- Current
- Browse
- Collections
-
For contributors
- For Authors
- Instructions to authors
- Article processing charge
- e-submission
- For Reviewers
- Instructions for reviewers
- How to become a reviewer
- Best reviewers
- For Readers
- Readership
- Subscription
- Permission guidelines
- About
- Editorial policy
Articles
- Page Path
- HOME > Diabetes Metab J > Volume 42(6); 2018 > Article
-
Original ArticleEpidemiology Association of Thigh Muscle Mass with Insulin Resistance and Incident Type 2 Diabetes Mellitus in Japanese Americans
-
Seung Jin Han1,2
 , Edward J. Boyko2,3, Soo-Kyung Kim4, Wilfred Y. Fujimoto3, Steven E. Kahn3,5, Donna L. Leonetti6
, Edward J. Boyko2,3, Soo-Kyung Kim4, Wilfred Y. Fujimoto3, Steven E. Kahn3,5, Donna L. Leonetti6 -
Diabetes & Metabolism Journal 2018;42(6):488-495.
DOI: https://doi.org/10.4093/dmj.2018.0022
Published online: September 5, 2018
1Department of Endocrinology and Metabolism, Ajou University School of Medicine, Suwon, Korea.
2Seattle Epidemiologic Research and Information Center, VA Puget Sound Health Care System, Seattle, WA, USA.
3Department of Medicine, University of Washington School of Medicine, Seattle, WA, USA.
4Department of Internal Medicine, CHA Bundang Medical Center, CHA University School of Medicine, Seongnam, Korea.
5Hospital and Specialty Medicine Service, VA Puget Sound Health Care System, Seattle, WA, USA.
6Department of Anthropology, University of Washington, Seattle, WA, USA.
- Corresponding author: Seung Jin Han. Department of Endocrinology and Metabolism, Ajou University School of Medicine, 164 World cup-ro, Yeongtong-gu, Suwon 16499, Korea. hsj@ajou.ac.kr
Copyright © 2018 Korean Diabetes Association
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/4.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
ABSTRACT
-
Background
- Skeletal muscle plays a major role in glucose metabolism. We investigated the association between thigh muscle mass, insulin resistance, and incident type 2 diabetes mellitus (T2DM) risk. In addition, we examined the role of body mass index (BMI) as a potential effect modifier in this association.
-
Methods
- This prospective study included 399 Japanese Americans without diabetes (mean age 51.6 years) who at baseline had an estimation of thigh muscle mass by computed tomography and at baseline and after 10 years of follow-up a 75-g oral glucose tolerance test and determination of homeostasis model assessment of insulin resistance (HOMA-IR). We fit regression models to examine the association between thigh muscle area and incidence of T2DM and change in HOMA-IR, both measured over 10 years.
-
Results
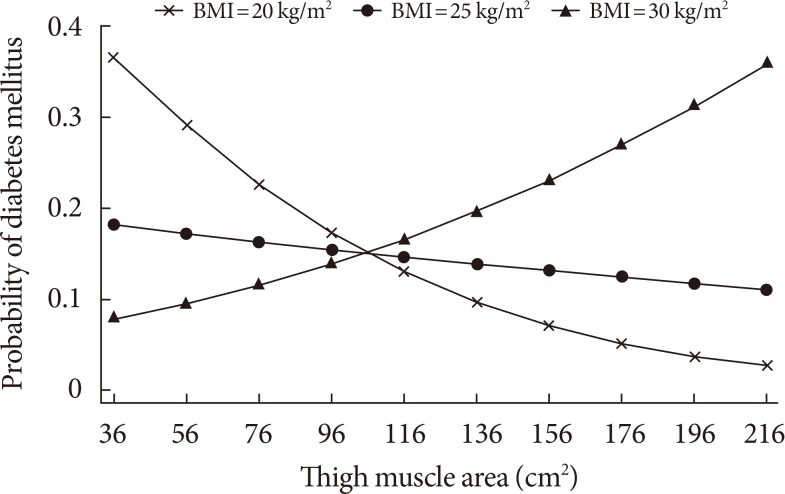
- Thigh muscle area was inversely associated with future HOMA-IR after adjustment for age, sex, BMI, HOMA-IR, fasting plasma glucose, total abdominal fat area, and thigh subcutaneous fat area at baseline (P=0.033). The 10-year cumulative incidence of T2DM was 22.1%. A statistically significant interaction between thigh muscle area and BMI was observed, i.e., greater thigh muscle area was associated with lower risk of incident T2DM for subjects at lower levels of BMI, but this association diminished at higher BMI levels.
-
Conclusion
- Thigh muscle mass area was inversely associated with future insulin resistance. Greater thigh muscle area predicts a lower risk of incident T2DM for leaner Japanese Americans.
- Type 2 diabetes mellitus (T2DM) is one of the most common metabolic disorders in the world, increasing in prevalence with age [1]. Muscle mass gradually decreases with age after the fourth decade of life, even if body weight or body fat mass is unchanged or slightly increased [2]. Skeletal muscle is a major site for glucose uptake and deposition, and hence plays an important role in the regulation of blood glucose levels [3]. Therefore, low muscle mass may affect the development of insulin resistance and T2DM.
