
- Current
- Browse
- Collections
-
For contributors
- For Authors
- Instructions to authors
- Article processing charge
- e-submission
- For Reviewers
- Instructions for reviewers
- How to become a reviewer
- Best reviewers
- For Readers
- Readership
- Subscription
- Permission guidelines
- About
- Editorial policy
Articles
- Page Path
- HOME > Diabetes Metab J > Volume 40(1); 2016 > Article
-
ReviewFibroblast Growth Factor 21 Protects against Atherosclerosis via Fine-Tuning the Multiorgan Crosstalk
-
Leigang Jin1,2, Zhuofeng Lin3, Aimin Xu1,2,4

-
Diabetes & Metabolism Journal 2016;40(1):22-31.
DOI: https://doi.org/10.4093/dmj.2016.40.1.22
Published online: January 29, 2016
1State Key Laboratory of Pharmaceutical Biotechnology, the University of Hong Kong, Hong Kong.
2Department of Pharmacology and Pharmacy, the University of Hong Kong, Hong Kong.
3School of Pharmacology, Wenzhou Medical University, Wenzhou, China.
4Department of Medicine, the University of Hong Kong, Hong Kong.
- Corresponding author: Aimin Xu. Department of Medicine, the University of Hong Kong, L8-40, New Laboratory Block, 21 Sassoon Road, Hong Kong. amxu@hkucc.hku.hk
Copyright © 2016 Korean Diabetes Association
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
- ABSTRACT
- INTRODUCTION
- BASIC BIOLOGY AND PHYSIOLOGY OF FGF21
- FGF21 AS A SENSOR OF METABOLIC STRESS AND A MEDIATOR OF MULTIORGAN CROSSTALK
- FGF21 AS A PHYSIOLOGICAL PROTECTOR AGAINST ATHEROSCLEROSIS
- ADIPONECTIN AS A DOWNSTREAM EFFECTOR OF FGF21 IN PROTECTION AGAINST ATHEROSCLEROSIS
- LIVER AS A MAJOR TARGET OF FGF21 FOR SUPPRESSION OF HYPERCHOLESTEROLEMIA
- CONCLUSIONS
- ACKNOWLEDGMENTS
- NOTES
- REFERENCES
ABSTRACT
- Fibroblast growth factor 21 (FGF21) is a metabolic hormone with pleiotropic effects on energy metabolism and insulin sensitivity. Besides its antiobese and antidiabetic activity, FGF21 also possesses the protective effects against atherosclerosis. Circulating levels of FGF21 are elevated in patients with atherosclerosis, macrovascular and microvascular complications of diabetes, possibly due to a compensatory upregulation. In apolipoprotein E-deficient mice, formation of atherosclerotic plaques is exacerbated by genetic depletion of FGF21, but is attenuated upon replenishment with recombinant FGF21. However, the blood vessel is not the direct target of FGF21, and the antiatherosclerotic activity of FGF21 is attributed to its actions in adipose tissues and liver. In adipocytes, FGF21 promotes secretion of adiponectin, which in turn acts directly on blood vessels to reduce endothelial dysfunction, inhibit proliferation of smooth muscle cells and block conversion of macrophages to foam cells. Furthermore, FGF21 suppresses cholesterol biosynthesis and attenuates hypercholesterolemia by inhibiting the transcription factor sterol regulatory element-binding protein-2 in hepatocytes. The effects of FGF21 on elevation of adiponectin and reduction of hypercholesterolemia are also observed in a phase-1b clinical trial in patients with obesity and diabetes. Therefore, FGF21 exerts its protection against atherosclerosis by fine-tuning the interorgan crosstalk between liver, brain, adipose tissue, and blood vessels.
