- Current
- Browse
- Collections
-
For contributors
- For Authors
- Instructions to authors
- Article processing charge
- e-submission
- For Reviewers
- Instructions for reviewers
- How to become a reviewer
- Best reviewers
- For Readers
- Readership
- Subscription
- Permission guidelines
- About
- Editorial policy
Articles
- Page Path
- HOME > Diabetes Metab J > Volume 40(1); 2016 > Article
-
Original ArticlePathophysiology Protective Effects of Ginger (
Zingiber officinale ) Extract against Diabetes-Induced Heart Abnormality in Rats -
Behrouz Ilkhanizadeh1, Alireza Shirpoor2
 , Mohamad hasan Khadem Ansari3, Samira Nemati2, Yusef Rasmi3
, Mohamad hasan Khadem Ansari3, Samira Nemati2, Yusef Rasmi3 -
Diabetes & Metabolism Journal 2016;40(1):46-53.
DOI: https://doi.org/10.4093/dmj.2016.40.1.46
Published online: February 19, 2016
1Department of Pathology, Urmia University of Medical Sciences Faculty of Medicine, Urmia, Iran.
2Department of Physiology, Urmia University of Medical Sciences Faculty of Medicine, Urmia, Iran.
3Department of Biochemistry, Urmia University of Medical Sciences Faculty of Medicine, Urmia, Iran.
- Corresponding author: Alireza Shirpoor. Department of Physiology, Urmia University of Medical Sciences Faculty of Medicine, Urmia, Iran. ashirpoor@yahoo.com
• Received: August 2, 2015 • Accepted: October 12, 2015
Copyright © 2016 Korean Diabetes Association
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
ABSTRACT
-
Background
- Diabetic cardiomyopathy is an important causal factor in morbidity and mortality among diabetic patients, and currently, no effective means are available to reverse its pathological progress. The purpose of the present study was to investigate the effect of ginger extract on apolipoproteins (apo) A and B, hyperhomocysteinemia, cathepsin G and leptin changes, as well as cardiac fibrosis and heart muscle cell proliferation under hyperglycemic conditions in vivo.
-
Methods
- Twenty-four male Wistar rats were divided into three groups, namely: control, non-treated diabetic, and ginger extract-treated diabetic groups. The ginger extract-treated diabetic group received a 50 mg daily dose of ginger extract intragastrically for 6 weeks.
-
Results
- The results revealed concurrent significant increases in plasma C-reactive protein (CRP), homocysteine (Hcy), cathepsin G and apoB levels and decreases in apoA and leptin levels in the non-treated diabetic group compared to the control group. Moreover, heart structural changes, including fibrosis and heart muscle cell proliferation, were observed in non-treated diabetic rats compared to the control rats. Significant amelioration of changes in the heart structure together with restoration of the elevated levels of Hcy and CRP, leptin, cathepsin G, and apoA and B were found in the ginger extract-treated diabetic group compared to the non-treated diabetic group.
-
Conclusion
- The findings indicated that ginger extract significantly reduces heart structural abnormalities in diabetic rats and that these effects might be associated with improvements in serum apo, leptin, cathepsin G, and Hcy levels and with the antioxidant properties of ginger extract.
- Cardiovascular complications resulting from diabetes are the main cause of death in diabetic patients; indeed, more than 65% of deaths among diabetic patients are associated with heart failure and vascular abnormalities [1]. Histological changes including apoptosis, fibrosis, hypertrophy, autonomic neuropathy, alterations in Ca2+ transport, as well as intracellular Ca2+ homeostasis, which are induced in the heart by diabetes, have been documented in recent articles [234]. Compelling evidence indicates that diabetes causes heart abnormalities through oxidative stress and inflammation. Certain markers of inflammation and oxidative stress such as C-reactive protein (CRP), interleukin 6 (IL-6), tumor necrosis factor α (TNF-α), oxidized low density lipoprotein (LDL), and protein oxidation, as well as homocysteine (Hcy), have repeatedly been associated with diabetes-related cardiomyopathy in different settings [5]. Nevertheless, few studies have examined possible associations between other parts of the inflammatory process (such as cathepsin G and leptin) and heart structural changes (such as fibrosis and heart muscle cell proliferation) under hyperglycemic conditions and diabetes. Cathepsin G is best known as a neutrophil and mast cell-derived serine protease, and its role in the progression of heart failure has recently become more evident [67]. Recently, the adipocyte-derived leptin has attracted attention due to its ambiguous role in cardiovascular function. Many studies have demonstrated that elevated leptin levels are significantly correlated with heart failure and myocardial infarction, probably through the increased production of proinflammatory cytokines and oxidative stress [89]. Other studies have found that low leptin levels (as occur in genetic models of leptin deficiency) increase the incidence of hypertension, atherosclerosis, and myocardial infarction [1011]. Accordingly, the relationship between leptin levels and the incidence of cardiovascular system dysfunction appears biphasic and complex. Currently, daily insulin administration is the main intervention strategy for decreasing mortality in diabetic patients. Insulin therapy reduces hyperglycemia and glycosylated hemoglobin, and delays some diabetes-related morbidity. However, even with insulin therapy, some secondary complications associated with diabetes including heart disease, neuropathy, and hypertension remain overwhelming conditions in diabetic patients [12]. In addition, the lipogenic and cholesterologenic actions of insulin therapy on the one hand and the increased risk of hypoglycemia on the other lead to devastating and fatal results in patients under long-term insulin treatment [13]. Furthermore, the effect of insulin therapy on serum leptin is biphasic and complex. It has been reported that acute insulin treatment increases the serum level of leptin in rats and humans [14]. Contradictorily, it has also been reported that acute insulin exposure does not regulate leptin levels in human subjects [1516]. For these reasons, improved alternative anti-diabetic approaches are urgently needed. In addition, naturally derived anti-oxidants appear more favorable for reducing diabetes-induced side effects in diabetic patients. Among plants containing natural anti-oxidants, ginger exhibits unique antioxidant, and anti-inflammatory properties [17].