- Although a few cross-sectional studies support an inverse association between muscle mass and insulin resistance or T2DM prevalence [456], other studies have failed to show this [789]. In a recent cross-sectional study, thigh muscle area directly measured by computed tomography (CT) was negatively associated with T2DM in South Asians but not Europeans, suggesting the possibility of ethnic differences in this relationship [8]. To our knowledge, no longitudinal research exists on the association between thigh muscle area and change in insulin resistance.
- There are three prospective investigations that have examined the association between muscle mass and incident T2DM with conflicting results. First, overall muscle mass measured by dual-energy X-ray absorptiometry was not a risk factor for incident T2DM in an Australian community-dwelling cohort of men with 5-year follow-up in the Men Androgen Inflammation Lifestyle Environment and Stress study [10]. In the Health ABC study of white and black participants ages 70 to 79 years, no association was observed between abdominal and thigh muscle area measured by CT and incident T2DM [11]. However, there was a significant interaction with body mass index (BMI) for muscle mass in women, so that greater abdominal but not thigh muscle area predicted lower risk of incident T2DM for normal-weight women only, whereas higher thigh muscle mass was associated with greater risk of incident T2DM for overweight/obese women in adjusted models. On the contrary, Son et al. [12] reported low overall muscle mass was associated with a higher risk of T2DM in Korean adults using bioelectrical impedance for muscle measurement. These previous prospective studies have limitations, such as inclusion of male or elderly participants only, self-reported diagnosis of diabetes, and use of bioelectrical impedance, the accuracy of which can be affected by hydration status, age, medical conditions affecting fluid and electrolyte balance, and other factors [13].
- The aim of this study was therefore to investigate the association of thigh muscle mass measured by CT imaging with insulin sensitivity and incident T2DM over a 10-year follow-up in a Japanese American cohort of men and women. We also examined whether generalized adiposity as reflected by BMI modifies the association between thigh muscle mass and incident T2DM.
INTRODUCTION
- Study subjects
- The study received approval from the University of Washington Human Subjects Division and all subjects provided written informed consent (Institutional Review Board number: 34469). Study subjects were from the Japanese American Community Diabetes Study, a cohort of second- and third-generation Japanese Americans of 100% Japanese ancestry. Details of the selection and recruitment of the study subjects have been described previously [14]. Briefly, study participants were selected as volunteers from a community-wide comprehensive mailing list and telephone directory that included almost 95% of the Japanese American population in King County, WA, USA. Participants had follow-up visits 10 to 11 years after the baseline study visit. Among 658 subjects in the original cohort, 166 were excluded because they had diabetes at baseline. Additionally, we excluded 12 subjects who did not have CT measurements of thigh muscle at baseline and 81 subjects who did not complete follow-up examinations. Thus, a total of 399 subjects without diabetes mellitus at baseline were available for analysis.
- Clinical and laboratory examination
- Personal medical history and lifestyle factors were determined using a standardized questionnaire. Family history of diabetes was considered positive if any first-degree relative had diabetes. Smoking status was divided into two categories (current smoker, former/never smoker). Moderate alcohol consumption was defined as intake of more than 6 g of ethanol per day [15]. Physical activity level was estimated by the Paffenbarger physical activity index questionnaire (usual kilocalories spent weekly) [16], and regular physical activity was defined as more than moderate intensity physical activity.
- Blood samples were obtained after an overnight fast of at least 10 hours. Insulin sensitivity was estimated by homeostasis model assessment of insulin resistance (HOMA-IR) index calculated as [fasting serum insulin (µU/mL)×fasting serum glucose (mg/dL)]/405 [17] and the quantitative insulin sensitivity check index (QUICKI) [18]. Diabetes was diagnosed by 75-g oral glucose tolerance test (OGTT), fasting glucose level ≥126 mg/dL or 2-hour post-glucose load ≥200 mg/day, or by treatment with oral glucose-lowering agents or insulin [19].
- A single (1-cm) CT scan slice was obtained of the left thigh at a level halfway between the greater trochanter and the superior margin of the patella and the abdomen at the level of the umbilicus [20]. CT scans were analyzed using density contour software. Tissue with a density of −250 to −50 Hounsfield units (HU) was classified as adipose tissue [20]. Total thigh and thigh subcutaneous fat cross-sectional areas (cm2) were measured and thigh muscle mass was estimated by calculating the difference between total thigh area and thigh subcutaneous fat area because directly measured thigh muscle area was unavailable. This proxy measure of thigh muscle area has been shown to be highly correlated with directly measured thigh muscle area in a validation study conducted in a Korean population (r=0.98; 95% confidence interval, 0.88 to 1.00) [21].
- Statistical analysis
- Continuous variables are expressed as mean±standard deviation, and categorical variables are expressed as numbers and percentages. The distribution of HOMA-IR was skewed and was therefore normalized using a logarithmic transformation for all analyses. An independent t-test was used to compare differences between means for continuous variables, and the chi-square test was used for categorical data. Multiple linear regression analysis was used to determine independent associations between insulin sensitivity at 10 years in relation to baseline thigh muscle mass area. Logistic regression analyses were conducted to estimate the associations between thigh muscle mass area and incident T2DM while adjusting for covariates. Interactions by BMI and sex with thigh muscle mass in relation to incident T2DM was assessed by insertion of first-order interaction terms into the logistic model. The data were analyzed using IBM SPSS Statistics for Windows version 22.0 (IBM Co., Armonk, NY, USA) and Stata version 14.2 (StataCorp, College station, TX, USA). A two-sided P<0.05 was considered to indicate statistical significance.