- Fibroblast growth factor 21 (FGF21), along with FGF19 and FGF23, are endocrine factors that structurally belong to the FGF superfamily [1]. Unlike the classic FGFs that require heparin for stable binding to the FGF receptors (FGFRs), the three endocrine members of the FGF superfamily lack the heparin-binding property, and therefore can be released into the circulation [2]. These hormone-like FGFs are involved in the regulation of diverse metabolic pathways. FGF15/19 controls cholesterol/bile acid synthesis. FGF23 modulates phosphate/vitamin D metabolism, whereas FGF21 regulates glucose and lipid metabolism [2]. Physiologically, FGF21 expression is markedly increased in response to fasting/starvation, and elevated FGF21 is a central mediator for fasting-induced fatty acid oxidation, ketogenesis and for inducing growth hormone resistance [34]. Pharmacologically, therapeutic intervention with recombinant FGF21 counteracts obesity and its related metabolic disorders in both rodents and nonhuman primates, including reduction of adiposity and amelioration of hyperglycemia, hyperinsulinemia, insulin resistance, dyslipidemia, and fatty liver disease [5]. Furthermore, FGF21 is the downstream target of both peroxisome proliferator-activated receptor (PPAR) α and PPARγ, and a growing body of evidence suggests that the glucose-lowering and insulin-sensitizing effects of the PPARγ agonists (thiazolidinediones [TZDs]) and the therapeutic benefits of the PPARα agonists (fenofibrates) on lipid profiles are mediated in part by induction of FGF21 [67].
- In addition to its multiple metabolic actions, emerging evidence suggests that FGF21 also possesses vascular protective activities, independent of its effects on glycemic control and insulin sensitivity. This review aims to summarize recent clinical and animal studies in this field, and discuss the molecular pathways underlying the antiatherosclerotic activities of FGF21.
INTRODUCTION
- FGF21 was initially identified as a metabolic regulator by a high throughput glucose uptake screen in 3T3-L1 adipocytes [8]. Although liver is a major source for circulating FGF21 [49], expression of FGF21 is also detectable in several other metabolic organs, including adipose tissue [1011], pancreatic islets [12], and skeletal muscle [13]. Due to lack of the heparin-binding property, FGF21 itself is not sufficient to bind the FGFRs, but requires the recruitment of the single-pass transmembrane glycoprotein βKlotho as a co-receptor [14151617], through its direction with the carboxyl terminus of FGF21 [18]. Both in vivo and in vitro studies have demonstrated the essential role of βKlotho in conferring the metabolic effects of FGF21 [14151617]. Unlike the ubiquitous expression pattern of FGFRs, βKlotho expression is restricted to a number of metabolic tissues (adipose tissue, liver, brain, and pancreas) [19], which may explain the tissue selectivity of the FGF21 targets.
- The binding of FGF21 to its receptor complex has been shown to activate several downstream signaling pathways, including the mitogen-activated protein kinase (MAPK)/extracellular signal-regulated protein kinase 1/2 (ERK1/2) [8141520], protein kinase B (Akt) [12], adenosine monophosphate (AMP)-activated protein kinase, and sirtuin 1 (Sirt1) [21]. Although phosphorylation of ERK1/2 and its downstream targets has been widely used as a maker for FGF21 signaling, it is unlikely that ERK1/2 is a key player in mediating the multiple metabolic functions of FGF21. The molecular events that link the FGF21 receptor complex with its distal effects remain poorly characterized. It is also currently unclear why FGF21 does not have any mitogenic activity despite its ability to activate ERK1/2.
BASIC BIOLOGY AND PHYSIOLOGY OF FGF21
- In rodents, circulating levels of FGF21 are extremely low in fed status, but are markedly induced in multiple organs in response to diverse physiological or pathological stressors, such as starvation, nutrient excess, autophagy deficiency, mitochondrial dysfunction, exercise, drug toxicity, and cold exposure [22]. Starvation triggers lipolysis in adipose tissue to release free fatty acids, which in turn activate hepatic PPARα for production of FGF21. Elevated FGF21 released from the liver travels across the blood-brain barrier and enters the brain to activate hypothalamic neurons for production of corticotropin-releasing hormone, which in turn triggers the pituitary release of adrenocorticotropic hormone and subsequent secretion of corticosterone from adrenal gland. Corticosterone acts on the hepatocytes to promote hepatic gluconeogenesis [23]. Therefore, FGF21 maintain fasting glucose homeostasis by mediating the crosstalk between liver, adipose tissue and brain via activation of the hypothalamus-pituitary-adrenal (HPA) axis [2324].