- The aim of the present study was to examine the protective effect of ginger on heart structural changes induced by diabetes in which cathepsin G and leptin play a central role. The study also examined whether treatment with ginger can restore altered oxidative and inflammatory stress agents in the heart of STZ-induced diabetic rats.
INTRODUCTION
- All procedures utilizing rats were performed according to the "Principles of Laboratory Animal Care" (NIH publication no. 85–23, revised 1985) and were approved by the Urmia University of Medical Sciences Animal Care Committee. Twenty-four 6-month-old male Wistar rats with initial body weights of approximately 220±20 g and normal blood glucose levels (145±5 mg/dL) were included in this study. The rats were divided into three groups (eight animals in each): control (C), non-treated diabetic (NTD), and ginger extract-treated diabetic (GETD) groups. Diabetes was induced in 16 rats by a single intraperitoneal injection of STZ (60 mg/kg), which was buffered in cold sodium citrate (pH 4.5). Forty-eight hours after the STZ injection, hyperglycemia was determined by measuring the tail vein blood glucose content using a glucose oxidase-based Biosystem kit (Biosystems, Barcelona, Spain). Rats with blood glucose of higher than 300 mg/dL were considered diabetic.
- In addition to their regular diet, GETD rats daily received 50 mg/kg body weight of hydroalcoholic ginger extract solution in tap water (20% w/v) intragastrically by gavage. The control and NTD groups were treated with vehicle only (tap water).
- For ginger extraction, dried ginger rhizome (origin: China) was purchased from a local market and powdered in an electric grinder. A hydroalcoholic extract was prepared by mixing 3 kg of powder with 6 L of 70% ethanol in a suitable container and incubating at room temperature for 72 hours. The extract was then filtered through filter paper and concentrated using a rotary evaporator. The obtained extract was stored in a refrigerator until use.
- The rats were anesthetized using 10% chloral hydrate (0.5 mL/kg body weight, IP) after 6 weeks of treatment, and the depth of anesthesia was assessed by pinching a hind paw. After weighing the rats, the thoracic cavity was opened, and blood samples were collected directly from the hearts; within 30 minutes of collection, the samples were mixed with ethylenediaminetetraacetic acid as an anticoagulant and then centrifuged at 4,000 ×g for 20 minutes. The plasma samples were stored at –80℃ without repeated freeze-thaw cycles. Next, the heart was dissected. The excised heart was freed from adventitial tissues, fat, and blood clots, and then weighed. Then, the entire left ventricular wall (without septum) was excised from the heart, weighed, and divided into two parts. For histopathological investigation, a part of the ventricle was fixed in buffered formalin and embedded in paraffin after standard dehydration methods.
- For biochemical analysis, other parts of the ventricle were washed with ice-cold physiological saline and then dried on filter papers. An ice-cold extraction buffer (10% wt/vol) containing 50 mM phosphate buffer (pH 7.4) was added, and the sample was subsequently homogenized using Ultra Turrax (T10B; IKA, Staufen, Germany). The homogenates were then centrifuged at 10,000 ×g for 20 minutes at 4℃. The supernatants were collected and stored at –80℃ until analyzed.
- Biochemical assay
- The concentration of cathepsin G in the rat heart was measured using a solid phase enzyme immunoassay method (Cathepsin G Kit; AESKULISA, Dresden, Germany) following the manufacturer's instructions. Hcy levels were measured using an Axis Shield kit (Axis Shield, Dundee, UK) following the manufacturer's recommended protocol.
- The level of CRP in the serum was assessed using nephelometric methods and MININEPHTM following the manufacturer's instructions (ZK044.L.R; The Binding Site Ltd., Birmingham, UK). Apolipoprotein (apo) A and B amounts were measured using the nephelometric method and the Mono Binding kit (Binding Site, Birmingham, UK) according to the manufacturer's guidelines. The amount of leptin was measured using a commercial enzyme immunoassay kit (Labor Diagnostika Nord GmbH, Nordhorn, Germany).
- Histopathological examination
- Histological sections (5-µm thick) from paraffin-embedded heart tissues were subjected to histopathological staining. Harris' H&E staining protocols were applied to evaluate the general structure of the heart and morphological changes.
- To assess the heart and coronary vessel proliferation rates, a proliferation cell nuclear antigen (PCNA) anti-body staining protocol was used in accordance with the included instructions. Briefly, tissue sections (4-µm thick) from paraffin-embedded heart tissues were stained by Monoclonal anti-PCNA antibody (Dako Denmark A/S, Glostrup, Denmark) after tissue processing including deparaffinization, rehydration by gradual ethanol passage, and final washing in Tris buffer.
- Optimal results were achieved using the EnVisio visualization system (Dako Denmark A/S). Hematoxylin was used as a counterstain. Appropriate negative controls were included in the assessment. Moreover, all slides were independently inspected by two expert pathologists. PCNA-positive indices were considered indicators of muscle cell proliferation.