METHODS
- Baseline clinical characteristics
- At baseline, study participants had a mean age of 51.6 years, 51.5% were men and they had a mean BMI of 24.2 kg/m2 (Table 1). Over a 10-year follow-up period, 88 of the 399 participants (22.1%) developed T2DM. Those developing incident T2DM were older, had higher BMI, fasting plasma glucose, HOMA-IR, QUICKI, total abdominal fat area, and a higher prevalence of a family history of diabetes compared to those who remained nondiabetic.
- The association between thigh muscle area and insulin resistance
- Table 2 lists the association of baseline thigh muscle area with HOMA-IR and QUICKI at 10-year follow-up. Thigh muscle area was inversely associated with future HOMA-IR after adjustment for age, sex, BMI, and baseline HOMA-IR (Model 1). This association persisted after further adjustment for fasting plasma glucose, total abdominal fat area, and thigh subcutaneous fat area (Model 2). In addition, thigh muscle area was independently associated with QUICKI as another insulin sensitivity surrogate index.
- The association between thigh muscle area and incident T2DM
- When we investigated the association between thigh muscle area and incident T2DM in multivariate logistic regression analysis, there was no significant association between thigh muscle area and incident T2DM after adjustment for age, sex, BMI, family history of diabetes, and HOMA-IR. However, when we examined an interaction between BMI and thigh muscle area using the first-order interaction terms inserted into the same logistic model, there was a significant positive interaction between BMI and estimated thigh muscle area (interaction term coefficient 0.003, P=0.022), with the coefficient for thigh muscle having a negative sign and being statistically significant (−0.071; P=0.030) (Table 3, model 1). In addition, interaction between BMI and thigh muscle area remained significant after insertion of fasting plasma glucose, total abdominal fat area, and thigh subcutaneous fat area as additional covariates into the model of Table 3 (model 2). There was no significant interaction between sex and thigh muscle (interaction term coefficient, −0.024; P=0.159) when these interactions were inserted into the logistic model in Table 3.
- In Fig. 1, the calculated probability of incident T2DM from the logistic regression model from Table 3 that includes the thigh muscle×BMI interaction is shown across the range of thigh muscle area for three values of BMI, demonstrating that the protective effect exists for lower levels of BMI but disappears in the obese range (BMI=30 kg/m2).
RESULTS
- These prospective data demonstrate that baseline thigh muscle mass area was inversely associated with future insulin resistance. We also observed an interaction between thigh muscle area and BMI in relation to T2DM. Namely, greater thigh muscle area was associated with lower risk of incident T2DM over 10 years for leaner subjects independent of age, sex, family history of diabetes, HOMA-IR, fasting plasma glucose, total abdominal fat area, and thigh subcutaneous fat area at baseline. We observed an inverse association between thigh muscle area and HOMA-IR change over 10 years that did not significantly vary by BMI. Thus greater muscle area was associated with less insulin resistance after 10 years but not a lower risk of T2DM in the presence of greater general adiposity. The potential benefit of less insulin resistance in association with greater muscle area may be overridden by higher general adiposity with regard to future T2DM risk.
- Previous studies reported that lower body muscle such as leg muscle was more associated with insulin resistance than was upper body muscle [2223]. Furthermore, the rate of loss of lower limb muscle was more than twice the rate of loss of upper limb muscle with aging [22425]. Considering these characteristics and their large muscle content, preservation of leg muscles should be considered as a main target in the strategy to prevent insulin resistance and incident T2DM. However, there has been little prospective research on this topic.
- We found that low thigh muscle area is independently associated with greater insulin resistance after 10 years in relatively healthy middle-aged and older Japanese Americans. To our knowledge, this is the first longitudinal study showing the long term association between thigh muscle mass and future insulin resistance. These findings support a previous cross-sectional study that demonstrated an inverse association between muscle mass and insulin resistance [4].
- The association between baseline thigh muscle area and incident T2DM was modified by BMI in our cohort; that is, greater thigh muscle area predicted lower risk of incident T2DM among leaner participants. The association between thigh muscle area and T2DM risk was diminished at higher levels of BMI. Our findings thus support earlier findings from cross-sectional studies. Although measurement of lower limb circumferences is a simple method to reflect both leg muscle and fat mass, there was also a strong interaction between thigh circumference and obesity in relation to diabetes prevalence in a large-scale, cross-sectional study [26]. Smaller thigh circumference was associated with diabetes, but this association was stronger among non-obese individuals. This study, though, could not convincingly demonstrate an association between thigh muscle and diabetes risk due to its cross-sectional design, which prevents ascertainment of temporal sequence, and also its measurement of thigh circumference which reflects a combination of thigh fat and muscle mass. In another cross-sectional study of postmenopausal women that also suggested an association between greater muscle mass and lower T2DM prevalence, abdominal muscle area was inversely related with T2DM prevalence, especially for women with a BMI <25 kg/m2 [5].
- To our knowledge the current study extends findings of the only other investigation that assessed risk of incident T2DM by regional muscle mass area using imaging (Health ABC Study), and which was conducted in an elderly Caucasian and African American population [10]. Ethnicity is an important determinant of body composition, with Asians known to have less muscle mass and more body fat compared to Western populations at the same BMI [27]. These characteristics are related with a higher risk of insulin resistance and T2DM [2829]. In addition, our population with a mean age of 51.6 years was relatively younger than those of the Health ABC Study, so our findings reflect the impact of a relatively higher range of muscle mass on the development of T2DM before advanced age-related sarcopenia. In addition, we used the OGTT along with a history of clinical diagnosis for identifying incident T2DM, and therefore were more likely to detect its occurrence as compared to the Health ABC Study that relied on fasting glucose and clinical history only [30].