- In response to overdose of acetaminophen, both circulating levels of FGF21 and its hepatic production are drastically elevated [25], which in turn act in a compensatory manner to protect against the drug-induced hepatotoxicity by enhancing PGC-1α/Nrf2-mediated antioxidant capacity in the liver. Mitochondrial dysfunction caused by autophagy deficiency triggers the release of FGF21 from skeletal muscle, which in turn acts as a myokine to protect against high fat diet-induced obesity and insulin resistance possibly via its actions in adipose tissues [26]. On the other hand, cold exposure causes elevated expression in both brown and white adipose tissues, but not in the liver [27]. Notably, FGF21 has been identified as an obligatory mediator of environmental cold-induced browning and thermogenesis of white adipose tissues in mice [28]. However, it is currently unclear whether such a browning effect of FGF21 is attributed to its autocrine action adipose tissue, or endocrine actions via interorgan crosstalk. A recent study showed that the central actions of FGF21 may contribute to its browning activity in white adipose tissue [29], suggesting that FGF21 is also a mediator for the crosstalk between brain and adipose tissue.
FGF21 AS A SENSOR OF METABOLIC STRESS AND A MEDIATOR OF MULTIORGAN CROSSTALK
- Although early studies of FGF21 focused predominantly on its metabolic activities, emerging evidence suggests that FGF21 is also a physiological protector of vascular functions in both rodents and humans. In a study cohort including 670 Chinese subjects with measurement of carotid intima-media thickness, we have observed a positive correlation between elevated serum FGF21 levels and carotid atherosclerosis, independent of the traditional risk factors including smoking, creatinine, C-reactive protein, dysglycemia, and dyslipidemia [30]. Furthermore, a strong association of serum FGF21 levels with subclinical atherosclerosis and low-extremity atherosclerosis has also been observed in patients with type 2 diabetes [3132]. In line with these clinical observations, circulating levels of FGF21 are progressively elevated with ageing in apolipoprotein E-deficient mice (apoE-/-), a well-known model with spontaneous development of atherosclerosis [33].
- To address the pathophysiological roles of FGF21 in the development of atherosclerosis, we have recently generated FGF21 and apoE double knockout (DKO) mice by crossing FGF21 knockout mice with apoE-/- mice in C57BL/J background [33]. Notably, both ageing- and western diet-induced atherosclerotic plaque formation (as determined by oil red O staining of entire aorta) in DKO mice is markedly exacerbated compared to that in apoE-/- littermates. Genetic depletion of FGF21 in apoE-/- mice also leads to premature death, possibly due to the development of atherosclerotic heart disease. In the atherosclerotic lesion areas of DKO mice, smooth muscle proliferation, macrophage infiltration, and collagen deposition are also significantly increased in comparison with apoE-/- mice [33]. There is no obvious difference in body weight, glucose tolerance and insulin sensitivity between DKO mice and apoE-/- mice on either standard or western diet. On the other hand, DKO mice exhibit markedly increased levels of proinflammatory chemokines (intercellular adhesion molecule-1 [ICAM-1], vascular cell adhesion protein-1 [VCAM-1], and monocyte chemotactic protein-1 [MCP-1]) and cytokines (tumor necrosis factor α [TNFα]) in both atherosclerotic lesion areas and bloodstream, and also worsened lipid profiles (hypercholesterolemia, decreased high density lipoprotein [HDL]/low density lipoprotein [LDL] ratio). All these changes can be reversed by treatment with recombinant FGF21. Taken together, these findings support the notion that FGF21 is a physiological protector against atherosclerosis possibly via its effects on alleviation of vascular inflammation and hypercholesterolemia, independent of adiposity, glycemic control, and insulin sensitivity. The elevated levels of circulating FGF21, as observed in both patients and animals with atherosclerosis, may serve as an adaptive mechanism to defend against this disease.