- To assess the percentage of PCNA-positive indices, all cells contained in each cross section (between 3 and 5 cross sections) of the heart and the coronary artery were scored. The samples were scored based on the PCNA-positive indices as follows: normal, PCNA-positive indices present in less than 5% of muscle cells; mild, PCNA-positive indices present in less than 25% of muscle cells; mild to moderate, PCNA-positive indices present in 25% to 50% of muscle cells; moderate to severe, PCNA-positive indices present in 50% to 75% of muscle cells; and severe, PCNA-positive indices present in 75% to 100% of muscle cells. To evaluate the heart and coronary vessel fibrosis, 4-µm heart tissue sections were stained with Masson Trichrome, following the manufacturer's instructions (Asia Pajohesh, Amol, Iran). The severity of tissue fibrosis was assessed using the semi-quantitative method described by Ashcroft et al. [18]. A score ranging from 0 (normal heart) to 8 (total fibrosis) was allocated. Heart fibrosis was scored as follows: grade 0, normal heart; grade 1, minimal fibrosis thickening of the myocardial or coronary vessel walls; grades 2 and 3, moderate thickening of the myocardial or coronary vessels walls without obvious damage to the heart tissue structure; grades 4 and 5, increased fibrosis with definite damage to the heart architecture and the formation of fibrosis bands or small fibrosis masses; grades 6 and 7, severe structure distortion and large fibrosis areas; and grade 8, total fibrotic obliteration.
- Statistical analysis
- The normal distribution of data within each group was verified using the Kolmogorov-Smirnov test. Significant differences between the groups were tested by performing a one-way analysis of variance using SPSS version 16 (SPSS Inc., Chicago, IL, USA), followed by Tukey post hoc test. For each test, the data are expressed as the mean±standard deviation, and P<0.05 was considered statistically significant.
METHODS
- Plasma apoB levels were significantly higher in the NTD group than in the control group (P=0.001). No significant difference in plasma apoB level was found between the control and GETD groups (P=0.89). The plasma apoA level was significantly lower in the NTD group than that in the control group (P=0.0001). Ginger extract administration significantly increased the apoA level compared with the NTD group (P=0.012), but the plasma apoA level in this group remained significantly lower than that in the control group (P=0.001). The apoB/apoA ratio was significantly higher in the NTD group than in the control group (P=0.001). No significant differences in the apoB/apoA ratio were found between the GETD group and the control group (Table 1).
- The amounts of cathepsin G and Hcy in heart tissue were significantly higher in the NTD group than in the control group (P=0.0001) but were significantly lower in the GETD group than in the NTD group (P=0.001).
- The plasma CRP level was significantly higher in NTD rats than in control rats (P=0.001). Ginger extract treatment significantly decreased plasma levels of CRP compared to the NTD group (P=0.001), but levels remained significantly higher than those in the control group (P=0.001). The plasma leptin level in NTD group was significantly lower than that in the control group (P=0.001). Ginger extract treatment significantly increased the plasma level of leptin compared to that in the NTD group (P=0.001), but leptin levels remained significantly lower than those measured in the control group (P=0.05) (Table 1).
- There were no significant differences in body weight among the groups at the study outset. Body weight and body weight gain were significantly lower in the NTD group compared to the control group at the end of the study (P=0.001). Ginger extract treatment enhanced body weight and body weight gain non-significantly compared to the NTD group (P=0.26). Body weight and body weight gain were significantly lower in the GEDP group than in the control group (P=0.05) (Table 1).
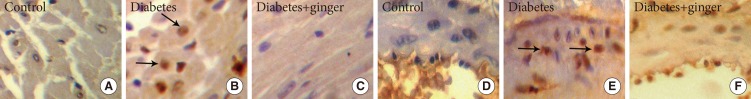
- The ratio of heart myocytes proliferated cells (PCNA positive indices) in the heart tissue of the control, NTD, and GETD groups were 4%±1%, 47%±3%, and 14%±4%, respectively. The PCNA-positive indices (as indicators of proliferation) were significantly higher in NTD hearts than in control hearts (P=0.002). Treatment with ginger extract significantly reduced the PCNA-positive indices compared to those found in the NTD group, but the indices in the GETD group remained significantly higher than those in the control group (P=0.001) (Fig. 1). PCNA-positive indices in the coronary vessel smooth muscle cells of NTD rats (51%±2%) were significantly higher than those in cells of the control rats (3%±1%; P=0.001). Ginger extract consumption with ethanol significantly decreased PCNA-positive indices (4%±1%) compared to those found in the NTD group (P=0.05). PCNA-positive indices did not significantly differ between the GETD and control groups (Fig. 2).
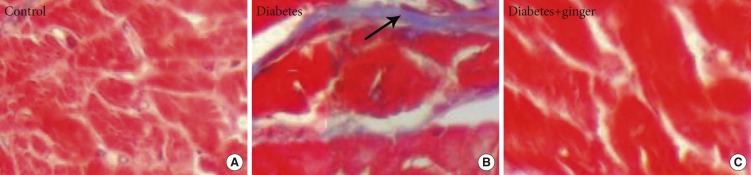
- Fig. 2 shows the microscopic fibrosis scores found in the heart tissue of rats of the different groups. The lesion score of the heart tissues of rats in the control group was 0 and that of rats in the NTD group was 2.5 (moderate thickening without obvious damage to the heart architecture); the latter was significantly higher than that of the control group (P=0.022). No significant differences were found between the GETD group and the control group (Fig. 2).
RESULTS
- The results of the current study showed that diabetes induced significant increases in cathepsin G, CRP, Hcy, and apoB. Moreover, the amounts of leptin and apoA were decreased. Structural changes including moderate fibrosis and mild to moderate heart and coronary muscle cell proliferation were also present in the hearts of diabetic rats when compared to the hearts of control group rats. All of these favor cardiovascular abnormalities. Significant amelioration or restoration of the heart tissue alterations and biochemical changes to those seen in the controls was found in the ginger extract-treated rats.
- In the current study, the apoA and apoB levels in the NTD group were significantly different from those in the control group; apoB levels and the apoB/apoA ratios were significantly higher and apoA levels were significantly lower in the NTD group than those in the control group.