- We previously found that BMI acts as an effect modifier in the relationship between hand grip strength and incident T2DM in Japanese Americans, i.e., greater hand grip strength predicts a lower risk of incident T2DM among normal weight participants [31]. Combining this finding in the same cohort with the finding from the current analysis demonstrating a lower risk of T2DM with greater thigh muscle area in normal weight participants, it appears that obesity modifies the associations between muscle mass or muscle strength and incident T2DM in a similar pattern in our population. Based on these findings, we can assume that interventions aimed at increasing muscle mass and strength in lean individuals may be more effective to reduce the risk of T2DM than in overweight or obese individuals.
- The underlying mechanism for the diminution of the inverse relationship between baseline thigh muscle area and T2DM risk at higher levels of BMI is not known. A possible reason is thigh muscle mass may have a relatively smaller impact on the development of T2DM in obese subjects due to preexisting metabolic imbalance caused by obesity. Therefore, the potential protective effects of greater muscle mass are overcome by much stronger effects of adiposity on T2DM risk. Our finding of a significant inverse association between thigh muscle area and future insulin resistance that did not vary by BMI further supports this theory, as this means that the potential mechanism for higher T2DM risk (i.e., greater insulin resistance) affects lean and non-lean similarly, and therefore must be overcome by other factors in the non-lean that prevent expression of this lower risk.
- We noted that greater thigh subcutaneous fat area was associated with higher insulin sensitivity at 10 years. This finding might therefore be expected to result in lower risk of developing T2DM. However, we did not identify an association between thigh subcutaneous fat area and risk of T2DM in a previous analysis of these same data [32].
- As for the strengths of our study, study participants were from a well-designed community-based cohort with a long-term follow-up period. This prospective study design permitted assessment of thigh muscle area prior to the outcome assessment, thereby helping to establish temporal sequence not possible in cross-sectional research. Additional strengths include use of imaging to measure muscle areas, and also our focus on regional muscle as opposed the generalized lean mass.
- There are several limitations to be considered in this study. First, we estimated thigh muscle mass instead of directly measuring thigh muscle area using imaging. However, this method has been previously validated [21]. Second, we used HOMA-IR and QUICKI as a surrogate for insulin sensitivity. Although not the gold standard method, it is a frequently used tool for assessing insulin sensitivity in large population based studies [1833]. Finally, as this study was restricted to middle-aged and older Japanese Americans, caution should be taken when generalizing the results to other age or ethnic groups.
- In conclusion, we found that thigh muscle area was inversely associated with future insulin resistance over 10 years in Japanese Americans. In addition, greater thigh muscle area was associated with lower risk of incident T2DM for leaner subjects independent of insulin resistance. These results suggest an important role for muscle mass in the development of T2DM that varies by degree of adiposity, and might lead to research to improve our understanding of the roles of both lean and non-lean body composition in the pathogenesis of hyperglycemia. Further intervention studies are needed to confirm whether increasing muscle mass can reduce the incidence of T2DM, and whether such effects vary according to BMI.
DISCUSSION
-
Acknowledgements
- National Institutes of Health grants DK-31170 and HL-49293. This work was supported by facilities and services provided by the Diabetes Research Center (DK-017047), Clinical Nutrition Research Unit (DK-035816), and the General Clinical Research Center (RR-000037) at the University of Washington. The funding entities had no role in the conduct of this study or interpretation of its results.
- We are grateful to the King County Japanese-American community for support and cooperation. VA Puget Sound Health Care System provided support for Drs. Boyko and Kahn's involvement in this research.
ACKNOWLEDGMENTS
-
CONFLICTS OF INTEREST: No potential conflict of interest relevant to this article was reported.