FGF21 AS A PHYSIOLOGICAL PROTECTOR AGAINST ATHEROSCLEROSIS
- Despite the potent antiatherosclerotic activity of FGF21, its co-receptor βKlotho is hardly detectable in blood vessels (L.J. and A.X., unpublished data). Therefore, we tested the possibility that FGF21 may exert its vascular protective effects indirectly, via induction of adiponectin, which is one of the most abundant adipokines secreted from adipocytes. Circulating levels of adiponectin are decreased in obesity and a cluster of obesity-related cardiometabolic diseases [3435]. Epidemiological studies in different ethnic groups have reproducibly identified low levels of adiponectin (hypoadiponectinemia) as an independent risk predictor for type 2 diabetes, atherosclerosis and coronary heart disease [3436]. Conversely, elevation of circulating adiponectin by either genetic or pharmacological approaches can reverse insulin resistance, hyperglycemia, metabolic inflammation, atherosclerosis, myocardial infarction and stroke in either rodents or pig models [37]. In particular, adiponectin exerts its cardiovascular protect effects through its direct actions in almost all major types of cells in both the blood vessels and heart [3738]. In endothelial cells, adiponectin induces production of nitric oxide via activation of endothelial nitric oxide synthase [39], reduces high glucose-induced oxidative stress and suppresses expression of adhesion molecules and the cell interaction with leucocytes [40]. In macrophages, adiponectin suppresses both endotoxin- and toxic lipids-induced inflammation and blocks foam cell formation by inhibiting uptake of oxidized LDL and enhancing cholesterol efflux [41]. Furthermore, adiponectin inhibits growth factors-induced proliferation and migration of smooth muscle cells [42], and prevents platelet aggregation and activation. Notably, adiponectin-deficient mice are refractory to the protective effects of rosiglitazone against diabetes-induced vascular damage [43], suggesting an obligatory role of adiponectin in mediating the vascular protection of this PPARγ agonist.
- Recent data from both us [44] and Scherer's group [45] has demonstrated the potent effects of FGF21 in inducing both expression and secretion of adiponectin. Notably, increased adiponectin is observed within 1 hour after administration with recombinant FGF21. Mechanistically, we found that both the acute and chronic effects of FGF21 on adiponectin production are mediated in part by PPARγ, a nuclear receptor that controls the gene transcription as well as the protein secretion of adiponectin [4647]. Furthermore, the stimulatory effects of the PPARγ agonists TZDs on adiponectin production in both mouse adipocytes and human adipose tissue are mediated by augmented autocrine actions of FGF21 [44]. In both fat explants isolated from PPARγ+/- mice and human fat explants pre-treated with the PPARγ antagonist GW9662, FGF21-induced production of adiponectin is partially abrogated, perhaps due to reduced expression of endoplasmic reticulum oxidoreductin-1-like α and disulfide-bond A oxidoreductase-like protein, both of which are molecular chaperones involved in adiponectin oligomerization and secretion [4849]. In support of this notion, a positive feedback loop between FGF21 and PPARγ in adipocytes has been shown to be critical in maintaining systemic glucose homeostasis [7]. The autocrine actions of FGF21 activate PPARγ possibly by modulation of sumoylation, which in turn leads to further induction of FGF21 expression via transcriptional activation. Consistent with these animal-based observations, a recent phase-1b clinical trial showed that treatment of obese human subjects with type 2 diabetes with LY2405319, a long-acting FGF21 analog, leads to a marked elevation in circulating adiponectin [50]. In adiponectin-deficient mice, both acute effects of FGF21 on decreasing hyperglycemia and hyperinsulinemia and chronic effects of FGF21 on alleviation of dietary obesity-induced insulin resistance, glucose intolerance, and fatty liver are severely impaired as compared to those in wild-type mice [44]. Similarly, the effects of FGF21 on reduction of ceramide and induction of energy expenditure are also dependent on adiponectin [45]. Taken together, these findings suggest that the metabolic benefits of FGF21 are mediated at least in part by induction of adiponectin.
