young-Jung Roh
Diabetes Care.2021; 44(10): 2419. CrossRef - Sodium-Glucose Cotransporter-2 Inhibitors Improve Cardiovascular Dysfunction in Type 2 Diabetic East Asians
Muhammad Afzal, Fahad Al-Abbasi, Muhammad Nadeem, Sultan Alshehri, Mohammed Ghoneim, Syed Imam, Waleed Almalki, Imran Kazmi
Metabolites.2021; 11(11): 794. CrossRef - Sodium-Glucose Cotransporter-2 Inhibitor for Renal Function Preservation in Patients with Type 2 Diabetes Mellitus: A Korean Diabetes Association and Korean Society of Nephrology Consensus Statement
Tae Jung Oh, Ju-Young Moon, Kyu Yeon Hur, Seung Hyun Ko, Hyun Jung Kim, Taehee Kim, Dong Won Lee, Min Kyong Moon
Diabetes & Metabolism Journal.2020; 44(4): 489. CrossRef - Sodium-glucose cotransporter-2 inhibitor for renal function preservation in patients with type 2 diabetes mellitus: A Korean Diabetes Association and Korean Society of Nephrology consensus statement
Tae Jung Oh, Ju-Young Moon, Kyu Yeon Hur, Seung Hyun Ko, Hyun Jung Kim, Taehee Kim, Dong Won Lee, Min Kyong Moon
Kidney Research and Clinical Practice.2020; 39(3): 269. CrossRef - Use and effectiveness of dapagliflozin in patients with type 2 diabetes mellitus: a multicenter retrospective study in Taiwan
Jung-Fu Chen, Yun-Shing Peng, Chung-Sen Chen, Chin-Hsiao Tseng, Pei-Chi Chen, Ting-I Lee, Yung-Chuan Lu, Yi-Sun Yang, Ching-Ling Lin, Yi-Jen Hung, Szu-Ta Chen, Chieh-Hsiang Lu, Chwen-Yi Yang, Ching-Chu Chen, Chun-Chuan Lee, Pi-Jung Hsiao, Ju-Ying Jiang, S
PeerJ.2020; 8: e9998. CrossRef - Long-Term Effectiveness and Safety of SGLT-2 Inhibitors in an Italian Cohort of Patients with Type 2 Diabetes Mellitus
Maria Mirabelli, Eusebio Chiefari, Patrizia Caroleo, Raffaella Vero, Francesco Saverio Brunetti, Domenica Maria Corigliano, Biagio Arcidiacono, Daniela Patrizia Foti, Luigi Puccio, Antonio Brunetti
Journal of Diabetes Research.2019; 2019: 1. CrossRef - An Age of Sodium-Glucose Cotransporter-2 Inhibitor Priority: Are We Ready?
Ji A Seo
Diabetes & Metabolism Journal.2019; 43(5): 578. CrossRef
- Figure
- Related articles
-
- Safety of COVID-19 Vaccines among Patients with Type 2 Diabetes Mellitus: Real-World Data Analysis (Diabetes Metab J 2023;47:356-65)
- Safety of COVID-19 Vaccines among Patients with Type 2 Diabetes Mellitus: Real-World Data Analysis (Diabetes Metab J 2023;47:356-65)
- Clinical and Lifestyle Determinants of Continuous Glucose Monitoring Metrics in Insulin-Treated Patients with Type 2 Diabetes Mellitus
- The Beneficial Effect of Glycemic Control against Adverse Outcomes in Patients with Type 2 Diabetes Mellitus and Chronic Kidney Disease
- Clinical Effects of a Home Care Pilot Program for Patients with Type 1 Diabetes Mellitus: A Retrospective Cohort Study

 KDA
KDA PubReader
PubReader Cite
Cite