- ApoA and apoB are included in the spectrum of lipoproteins such as those involved in LDL, very LDL, lipoprotein, and high density lipoprotein and determine the metabolic fate of these lipoproteins [19]. In general, apoB carries lipids from the liver and gut to tissues that use lipids, whereas apoA contains particles that mediate reverse lipid transport and carries excess lipids from peripheral tissues to the liver [19]. The concentrations of apoA and apoB are measured to assess cardiovascular disease because these concentrations reflect the particle numbers of corresponding lipoprotein classes and thus the opposite aspects cardiovascular risks [20]. A high apoB/apoA ratio designates a high number of atherogenic lipoprotein particles, which are likely to be deposited in the arterial wall [20]. Here, dramatic increases in apoB levels and apoB/apoA ratios were observed, together with coronary and heart muscle cell proliferation in the NTD group; these changes might favor the arterial deposition of lipids and consequent complications including proliferation. Another important finding of this study was the significantly lower leptin levels in the NTD group when compared to the levels in the control group.
- Recently, the role of the adipocyte-derived molecule leptin in maintaining normal energy homeostasis has been well documented [21]. Leptin adjusts the energy balance through central and peripheral pathways. Energy balance is centrally regulated by leptin through the inhibition of neuropeptide Y, which is the strongest stimulator of the hunger center. In addition, leptin stimulates the secretion of melanocyte-stimulating hormone, which also inhibits the appetite [22]. Peripherally, leptin adjusts the energy balance by modulating lipolysis and glucose tolerance by increasing the expression of enzymes that influence gluconeogenesis and glycogenolysis [2223]. According to previous studies, leptin has specific effects on the heart due to the expression of several isoforms of leptin receptors in the myocardium and in isolated cardiac myocytes [24]. A graph plotting the amount of leptin against cardiovascular function forms a U-shaped curve. In individuals with normoglycaemia and leptin sensitivity, in addition to its role in energy metabolism and homeostasis in myocytes, leptin reduces blood pressure by affecting mechanisms such as vasodilation through the release of nitric oxide from the endothelium [2526]. Meanwhile, accumulating evidence obtained in animal and human studies indicates a positive correlation between hyperleptinemia and structural and functional abnormalities of the heart (such as metabolic abnormalities, stroke, fibrosis, hypertrophy, hypertension, and heart failure) [2728].
- Indeed, abnormal energy homeostasis events, such as increased fatty acid utilization and decreased glucose utilization together with enhanced collagen deposition and matrix metalloproteinase activities have also been seen in rodent models that lack of leptin its receptors, thus resulting in diabetes or obesity [2930]. In this study, STZ-diabetic rats presented reduced levels of plasma leptin, as seen in previous studies [3031]. In addition, in the present study, leptin-deficient diabetic rats exhibited moderate fibrosis and muscle cell proliferation in heart tissue. Whether these structural changes were the results of leptin deficiency remains unclear. Previous studies indicated fibrosis of the heart tissue in obese animals with hyperleptinemia and in diabetic animals with leptin deficiency [3032]. In this study, moderate fibrosis and mild to moderate cell proliferation were found in the heart and coronary vessels of STZ-diabetic rats.
- Although fibrosis and vascular cell proliferation have been identified to be of potential significance for the pathogenesis and development of heart failure and atherosclerosis [33], the underlying mechanisms of heart cell proliferation and fibrosis have not been substantially investigated in diabetic hearts. Strong evidence exists showing that inflammatory biomarkers are essential components of fibrosis and muscle cell proliferation in heart tissue [3435].
- Our recent studies indicated that plasma serine proteases, proinflammatory cytokines, and cell adhesion molecules are elevated in STZ-diabetic rats [3637]. Due to the pivotal role of cathepsin G in tissue fibrosis, we examined the relationship between cathepsin G changes and heart cell proliferation and fibrosis in diabetic rats. Interestingly, we found that cathepsin G levels were increased with heart tissue fibrosis and proliferation in STZ-diabetic rats. To our knowledge, no other study has yet investigated the association between heart muscle cell proliferation, fibrosis, and cathepsin G as induced by diabetes in rats.
- A positive association was recently reported between cathepsin G elevation and heart tissue fibrosis. Cathepsin G induces myocyte hypertrophy and necrosis, and increases fibrosis by converting angiotension I to angiotension II [7]. Angiotension II, as a pro-fibrotic and pro-inflammatory mediator, activates the transforming growth factor β pathway, resulting in myocyte necrosis, hypertrophy, and fibrosis [67].
- Secondly, we examined the effect of ginger extract on rescuing the heart structural alteration, leptin, and inflammatory responses that are induced by diabetes in heart tissue. Previous work by us and others has shown that ginger administration has helpful features and protects against diabetes-induced abnormalities [3839]. The protective effect of ginger results from its antioxidant and anti-inflammatory properties. Due to the antioxidants contained in ginger, ginger supplementation increases the total antioxidant capacity and reduces lipid and protein oxidation in diabetes and other oxidative stress conditions [3840]. Accordingly, if the diabetes milieu induces functional and structural abnormalities through oxidative stress, as confirmed by previous studies, the effect of ginger supplementation on rescuing these abnormalities will be due to its antioxidant properties. It has also been shown that ginger has anti-inflammatory effects and suppresses the expression of pro-inflammatory cytokines such as TNF-α and inhibits of the arachidonic acid cascade [4142]. Mechanistically, the ginger compounds gingerol and shogaol inhibit prostaglandin and leukotriene biosynthesis through the suppression of 5-lipooxygenase synthetase [42]. Although various studies have shown the beneficial effects of ginger supplementation on inflammatory cytokines such as TNF-α, IL-6, protaglandins, and leukotrienes, this is the first in vivo study to demonstrate the protective effect of ginger extract against cathepsin G, together with heart tissue fibrosis and muscle cell proliferation, in diabetic hearts. Another interesting result was the elevation of leptin levels in the GETD group compared to the diabetic group. As mentioned above, at normal plasma levels, leptin plays a pivotal role not only in energy homeostasis but also in optimal cardiac function. Recent studies have indicated that leptin therapy alone reverses hyperglycemia and deaths caused by insulin signaling deficiencies [43], although the underlying mechanism of this golden effect of leptin is unknown. To our knowledge, the present study is the first to demonstrate that ginger supplementation improves leptin levels in diabetics and might be used in the management and treatment of diabetes-induced abnormities.