- In DKO mice, we found that the exacerbated atherosclerosis caused by FGF21 deficiency is significantly reduced by daily administration with either recombinant FGF21 or adiponectin [33]. However, the magnitude of reduction in atherosclerotic plaque size in adiponectin-treated mice is smaller than that in FGF21-treated mice. Further analysis showed that the effects of FGF21 and adiponectin administration on suppression of collagen composition, smooth muscle proliferation and macrophage infiltration, and reduction in expression of proinflammatory chemokines ICAM-1 and VCAM-1 and cytokines TNFα and monocyte MCP-1 are comparable. Consistently, our in vitro studies demonstrated that adiponectin, but not FGF21, exerts direct effects on suppressing proliferation and migration of human smooth muscle cells and uptake of oxidized LDL in human macrophages. On the other hand, while administration of recombinant FGF21 decreases total cholesterol in DKO mice to a level comparable to apoE-/- mice, whereas adiponectin has no effect on hypercholesterolemia caused by FGF21 deficiency. Therefore, these findings suggest that while adiponectin is critical for the protective effects of FGF21 on blood vessel cells, but is not required for the cholesterol-lowering activity of FGF21. Indeed, adiponectin does not have cholesterol-lowering activity despite its ability to reduce triglyceride [51].
ADIPONECTIN AS A DOWNSTREAM EFFECTOR OF FGF21 IN PROTECTION AGAINST ATHEROSCLEROSIS
- Dyslipidemia, especially elevated LDL-cholesterol, is a major contributor to atherosclerotic plaque formation. The cholesterol-lowering drugs, such as statins, have been used clinically to reduce the risk of atherosclerotic heart disease. Therapeutic administration of FGF21 has been shown to alleviate dyslipidemia in rodents [52], obese monkeys [53], and obese patients with type 2 diabetes [50], including reductions in total and LDL-cholesterol and triglycerides, elevations in HDL-cholesterol and a shift to a less atherogenic apolipoprotein profile.
- Consistent with the aforementioned pharmacological studies, FGF21 deficiency in apoE-/- mice causes a further aggravation of hypercholesterolemia and a shift of apolipoprotein profiles from HDL to LDL, which are accompanied by augmented de novo cholesterol biosynthesis and increased expression of several cholesterologenic genes in the liver, suggesting that endogenous FGF21 is a physiological suppressor of hepatic cholesterol production [33]. Mechanistically, we found that the inhibitory effects of FGF21 on hepatic cholesterol biosynthesis are attributed to its suppression of sterol regulatory element-binding protein 2 (Srebp-2), a member of the basic helix-loop-helix-leucine zipper transcription factor family and a master regulator of cholesterol biosynthesis by preferentially activating the transcription of key cholesterologenic genes [5455]. Adenovirus-mediated silencing of hepatic Srebp-2 expression is sufficient to counteract exacerbation of hypercholesterolemia and augmentation of hepatic cholesterol biosynthesis caused by FGF21 deficiency, whereas the therapeutic benefits of systemic FGF21 administration on inhibition of hepatic cholesterologenesis and reduction of hypercholesterolemia are abrogated by overexpression of Srebp-2. Interestingly, FGF21 specifically suppresses the transcription and activity of Srebp-2, but not Srebp-1, which preferentially activate transcription of genes involved in fatty acid synthesis [33].