NOTES
- 1. Guariguata L, Whiting DR, Hambleton I, Beagley J, Linnenkamp U, Shaw JE. Global estimates of diabetes prevalence for 2013 and projections for 2035. Diabetes Res Clin Pract 2014;103:137-149. ArticlePubMed
- 2. Janssen I, Heymsfield SB, Wang ZM, Ross R. Skeletal muscle mass and distribution in 468 men and women aged 18-88 yr. J Appl Physiol (1985) 2000;89:81-88. ArticlePubMed
- 3. DeFronzo RA, Tripathy D. Skeletal muscle insulin resistance is the primary defect in type 2 diabetes. Diabetes Care 2009;32(Suppl 2):S157-S163. ArticlePubMedPMCPDF
- 4. Srikanthan P, Karlamangla AS. Relative muscle mass is inversely associated with insulin resistance and prediabetes. Findings from the third National Health and Nutrition Examination Survey. J Clin Endocrinol Metab 2011;96:2898-2903. ArticlePubMed
- 5. Larsen BA, Allison MA, Laughlin GA, Araneta MR, Barrett-Connor E, Wooten WJ, Saad SD, Wassel CL. The association between abdominal muscle and type II diabetes across weight categories in diverse post-menopausal women. J Clin Endocrinol Metab 2015;100:E105-E109. ArticlePubMed
- 6. Atlantis E, Martin SA, Haren MT, Taylor AW, Wittert GA. Members of the Florey Adelaide Male Ageing Study. Inverse associations between muscle mass, strength, and the metabolic syndrome. Metabolism 2009;58:1013-1022. ArticlePubMed
- 7. Volpato S, Bianchi L, Lauretani F, Lauretani F, Bandinelli S, Guralnik JM, Zuliani G, Ferrucci L. Role of muscle mass and muscle quality in the association between diabetes and gait speed. Diabetes Care 2012;35:1672-1679. ArticlePubMedPMCPDF
- 8. Eastwood SV, Tillin T, Wright A, Mayet J, Godsland I, Forouhi NG, Whincup P, Hughes AD, Chaturvedi N. Thigh fat and muscle each contribute to excess cardiometabolic risk in South Asians, independent of visceral adipose tissue. Obesity (Silver Spring) 2014;22:2071-2079. PubMedPMC
- 9. Barzilay JI, Cotsonis GA, Walston J, Schwartz AV, Satterfield S, Miljkovic I, Harris TB. Health ABC Study. Insulin resistance is associated with decreased quadriceps muscle strength in nondiabetic adults aged >or=70 years. Diabetes Care 2009;32:736-738. PubMedPMC
- 10. Li JJ, Wittert GA, Vincent A, Atlantis E, Shi Z, Appleton SL, Hill CL, Jenkins AJ, Januszewski AS, Adams RJ. Muscle grip strength predicts incident type 2 diabetes: population-based cohort study. Metabolism 2016;65:883-892. ArticlePubMed
- 11. Larsen BA, Wassel CL, Kritchevsky SB, Strotmeyer ES, Criqui MH, Kanaya AM, Fried LF, Schwartz AV, Harris TB, Ix JH. Health ABC Study. Association of muscle mass, area, and strength with incident diabetes in older adults: The Health ABC Study. J Clin Endocrinol Metab 2016;101:1847-1855. ArticlePubMedPMCPDF
- 12. Son JW, Lee SS, Kim SR, Yoo SJ, Cha BY, Son HY, Cho NH. Low muscle mass and risk of type 2 diabetes in middle-aged and older adults: findings from the KoGES. Diabetologia 2017;60:865-872. ArticlePubMedPDF
- 13. Dehghan M, Merchant AT. Is bioelectrical impedance accurate for use in large epidemiological studies? Nutr J 2008;7:26ArticlePubMedPMCPDF
- 14. Fujimoto WY, Leonetti DL, Kinyoun JL, Newell-Morris L, Shuman WP, Stolov WC, Wahl PW. Prevalence of diabetes mellitus and impaired glucose tolerance among second-generation Japanese-American men. Diabetes 1987;36:721-729. ArticlePubMed
- 15. Koppes LL, Dekker JM, Hendriks HF, Bouter LM, Heine RJ. Moderate alcohol consumption lowers the risk of type 2 diabetes: a meta-analysis of prospective observational studies. Diabetes Care 2005;28:719-725. PubMed
- 16. Paffenbarger RS Jr, Wing AL, Hyde RT. Physical activity as an index of heart attack risk in college alumni. Am J Epidemiol 1978;108:161-175. ArticlePubMed
- 17. Matthews DR, Hosker JP, Rudenski AS, Naylor BA, Treacher DF, Turner RC. Homeostasis model assessment: insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia 1985;28:412-419. ArticlePubMedPDF
- 18. Katz A, Nambi SS, Mather K, Baron AD, Follmann DA, Sullivan G, Quon MJ. Quantitative insulin sensitivity check index: a simple, accurate method for assessing insulin sensitivity in humans. J Clin Endocrinol Metab 2000;85:2402-2410. ArticlePubMedPDF
- 19. Report of the expert committee on the diagnosis and classification of diabetes mellitus. Diabetes Care 1997;20:1183-1197. ArticlePubMedPDF
- 20. Shuman WP, Morris LL, Leonetti DL, Wahl PW, Moceri VM, Moss AA, Fujimoto WY. Abnormal body fat distribution detected by computed tomography in diabetic men. Invest Radiol 1986;21:483-487. ArticlePubMed
- 21. Han SJ, Kim SK, Fujimoto WY, Kahn SE, Leonetti DL, Boyko EJ. Effects of combination of change in visceral fat and thigh muscle mass on the development of type 2 diabetes. Diabetes Res Clin Pract 2017;134:131-138. ArticlePubMedPMC
- 22. Olsen DB, Sacchetti M, Dela F, Ploug T, Saltin B. Glucose clearance is higher in arm than leg muscle in type 2 diabetes. J Physiol 2005;565(Pt 2):555-562. ArticlePubMedPMC
- 23. Sacchetti M, Olsen DB, Saltin B, van Hall G. Heterogeneity in limb fatty acid kinetics in type 2 diabetes. Diabetologia 2005;48:938-945. ArticlePubMedPDF