- In conclusion, several notable findings were observed in this study. The rats with STZ-induced diabetes generated cathepsin G at higher levels and exhibited higher levels of Hcy, CRP, and apoB, as well as lower levels of apoA and leptin. In addition, we observed evidence of heart structure alterations, such as moderate fibrosis and mild to moderate heart and coronary muscle cell proliferation in the diabetic rats (compared to the control rats). Furthermore, the administration of ginger extract alleviated these structural changes and restored the levels of inflammatory cytokines, apoA and apoB, Hcy, and leptin in the experimental group (compared to the control group). These restorative effects of ginger extract support the hypothesis that diabetes-related alterations in the heart structure are caused by oxidative stress together with inflammatory responses and leptin deficiency. Further research is required to elucidate the details of the mechanisms by which ginger extract exerts its rescue effects against diabetes-related complications; such information would enable the development of feasible alternatives to insulin therapy for the treatment of diabetes complications.
DISCUSSION
-
Acknowledgements
- This research was supported by a grant from the Office of the Vice Chancellor for Research and Technology, Urmia University of Medical Sciences, Urmia, Iran.
ACKNOWLEDGMENTS
-
CONFLICTS OF INTEREST: No potential conflict of interest relevant to this article was reported.
NOTES
- 1. Poornima IG, Parikh P, Shannon RP. Diabetic cardiomyopathy: the search for a unifying hypothesis. Circ Res 2006;98:596-605. PubMed
- 2. Shirpoor A, Salami S, Khadem-Ansari MH, Ilkhanizadeh B, Pakdel FG, Khademvatani K. Cardioprotective effect of vitamin E: rescues of diabetes-induced cardiac malfunction, oxidative stress, and apoptosis in rat. J Diabetes Complications 2009;23:310-316. ArticlePubMed
- 3. Zhao J, Randive R, Stewart JA. Molecular mechanisms of AGE/RAGE-mediated fibrosis in the diabetic heart. World J Diabetes 2014;5:860-867. ArticlePubMedPMC
- 4. Okatan EN, Tuncay E, Turan B. Cardioprotective effect of selenium via modulation of cardiac ryanodine receptor calcium release channels in diabetic rat cardiomyocytes through thioredoxin system. J Nutr Biochem 2013;24:2110-2118. ArticlePubMed
- 5. Westermann D, Rutschow S, Jager S, Linderer A, Anker S, Riad A, Unger T, Schultheiss HP, Pauschinger M, Tschope C. Contributions of inflammation and cardiac matrix metalloproteinase activity to cardiac failure in diabetic cardiomyopathy: the role of angiotensin type 1 receptor antagonism. Diabetes 2007;56:641-646. PubMed
- 6. Jahanyar J, Youker KA, Loebe M, Assad-Kottner C, Koerner MM, Torre-Amione G, Noon GP. Mast cell-derived cathepsin g: a possible role in the adverse remodeling of the failing human heart. J Surg Res 2007;140:199-203. ArticlePubMed
- 7. Reilly CF, Tewksbury DA, Schechter NM, Travis J. Rapid conversion of angiotensin I to angiotensin II by neutrophil and mast cell proteinases. J Biol Chem 1982;257:8619-8622. ArticlePubMed
- 8. Soderberg S, Stegmayr B, Ahlbeck-Glader C, Slunga-Birgander L, Ahren B, Olsson T. High leptin levels are associated with stroke. Cerebrovasc Dis 2003;15:63-69. ArticlePubMedPDF
- 9. Wannamethee SG, Shaper AG, Whincup PH, Lennon L, Sattar N. Obesity and risk of incident heart failure in older men with and without pre-existing coronary heart disease: does leptin have a role? J Am Coll Cardiol 2011;58:1870-1877. PubMed
- 10. Ku IA, Farzaneh-Far R, Vittinghoff E, Zhang MH, Na B, Whooley MA. Association of low leptin with cardiovascular events and mortality in patients with stable coronary artery disease: the Heart and Soul Study. Atherosclerosis 2011;217:503-508. ArticlePubMed
- 11. Hou N, Luo JD. Leptin and cardiovascular diseases. Clin Exp Pharmacol Physiol 2011;38:905-913. ArticlePubMed
- 12. Bluestone JA, Herold K, Eisenbarth G. Genetics, pathogenesis and clinical interventions in type 1 diabetes. Nature 2010;464:1293-1300. ArticlePubMedPMCPDF
- 13. Liu HY, Cao SY, Hong T, Han J, Liu Z, Cao W. Insulin is a stronger inducer of insulin resistance than hyperglycemia in mice with type 1 diabetes mellitus (T1DM). J Biol Chem 2009;284:27090-27100. ArticlePubMedPMC
- 14. Utriainen T, Malmstrom R, Makimattila S, Yki-Jarvinen H. Supraphysiological hyperinsulinemia increases plasma leptin concentrations after 4 h in normal subjects. Diabetes 1996;45:1364-1366. ArticlePubMed