- Although FGF21 is predominantly produced from the liver, it is still debatable whether hepatocyte itself is a direct target of FGF21 [356]. While βKlotho is highly expressed in the liver, the expression of FGFR1, which is postulated as a major form of FGF21 receptor in adipocytes, is hardly detectable in this tissue [33]. Despite the fact that hepatic production of FGF21 is markedly induced upon prolonged fasting, FGF21 regulates hepatic gluconeogenesis indirectly via activation of the HPA axis, but not via its direct actions on hepatocytes [323]. On the other hand, direct effects of FGF21 on inhibition of gluconeogenesis and augmentation of fatty acid oxidation have also been reported [5758]. To address whether the regulatory effects of FGF21 on cholesterol homeostasis are attributed to its direct hepatic actions, we generated the βKlotho liver-specific knockout (βKlotho-LKO) mice by intravenous injection of adenovirus-associated virus encoding Cre recombinase into βKlotho-floxed mice [33]. Notably, the effects of FGF21 on reduction of hypercholesterolemia and inhibition of hepatic expression of Srebp-2 and several cholesterologenic genes are largely abrogated in βKlotho-LKO mice, despite a similar level of adiponectin between βKlotho-LKO and wild-type littermates. By using both gain-of-function and loss-of-function experiments, we identified FGFR2, which is highly expressed in hepatocytes, mediates the cholesterol-lowering effect of FGF21 in the liver. These findings collectively support the notion that the cholesterol-lowering effects of FGF21 are attributed to its direct actions on hepatocytes via the FGFR2-βKlotho receptor complex. However, the intracellular signaling pathways that link FGF21-mediated activation of the FGFR2-βKlotho receptor complex with the downstream Srebp-2 expression remain elusive.
LIVER AS A MAJOR TARGET OF FGF21 FOR SUPPRESSION OF HYPERCHOLESTEROLEMIA
- In addition to multiple metabolic benefits of FGF21, accumulating evidence from both animal and clinical studies demonstrate that this hormone also possesses potent protective effects against vascular inflammation and atherosclerotic lesions by mediating the crosstalk among several key metabolic and vascular organs (Fig. 1). First, FGF21 acts on its main target adipose tissues to promote the expression and release of adiponectin possibly by activation of PPARγ, and adiponectin in turn exerts its vascular protective activities via its direct effects on all the major types of cells in the blood vessels. Second, FGF21 alleviates hypercholesterolemia via its autocrine actions in hepatocytes, where it suppresses Srebp-2-mediated cholesterol biosynthesis via the FGFR2-βKlotho receptor complex. In addition, several other effects of FGF21 may also contribute indirectly to the antiatherosclerotic activity of FGF21, including its inhibition of lipolysis, which may in turn prevent lipotoxicity-induced metabolic and vascular damages [59]. Furthermore, FGF21 has been shown to promote the browning of white adipose tissues via either its direct actions in adipocytes or via mediating the crosstalk between brain and adipose tissues [272829]. Notably, transplantation of brown adipocytes has been shown to alleviate atherosclerosis in animals [60]. In humans, the amount and activity of brown adipocytes are inversely correlated with cardiovascular diseases [61].
- Apart from the anti-atherosclerotic activities of FGF21, its cardiac protective effects have also been observed in several recent studies, including prevention of myocardial injury and apoptosis after ischemia reperfusion via FGFR1/βKlotho/Akt signaling cascade in cardiomyocytes [62]. FGF21 has also been reported to prevent cardiomyocytes apoptosis by activation of adiponectin-dependent signaling [63]. Furthermore, FGF21 alleviates diabetic cardiomyopathy by reducing cardiac lipid accumulation [64]. Taken together, these animal-based studies suggest FGF21 analogs represent a promising therapeutic agent for treatment and prevention of a cluster of cardiometabolic disorders. Although FGF21 was initially identified as glucose-lowering agent, a recent clinical in obese patients with type 2 diabetes showed that chronic administration of a long-acting form of FGF21 causes a marked elevation of adiponectin and an obvious reduction in total and LDL-cholesterol, but has little effect on hyperglycemia [50], further supporting the notion that FGF21 or its agonists might be more effective in reducing cardiovascular risks, instead of diabetes.