- 24. Borkan GA, Hults DE, Gerzof SG, Robbins AH, Silbert CK. Age changes in body composition revealed by computed tomography. J Gerontol 1983;38:673-677. ArticlePubMed
- 25. Gallagher D, Visser M, De Meersman RE, Sepulveda D, Baumgartner RN, Pierson RN, Harris T, Heymsfield SB. Appendicular skeletal muscle mass: effects of age, gender, and ethnicity. J Appl Physiol (1985) 1997;83:229-239. ArticlePubMed
- 26. Jung KJ, Kimm H, Yun JE, Jee SH. Thigh circumference and diabetes: obesity as a potential effect modifier. J Epidemiol 2013;23:329-336. ArticlePubMedPMC
- 27. Nakagami T, Qiao Q, Carstensen B, Nhr-Hansen C, Hu G, Tuomilehto J, Balkau B, Borch-Johnsen K. DECODE-DECODA Study Group. Age, body mass index and type 2 diabetes-associations modified by ethnicity. Diabetologia 2003;46:1063-1070. ArticlePubMedPDF
- 28. Kim TN, Park MS, Lim KI, Choi HY, Yang SJ, Yoo HJ, Kang HJ, Song W, Choi H, Baik SH, Choi DS, Choi KM. Relationships between sarcopenic obesity and insulin resistance, inflammation, and vitamin D status: the Korean Sarcopenic Obesity Study. Clin Endocrinol (Oxf) 2013;78:525-532. ArticlePubMed
- 29. Kim TN, Choi KM. The implications of sarcopenia and sarcopenic obesity on cardiometabolic disease. J Cell Biochem 2015;116:1171-1178. ArticlePubMed
- 30. Han SJ, Kim HJ, Kim DJ, Lee KW, Cho NH. Incidence and predictors of type 2 diabetes among Koreans: a 12-year follow up of the Korean Genome and Epidemiology Study. Diabetes Res Clin Pract 2017;123:173-180. ArticlePubMed
- 31. Wander PL, Boyko EJ, Leonetti DL, McNeely MJ, Kahn SE, Fujimoto WY. Greater hand-grip strength predicts a lower risk of developing type 2 diabetes over 10 years in leaner Japanese Americans. Diabetes Res Clin Pract 2011;92:261-264. ArticlePubMedPMC
- 32. Hoyer D, Boyko EJ, McNeely MJ, Leonetti DL, Kahn SE, Fujimoto WY. Subcutaneous thigh fat area is unrelated to risk of type 2 diabetes in a prospective study of Japanese Americans. Diabetologia 2011;54:2795-2800. ArticlePubMedPMCPDF
- 33. Wallace TM, Levy JC, Matthews DR. Use and abuse of HOMA modeling. Diabetes Care 2004;27:1487-1495. ArticlePubMedPDF
REFERENCES
Adjusted marginal plots showing body mass index (BMI) interaction effects on the association between thigh muscle area and probability of 10-year incident type 2 diabetes mellitus. Probabilities of type 2 diabetes mellitus are shown for subjects with BMI 20, 25, 30 kg/m2 after adjusting for age, sex, family history of diabetes, and homeostasis model assessment of insulin resistance.
Clinical characteristics of study subjects by incident type 2 diabetes mellitus status at 10-year follow-up
Values are presented as mean±standard deviation or number (%).
HOMA-IR, homeostasis model assessment of insulin resistance; QUICKI, quantitative insulin sensitivity check index.
aLog-transformed values were used for statistical comparisons. P values represent the difference between groups for each variable using independent t-tests and chi-square tests, as appropriate.
Multivariate linear regression analysis of the prediction of HOMA-IR and QUICKI at 10-year follow-up
Model 1: adjusted for baseline traits (age, sex, BMI, thigh muscle area, and HOMA-IR); Model 2: model 1+fasting plasma glucose, total abdominal fat area, and thigh subcutaneous fat area.
HOMA-IR, homeostasis model assessment of insulin resistance; QUICKI, quantitative insulin sensitivity check index; BMI, body mass index.
aData are expressed as standardized β.
Multivariate logistic regression analysis of the prediction of incident type 2 diabetes mellitus at 10-year follow-up
Figure & Data
References
Citations

- More appendicular lean mass relative to body mass index is associated with lower incident diabetes in middle-aged adults in the CARDIA study
Melanie S. Haines, Aaron Leong, Bianca C. Porneala, Victor W. Zhong, Cora E. Lewis, Pamela J. Schreiner, Karen K. Miller, James B. Meigs, Mercedes R. Carnethon
Nutrition, Metabolism and Cardiovascular Diseases.2023; 33(1): 105. CrossRef - Shape phenotype of thigh fat and muscle and risk of major adverse cardiovascular events after fragility hip fracture
Sheen‐Woo Lee, Seung‐Chan Kim, Jeong‐Eun Yi
Journal of Cachexia, Sarcopenia and Muscle.2023;[Epub] CrossRef - Involvement of the fecal amino acid profile in a clinical and anthropometric study of Mexican patients with insulin resistance and type 2 diabetes mellitus
Mayra Paloma Macías-Acosta, Lorena Valerdi-Contreras, Ericka Denise Bustos-Angel, Rudy Antonio García-Reyes, Monserrat Alvarez-Zavala, Marisela González-Ávila
Amino Acids.2022; 54(1): 47. CrossRef - Association of serum creatinine levels and risk of type 2 diabetes mellitus in Korea: a case control study
Do Kyeong Song, Young Sun Hong, Yeon-Ah Sung, Hyejin Lee
BMC Endocrine Disorders.2022;[Epub] CrossRef - The Relationship Between Fat-Free Mass and Glucose Metabolism in Children and Adolescents: A Systematic Review and Meta-Analysis
Lijun Wu, Fangfang Chen, Junting Liu, Dongqing Hou, Tao Li, Yiren Chen, Zijun Liao