- 15. Kolaczynski JW, Nyce MR, Considine RV, Boden G, Nolan JJ, Henry R, Mudaliar SR, Olefsky J, Caro JF. Acute and chronic effects of insulin on leptin production in humans: studies in vivo and in vitro. Diabetes 1996;45:699-701. ArticlePubMed
- 16. Boden G, Chen X, Kolaczynski JW, Polansky M. Effects of prolonged hyperinsulinemia on serum leptin in normal human subjects. J Clin Invest 1997;100:1107-1113. ArticlePubMedPMC
- 17. Nile SH, Park SW. Chromatographic analysis, antioxidant, anti-inflammatory, and xanthine oxidase inhibitory activities of ginger extracts and its reference compounds. Ind Crops Prod 2015;70:238-244.Article
- 18. Ashcroft T, Simpson JM, Timbrell V. Simple method of estimating severity of pulmonary fibrosis on a numerical scale. J Clin Pathol 1988;41:467-470. ArticlePubMedPMC
- 19. Marcovina S, Packard CJ. Measurement and meaning of apolipoprotein AI and apolipoprotein B plasma levels. J Intern Med 2006;259:437-446. ArticlePubMed
- 20. Walldius G, Jungner I. The apoB/apoA-I ratio: a strong, new risk factor for cardiovascular disease and a target for lipid-lowering therapy: a review of the evidence. J Intern Med 2006;259:493-519. ArticlePubMed
- 21. Ahima RS, Osei SY. Leptin signaling. Physiol Behav 2004;81:223-241. ArticlePubMed
- 22. Kalra SP. Central leptin insufficiency syndrome: an interactive etiology for obesity, metabolic and neural diseases and for designing new therapeutic interventions. Peptides 2008;29:127-138. ArticlePubMed
- 23. Gutierrez-Juarez R, Obici S, Rossetti L. Melanocortin-independent effects of leptin on hepatic glucose fluxes. J Biol Chem 2004;279:49704-49715. ArticlePubMed
- 24. Rajapurohitam V, Gan XT, Kirshenbaum LA, Karmazyn M. The obesity-associated peptide leptin induces hypertrophy in neonatal rat ventricular myocytes. Circ Res 2003;93:277-279. ArticlePubMed
- 25. Lembo G, Vecchione C, Fratta L, Marino G, Trimarco V, d'Amati G, Trimarco B. Leptin induces direct vasodilation through distinct endothelial mechanisms. Diabetes 2000;49:293-297. ArticlePubMedPDF
- 26. Vecchione C, Maffei A, Colella S, Aretini A, Poulet R, Frati G, Gentile MT, Fratta L, Trimarco V, Trimarco B, Lembo G. Leptin effect on endothelial nitric oxide is mediated through Akt-endothelial nitric oxide synthase phosphorylation pathway. Diabetes 2002;51:168-173. ArticlePubMedPDF
- 27. Sweeney G. Cardiovascular effects of leptin. Nat Rev Cardiol 2010;7:22-29. ArticlePubMedPDF
- 28. Guzman-Ruiz R, Somoza B, Gil-Ortega M, Merino B, Cano V, Attane C, Castan-Laurell I, Valet P, Fernandez-Alfonso MS, Ruiz-Gayo M. Sensitivity of cardiac carnitine palmitoyltransferase to malonyl-CoA is regulated by leptin: similarities with a model of endogenous hyperleptinemia. Endocrinology 2010;151:1010-1018. ArticlePubMedPDF
- 29. Abel ED, Litwin SE, Sweeney G. Cardiac remodeling in obesity. Physiol Rev 2008;88:389-419. ArticlePubMed
- 30. McGaffin KR, Witham WG, Yester KA, Romano LC, O'Doherty RM, McTiernan CF, O'Donnell CP. Cardiac-specific leptin receptor deletion exacerbates ischaemic heart failure in mice. Cardiovasc Res 2011;89:60-71. ArticlePubMed
- 31. Schram K, Sweeney G. Implications of myocardial matrix remodeling by adipokines in obesity-related heart failure. Trends Cardiovasc Med 2008;18:199-205. ArticlePubMed
- 32. Zibadi S, Cordova F, Slack EH, Watson RR, Larson DF. Leptin's regulation of obesity-induced cardiac extracellular matrix remodeling. Cardiovasc Toxicol 2011;11:325-333. ArticlePubMedPDF
- 33. Shirpoor A, Norouzi L, Khadem Ansari MH, Ilkhanizadeh B, Gharaaghaji R. Vasoprotective effect of vitamin E: rescue of ethanol-induced atherosclerosis and inflammatory stress in rat vascular wall. Int Immunopharmacol 2013;16:498-504. ArticlePubMed
- 34. Passino C, Barison A, Vergaro G, Gabutti A, Borrelli C, Emdin M, Clerico A. Markers of fibrosis, inflammation, and remodeling pathways in heart failure. Clin Chim Acta 2015;443:29-38. ArticlePubMed
- 35. Shirpoor A, Nemati S, Ansari MH, Ilkhanizadeh B. The protective effect of vitamin E against prenatal and early postnatal ethanol treatment-induced heart abnormality in rats: a 3-month follow-up study. Int Immunopharmacol 2015;26:72-79. ArticlePubMed
- 36. Shirpoor A, Khadem Ansari MH, Heshmatian B, Ilkhanizadeh B, Noruzi L, Abdollahzadeh N, Saboory E. Decreased blood pressure with a corresponding decrease in adhesive molecules in diabetic rats caused by vitamin E administration. J Diabetes 2012;4:362-368. ArticlePubMed
- 37. Alireza S, Leila N, Siamak S, Mohammad-Hasan KA, Behrouz I. Effects of vitamin E on pathological changes induced by diabetes in rat lungs. Respir Physiol Neurobiol 2013;185:593-599. ArticlePubMed