- Despite the promising results on the cardiovascular protective effects of FGF21, it is important to note that almost all these studies are mainly based on rodent models. In light of the fact that there is a big difference in lipid metabolism and cardiovascular structure between rodents and humans, the pathophysiological relevance of these findings remains to be confirmed in humanoid large animals (such as pigs) and in clinical investigations. Furthermore, our knowledge on the receptor and post-receptor signaling events underlying the multiple physiological and/or pharmacological actions of FGF21 remain largely fragmented, and further extensive studies on FGF21 signaling cascade are needed to assist the rational design of FGF21 or its receptor agonists for future therapeutic applications to reduce the morbidity and mortality of cardiovascular diseases.
CONCLUSIONS
-
Acknowledgements
- This study was supported by Hong Kong Research grant council (HKU783413M), Hong Kong Health and Medical Research Fund (2132836), and matching fund for state key laboratory of pharmaceutical biotechnology from the University of Hong Kong.
ACKNOWLEDGMENTS
-
CONFLICTS OF INTEREST: No potential conflict of interest relevant to this article was reported.
NOTES
- 1. Itoh N. Hormone-like (endocrine) Fgfs: their evolutionary history and roles in development, metabolism, and disease. Cell Tissue Res 2010;342:1-11. ArticlePubMedPMC
- 2. Fukumoto S. Actions and mode of actions of FGF19 subfamily members. Endocr J 2008;55:23-31. ArticlePubMed
- 3. Potthoff MJ, Inagaki T, Satapati S, Ding X, He T, Goetz R, Mohammadi M, Finck BN, Mangelsdorf DJ, Kliewer SA, Burgess SC. FGF21 induces PGC-1alpha and regulates carbohydrate and fatty acid metabolism during the adaptive starvation response. Proc Natl Acad Sci U S A 2009;106:10853-10858. PubMedPMC
- 4. Badman MK, Pissios P, Kennedy AR, Koukos G, Flier JS, Maratos-Flier E. Hepatic fibroblast growth factor 21 is regulated by PPARalpha and is a key mediator of hepatic lipid metabolism in ketotic states. Cell Metab 2007;5:426-437. PubMed
- 5. Ge X, Wang Y, Lam KS, Xu A. Metabolic actions of FGF21: molecular mechanisms and therapeutic implications. Acta Pharm Sin B 2012;2:350-357.Article
- 6. Li H, Gao Z, Zhang J, Ye X, Xu A, Ye J, Jia W. Sodium butyrate stimulates expression of fibroblast growth factor 21 in liver by inhibition of histone deacetylase 3. Diabetes 2012;61:797-806. ArticlePubMedPMCPDF
- 7. Dutchak PA, Katafuchi T, Bookout AL, Choi JH, Yu RT, Mangelsdorf DJ, Kliewer SA. Fibroblast growth factor-21 regulates PPARgamma activity and the antidiabetic actions of thiazolidinediones. Cell 2012;148:556-567. ArticlePubMedPMC
- 8. Kharitonenkov A, Shiyanova TL, Koester A, Ford AM, Micanovic R, Galbreath EJ, Sandusky GE, Hammond LJ, Moyers JS, Owens RA, Gromada J, Brozinick JT, Hawkins ED, Wroblewski VJ, Li DS, Mehrbod F, Jaskunas SR, Shanafelt AB. FGF-21 as a novel metabolic regulator. J Clin Invest 2005;115:1627-1635. ArticlePubMedPMC
- 9. Inagaki T, Dutchak P, Zhao G, Ding X, Gautron L, Parameswara V, Li Y, Goetz R, Mohammadi M, Esser V, Elmquist JK, Gerard RD, Burgess SC, Hammer RE, Mangelsdorf DJ, Kliewer SA. Endocrine regulation of the fasting response by PPARalpha-mediated induction of fibroblast growth factor 21. Cell Metab 2007;5:415-425. PubMed