Frontiers in Pediatrics.2022;[Epub] CrossRef - Is imaging-based muscle quantity associated with risk of diabetes? A meta-analysis of cohort studies
Shanhu Qiu, Xue Cai, Yang Yuan, Bo Xie, Zilin Sun, Tongzhi Wu
Diabetes Research and Clinical Practice.2022; 189: 109939. CrossRef - Effects of Exercise Intervention on Type 2 Diabetes Patients With Abdominal Obesity and Low Thigh Circumference (EXTEND): Study Protocol for a Randomized Controlled Trial
Dan Liu, Ying Zhang, Liang Wu, Jingyi Guo, Xiangtian Yu, Huasheng Yao, Rui Han, Tianshu Ma, Yuchan Zheng, Qiongmei Gao, Qichen Fang, Yan Zhao, Yanan Zhao, Biao Sun, Weiping Jia, Huating Li
Frontiers in Endocrinology.2022;[Epub] CrossRef - Association between diabetic peripheral neuropathy and sarcopenia: A systematic review and meta‐analysis
Thapat Wannarong, Persen Sukpornchairak, Weerakit Naweera, Christopher D. Geiger, Patompong Ungprasert
Geriatrics & Gerontology International.2022; 22(9): 785. CrossRef - Synergistic Interaction between Hyperuricemia and Abdominal Obesity as a Risk Factor for Metabolic Syndrome Components in Korean Population
Min Jin Lee, Ah Reum Khang, Yang Ho Kang, Mi Sook Yun, Dongwon Yi
Diabetes & Metabolism Journal.2022; 46(5): 756. CrossRef - HOMA-IR as a predictor of Health Outcomes in Patients with Metabolic Risk Factors: A Systematic Review and Meta-analysis
José G. González-González, Jorge R. Violante-Cumpa, Miguel Zambrano-Lucio, Erick Burciaga-Jimenez, Patricia L. Castillo-Morales, Mariano Garcia-Campa, Ricardo César Solis, Alejandro D González-Colmenero, René Rodríguez-Gutiérrez
High Blood Pressure & Cardiovascular Prevention.2022; 29(6): 547. CrossRef - Maternal height as a predictor of glucose intolerance in the postpartum and its relationship with maternal pre-gestational weight
Catarina Cidade-Rodrigues, Filipe M. Cunha, Catarina Chaves, Fabiana Castro, Catarina Pereira, Sílvia Paredes, Margarida Silva-Vieira, Anabela Melo, Odete Figueiredo, Cláudia Nogueira, Ana Morgado, Mariana Martinho, Maria C. Almeida, Margarida Almeida
Archives of Gynecology and Obstetrics.2022; 307(2): 601. CrossRef - Correlation of body composition in early pregnancy on gestational diabetes mellitus under different body weights before pregnancy
Li Xintong, Xu Dongmei, Zhang Li, Cao Ruimin, Hao Yide, Cui Lingling, Chen Tingting, Guo Yingying, Li Jiaxin
Frontiers in Endocrinology.2022;[Epub] CrossRef - Relationship between low skeletal muscle mass, sarcopenic obesity and left ventricular diastolic dysfunction in Korean adults
Jee Hee Yoo, Sung Woon Park, Ji Eun Jun, Sang‐Man Jin, Kyu Yeon Hur, Moon‐Kyu Lee, Mira Kang, Gyuri Kim, Jae Hyeon Kim
Diabetes/Metabolism Research and Reviews.2021;[Epub] CrossRef - Skeletal Muscle and Metabolic Health: How Do We Increase Muscle Mass and Function in People with Type 2 Diabetes?
Ebaa Al-Ozairi, Dalal Alsaeed, Dherar Alroudhan, Nia Voase, Amal Hasan, Jason M R Gill, Naveed Sattar, Paul Welsh, Cindy M Gray, Jirapitcha Boonpor, Carlos Celis-Morales, Stuart R Gray
The Journal of Clinical Endocrinology & Metabolism.2021; 106(2): 309. CrossRef - Association of the triglyceride and glucose index with low muscle mass: KNHANES 2008–2011
Jung A. Kim, Soon Young Hwang, Ji Hee Yu, Eun Roh, So-hyeon Hong, You-Bin Lee, Nam Hoon Kim, Hye Jin Yoo, Ji A. Seo, Nan Hee Kim, Sin Gon Kim, Sei Hyun Baik, Kyung Mook Choi
Scientific Reports.2021;[Epub] CrossRef - ADAS-viewer: web-based application for integrative analysis of multi-omics data in Alzheimer’s disease
Seonggyun Han, Jaehang Shin, Hyeim Jung, Jane Ryu, Habtamu Minassie, Kwangsik Nho, Insong Koh, Younghee Lee
npj Systems Biology and Applications.2021;[Epub] CrossRef - Relative associations of abdominal and thigh compositions with cardiometabolic diseases in African Caribbean men
Curtis Tilves, Joseph M. Zmuda, Allison L. Kuipers, Sangeeta Nair, John Jeffrey Carr, James G. Terry, Shyamal Peddada, Victor Wheeler, Iva Miljkovic
Obesity Science & Practice.2021; 7(6): 738. CrossRef - Changes in creatinine‐to‐cystatin C ratio over 4 years, risk of diabetes, and cardiometabolic control: The China Health and Retirement Longitudinal Study
Shanhu Qiu, Xue Cai, Yang Yuan, Bo Xie, Zilin Sun, Tongzhi Wu
Journal of Diabetes.2021; 13(12): 1025. CrossRef - Associations Between Glucose Tolerance, Insulin Secretion, Muscle and Fat Mass in Cystic Fibrosis
Bibi Uhre Nielsen, Daniel Faurholt-Jepsen, Peter Sandor Oturai, Tavs Qvist, Rikke Krogh-Madsen, Terese Lea Katzenstein, James Shaw, Christian Ritz, Tacjana Pressler, Thomas Peter Almdal, Inger Hee Mabuza Mathiesen
Clinical Medicine Insights: Endocrinology and Diabetes.2021; 14: 117955142110382. CrossRef - Total and regional fat‐to‐muscle mass ratio measured by bioelectrical impedance and risk of incident type 2 diabetes
Ningjian Wang, Ying Sun, Haojie Zhang, Chi Chen, Yuying Wang, Jihui Zhang, Fangzhen Xia, Christian Benedict, Xiao Tan, Yingli Lu
Journal of Cachexia, Sarcopenia and Muscle.2021; 12(6): 2154. CrossRef - How was the Diabetes Metabolism Journal added to MEDLINE?