- 38. Taghizadeh Afshari A, Shirpoor A, Farshid A, Saadatian R, Rasmi Y, Saboory E, Ilkhanizadeh B, Allameh A. The effect of ginger on diabetic nephropathy, plasma antioxidant capacity and lipid peroxidation in rats. Food Chem 2007;101:148-153.Article
- 39. Shanmugam KR, Mallikarjuna K, Nishanth K, Kuo CH, Sathyavelu Reddy K. Protective effect of dietary ginger on antioxidant enzymes and oxidative damage in experimental diabetic rat tissues. Food Chem 2011;124:1436-1442.Article
- 40. Ramudu SK, Korivi M, Kesireddy N, Chen CY, Kuo CH, Kesireddy SR. Ginger feeding protects against renal oxidative damage caused by alcohol consumption in rats. J Ren Nutr 2011;21:263-270. ArticlePubMed
- 41. Isa Y, Miyakawa Y, Yanagisawa M, Goto T, Kang MS, Kawada T, Morimitsu Y, Kubota K, Tsuda T. 6-Shogaol and 6-gingerol, the pungent of ginger, inhibit TNF-alpha mediated downregulation of adiponectin expression via different mechanisms in 3T3-L1 adipocytes. Biochem Biophys Res Commun 2008;373:429-434. PubMed
- 42. Srivastava KC, Mustafa T. Ginger (Zingiber officinale) in rheumatism and musculoskeletal disorders. Med Hypotheses 1992;39:342-348. ArticlePubMed
- 43. German JP, Wisse BE, Thaler JP, Oh IS, Sarruf DA, Ogimoto K, Kaiyala KJ, Fischer JD, Matsen ME, Taborsky GJ Jr, Schwartz MW, Morton GJ. Leptin deficiency causes insulin resistance induced by uncontrolled diabetes. Diabetes 2010;59:1626-1634. ArticlePubMedPMCPDF
REFERENCES
Fig. 1
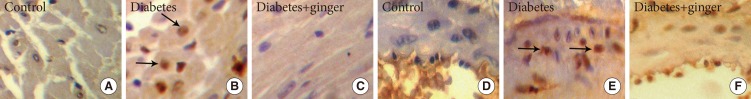
Immunohistochemical staining of heart and coronary tissue for proliferating cell nuclear antigen (PCNA) indicate the proliferation of heart muscle (A-C) and coronary smooth muscle cells (D-F) in the diabetic group compared to the control group. Ginger extract treatment along diabetes reduced cell proliferation in both tissues (×400). PCNA positive indices (arrows).

Fig. 2
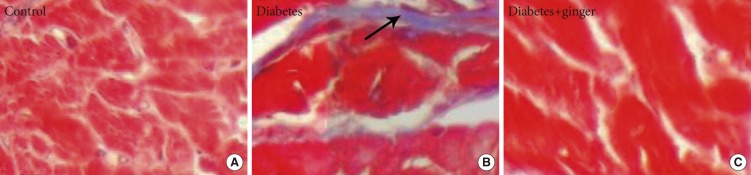
(A-C) Masson trichrome staining showing fibrosis bonds in the heart tissue of diabetic rats compared to the control group. No fibrosis bond or accumulation was observed in the heart tissue of the ginger-extract treated group (×400). Fibrosis bond (arrow).

Table 1
![dmj-40-46-i001.jpg]()
Effect of ginger extract on diabetes-induced changes in plasma apolipoproteins, leptin, cathepsin G, CRP, and Hcy
Figure & Data
References
Citations
Citations to this article as recorded by 

- Cardioprotective Activities of some Indian Spices: An Insight into
Pharmacology and Phytochemical Investigation
Kalyani Pathak, Manash Pratim Pathak, Riya Saikia, Urvashee Gogoi, Jon Jyoti Sahariah, Aparoop Das, Mohammad Zaki Ahmad, Tirna Paul, Jyotirmoy Das, Saif Aboud M. Alqahtani
Current Traditional Medicine.2024;[Epub] CrossRef - Network-pharmacology-based research on protective effects and underlying mechanism of Shuxin decoction against myocardial ischemia/reperfusion injury with diabetes
Ling Yang, Yang Jian, Zai-Yuan Zhang, Bao-Wen Qi, Yu-Bo Li, Pan Long, Yao Yang, Xue Wang, Shuo Huang, Jing Huang, Long-Fu Zhou, Jie Ma, Chang-Qing Jiang, Yong-He Hu, Wen-Jing Xiao
World Journal of Diabetes.2023; 14(7): 1057. CrossRef - Evaluating the effect of Ginger Powder on Bilirubin, Creatinine, Urea and Uric Acid on Experimental Diabetic Wistar Rats in Randomized Control Trial
Maryam Maqsood, Saima Naaz, Huma Bader Ul Ain, Zunaira Mushtaq, Makia Nasir, Aiza Qamar
Pakistan BioMedical Journal.2022; 5(1): 351. CrossRef - The effect of red ginger bread consumption on the physiological parameters of healthy subjects
Titin Sulastri, Marleen Sunyoto, Marvel Reuben Suwitono, Jutti Levita
Journal Of Advanced Pharmacy Education And Research.2022; 12(3): 28. CrossRef - Protective effects of medicinal plant against diabetes induced cardiac disorder: A review
Sadegh Shabab, Zahra Gholamnezhad, Maryam Mahmoudabady
Journal of Ethnopharmacology.2021; 265: 113328. CrossRef - Ginger (Zingiber Officinale Roscoe) Extract Protects the Heart Against Inflammation and Fibrosis in Diabetic Rats
Tara Abdi, Maryam Mahmoudabady, Hadi Zare Marzouni, Saeed Niazmand, Majid Khazaei
Canadian Journal of Diabetes.2021; 45(3): 220. CrossRef - Combined ginger and garlic extract improves serum lipid profile, oxidative stress markers and reduced IL-6 in diet induced obese rats
Peter Ifeoluwa Adegbola, Olumide Samuel Fadahunsi, Bamidele Stephen Ajilore, Adebola Olayemi Akintola, Olubukola Sinbad Olorunnisola
Obesity Medicine.2021; 23: 100336. CrossRef - The Effects of Medicinal Plants and Bioactive Natural Compounds on Homocysteine