Hye Jin Yoo
Science Editing.2020; 7(2): 201. CrossRef - An update on nutrient modulation in the management of disease-induced muscle wasting: evidence from human studies
Matthew S. Brook, Daniel J. Wilkinson, Philip J. Atherton
Current Opinion in Clinical Nutrition & Metabolic Care.2020; 23(3): 174. CrossRef - The functional nutritional and regulatory activities of calcium supplementation from eggshell for obesity disorders management
Marwa El‐Zeftawy, Samar Abd‐El Mohsen Ali, Sally Salah, Hani S. Hafez
Journal of Food Biochemistry.2020;[Epub] CrossRef - Dose‐response associations between serum creatinine and type 2 diabetes mellitus risk: A Chinese cohort study and meta‐analysis of cohort studies
Pei Qin, Yanmei Lou, Liming Cao, Jing Shi, Gang Tian, Dechen Liu, Qionggui Zhou, Chunmei Guo, Quanman Li, Yang Zhao, Feiyan Liu, Xiaoyan Wu, Ranran Qie, Minghui Han, Shengbing Huang, Ping Zhao, Changyi Wang, Jianping Ma, Xiaolin Peng, Shan Xu, Hongen Chen
Journal of Diabetes.2020; 12(8): 594. CrossRef - Caloric restriction in heart failure: A systematic review
Vittorio Emanuele Bianchi
Clinical Nutrition ESPEN.2020; 38: 50. CrossRef - Association between the Thigh Muscle and Insulin Resistance According to Body Mass Index in Middle-Aged Korean Adults
Ji Eun Heo, Jee-Seon Shim, Hokyou Lee, Hyeon Chang Kim
Diabetes & Metabolism Journal.2020; 44(3): 446. CrossRef - Joint impact of muscle mass and waist circumference on type 2 diabetes in Japanese middle‐aged adults: The Circulatory Risk in Communities Study (CIRCS)
Mikako Yasuoka, Isao Muraki, Hironori Imano, Hiroshige Jinnouchi, Yasuhiko Kubota, Mina Hayama‐Terada, Mitsumasa Umesawa, Kazumasa Yamagishi, Tetsuya Ohira, Akihiko Kitamura, Takeo Okada, Masahiko Kiyama, Hiroyasu Iso
Journal of Diabetes.2020; 12(9): 677. CrossRef - Catch-Up Growth in Children Born Small for Gestational Age Related to Body Composition and Metabolic Risk at Six Years of Age in the UK
M. Loredana Marcovecchio, Samantha Gorman, Laura P.E. Watson, David B. Dunger, Kathryn Beardsall
Hormone Research in Paediatrics.2020; 93(2): 119. CrossRef - Response: Association of Thigh Muscle Mass with Insulin Resistance and Incident Type 2 Diabetes Mellitus in Japanese Americans (Diabetes Metab J 2018;42:488–95)
Seung Jin Han, Edward J. Boyko
Diabetes & Metabolism Journal.2019; 43(1): 125. CrossRef - Berberine chloride ameliorated PI3K/Akt‐p/SIRT‐1/PTEN signaling pathway in insulin resistance syndrome induced in rats
Marwa El‐Zeftawy, Doaa Ghareeb, Eman R. ElBealy, Rasha Saad, Salma Mahmoud, Nihal Elguindy, Attalla F. El‐kott, Mohamed El‐Sayed
Journal of Food Biochemistry.2019;[Epub] CrossRef - Letter: Association of Thigh Muscle Mass with Insulin Resistance and Incident Type 2 Diabetes Mellitus in Japanese Americans (Diabetes Metab J 2018;42:488–95)
Nan Hee Cho, Hye Soon Kim
Diabetes & Metabolism Journal.2019; 43(1): 123. CrossRef - Asian Indians With Prediabetes Have Similar Skeletal Muscle Mass and Function to Those With Type 2 Diabetes
Sucharita Sambashivaiah, Stephen D. R. Harridge, Nidhi Sharma, Sumithra Selvam, Priyanka Rohatgi, Anura V. Kurpad
Frontiers in Nutrition.2019;[Epub] CrossRef

 KDA
KDA PubReader
PubReader Cite
Cite