Mohammad Amin Atazadegan, Mohammad Bagherniya, Gholamreza Askari, Aida Tasbandi, Amirhossein Sahebkar
Molecules.2021; 26(11): 3081. CrossRef - Long-chain noncoding RNA-GAS5/hsa-miR-138-5p attenuates high glucose-induced cardiomyocyte damage by targeting CYP11B2
Xiaozhen Zhuo, Kai Bai, Yingxian Wang, Peining Liu, Wen Xi, Jianqing She, Junhui Liu
Bioscience Reports.2021;[Epub] CrossRef - Regulation of Apolipoprotein B by Natural Products and Nutraceuticals: A Comprehensive Review
Mohammad Bagherniya, Thomas P. Johnston, Amirhossein Sahebkar
Current Medicinal Chemistry.2021; 28(7): 1363. CrossRef - Ameliorative effects of Hydrolea zeylanica in streptozotocin-induced oxidative stress and metabolic changes in diabetic rats
Sandeep Kumar Swain, Umesh Chandra Dash, Satish Kanhar, Atish Kumar Sahoo
Journal of Ethnopharmacology.2020; 247: 112257. CrossRef - The effect of 8 weeks of high-intensity interval training and moderate-intensity continuous training on cardiac angiogenesis factor in diabetic male rats
Faramarz Yazdani, Fereshteh Shahidi, Pouran Karimi
Journal of Physiology and Biochemistry.2020; 76(2): 291. CrossRef - DNA methyltransferase-1 inactivation of androgen receptor axis triggers homocysteine induced cardiac fibroblast autophagy in diabetic cardiac fibrosis
Hui Tao, Peng Shi, Hai-Yang Xuan, Xuan-Sheng Ding
Archives of Biochemistry and Biophysics.2020; 692: 108521. CrossRef - Ameliorative and protective effects of ginger and its main constituents against natural, chemical and radiation-induced toxicities: A comprehensive review
Muhammad A. Alsherbiny, Wessam H. Abd-Elsalam, Shymaa A. El badawy, Ehab Taher, Mohamed Fares, Allan Torres, Dennis Chang, Chun Guang Li
Food and Chemical Toxicology.2019; 123: 72. CrossRef - SELECTION OF A FILLER FOR TABLETS MANUFACTURED WITH DIRECT COMPRESSION METHOD CONTAINING DRY GINGER EXTRACT
Оlena Ruban, Malek Alkhalaf, Nataliia Gerbina
EUREKA: Health Sciences.2019; 3: 26. CrossRef - Role of medicinal plants in the management of diabetes mellitus: a review
Bindu Jacob, Narendhirakannan R.T.
3 Biotech.2019;[Epub] CrossRef - Ginger (Zingiber officinale Roscoe) in the Prevention of Ageing and Degenerative Diseases: Review of Current Evidence
Nur Fatin Nabilah Mohd Sahardi, Suzana Makpol
Evidence-Based Complementary and Alternative Medicine.2019; 2019: 1. CrossRef - Roles of Spicy Foods and Their Bioactive Compounds in Management of Hypercholesterolemia
Yimin Zhao, Zhen-Yu Chen
Journal of Agricultural and Food Chemistry.2018; 66(33): 8662. CrossRef - Relaxant and vasoprotective effects of ginger extracts on porcine coronary arteries
Hsing‑Chen Wu, Chi‑Ting Horng, Shih‑Chang Tsai, You‑Li Lee, Shou‑Cheng Hsu, Yi‑Jen Tsai, Fuu‑Jen Tsai, Jo‑Hua Chiang, Daih‑Huang Kuo, Jai‑Sing Yang
International Journal of Molecular Medicine.2018;[Epub] CrossRef - Increased hepatic FAT/CD36, PTP1B and decreased HNF4A expression contributes to dyslipidemia associated with ethanol–induced liver dysfunction: Rescue effect of ginger extract
Alireza Shirpoor, Elaheh Heshmati, Fatemeh Kheradmand, Farzaneh Hosseini Gharalari, Leila Chodari, Roya Naderi, Farideh Nezami Majd, Mahrokh Samadi
Biomedicine & Pharmacotherapy.2018; 105: 144. CrossRef - Ginger extract mitigates ethanol-induced changes of alpha and beta – myosin heavy chain isoforms gene expression and oxidative stress in the heart of male wistar rats
Alireza Shirpoor, Mitra Zerehpoosh, Mohammad Hasan Khadem Ansari, Fatemeh Kheradmand, Yousef Rasmi
DNA Repair.2017; 57: 45. CrossRef - Single, repeated dose toxicity and genotoxicity assessment of herb formula KIOM2012H
Hwayong Park, Youn-Hwan Hwang, Jin Yeul Ma
Integrative Medicine Research.2017; 6(4): 361. CrossRef - Rescue effects of ginger extract on dose dependent radiation-induced histological and biochemical changes in the kidneys of male Wistar rats
Hassan Saberi, Behnaz Keshavarzi, Alireza Shirpoor, Farzaneh Hosseini Gharalari, Yousef Rasmi
Biomedicine & Pharmacotherapy.2017; 94: 569. CrossRef - Promising Antidiabetic Drugs, Medicinal Plants and Herbs: An Update
Mohd Iqbal Yatoo, Archana Saxena, Arumugam Gopalakris, Mahmoud Alagawany, Kuldeep Dhama
International Journal of Pharmacology.2017; 13(7): 732. CrossRef - Cardioprotective effects of rutin via alteration in TNF-α, CRP, and BNP levels coupled with antioxidant effect in STZ-induced diabetic rats
Ravi Saklani, Suresh Kumar Gupta, Ipseeta Ray Mohanty, Binit Kumar, Sushma Srivastava, Rajani Mathur
Molecular and Cellular Biochemistry.2016; 420(1-2): 65. CrossRef

 KDA
KDA PubReader
PubReader Cite
Cite