- 10. Zhang X, Yeung DC, Karpisek M, Stejskal D, Zhou ZG, Liu F, Wong RL, Chow WS, Tso AW, Lam KS, Xu A. Serum FGF21 levels are increased in obesity and are independently associated with the metabolic syndrome in humans. Diabetes 2008;57:1246-1253. ArticlePubMedPDF
- 11. Muise ES, Azzolina B, Kuo DW, El-Sherbeini M, Tan Y, Yuan X, Mu J, Thompson JR, Berger JP, Wong KK. Adipose fibroblast growth factor 21 is up-regulated by peroxisome proliferator-activated receptor gamma and altered metabolic states. Mol Pharmacol 2008;74:403-412. ArticlePubMed
- 12. Wente W, Efanov AM, Brenner M, Kharitonenkov A, Koster A, Sandusky GE, Sewing S, Treinies I, Zitzer H, Gromada J. Fibroblast growth factor-21 improves pancreatic beta-cell function and survival by activation of extracellular signal-regulated kinase 1/2 and Akt signaling pathways. Diabetes 2006;55:2470-2478. PubMed
- 13. Izumiya Y, Bina HA, Ouchi N, Akasaki Y, Kharitonenkov A, Walsh K. FGF21 is an Akt-regulated myokine. FEBS Lett 2008;582:3805-3810. ArticlePubMedPMC
- 14. Suzuki M, Uehara Y, Motomura-Matsuzaka K, Oki J, Koyama Y, Kimura M, Asada M, Komi-Kuramochi A, Oka S, Imamura T. betaKlotho is required for fibroblast growth factor (FGF) 21 signaling through FGF receptor (FGFR) 1c and FGFR3c. Mol Endocrinol 2008;22:1006-1014. PubMedPMC
- 15. Kurosu H, Choi M, Ogawa Y, Dickson AS, Goetz R, Eliseenkova AV, Mohammadi M, Rosenblatt KP, Kliewer SA, Kuro-o M. Tissue-specific expression of betaKlotho and fibroblast growth factor (FGF) receptor isoforms determines metabolic activity of FGF19 and FGF21. J Biol Chem 2007;282:26687-26695. PubMedPMC
- 16. Goetz R, Beenken A, Ibrahimi OA, Kalinina J, Olsen SK, Eliseenkova AV, Xu C, Neubert TA, Zhang F, Linhardt RJ, Yu X, White KE, Inagaki T, Kliewer SA, Yamamoto M, Kurosu H, Ogawa Y, Kuro-o M, Lanske B, Razzaque MS, Mohammadi M. Molecular insights into the klotho-dependent, endocrine mode of action of fibroblast growth factor 19 subfamily members. Mol Cell Biol 2007;27:3417-3428. ArticlePubMedPMCPDF
- 17. Ge X, Chen C, Hui X, Wang Y, Lam KS, Xu A. Fibroblast growth factor 21 induces glucose transporter-1 expression through activation of the serum response factor/Ets-like protein-1 in adipocytes. J Biol Chem 2011;286:34533-34541. ArticlePubMedPMC
- 18. Micanovic R, Raches DW, Dunbar JD, Driver DA, Bina HA, Dickinson CD, Kharitonenkov A. Different roles of N- and C- termini in the functional activity of FGF21. J Cell Physiol 2009;219:227-234. ArticlePubMed
- 19. Fon Tacer K, Bookout AL, Ding X, Kurosu H, John GB, Wang L, Goetz R, Mohammadi M, Kuro-o M, Mangelsdorf DJ, Kliewer SA. Research resource: comprehensive expression atlas of the fibroblast growth factor system in adult mouse. Mol Endocrinol 2010;24:2050-2064. ArticlePubMedPMC
- 20. Ogawa Y, Kurosu H, Yamamoto M, Nandi A, Rosenblatt KP, Goetz R, Eliseenkova AV, Mohammadi M, Kuro-o M. BetaKlotho is required for metabolic activity of fibroblast growth factor 21. Proc Natl Acad Sci U S A 2007;104:7432-7437. PubMedPMC
- 21. Chau MD, Gao J, Yang Q, Wu Z, Gromada J. Fibroblast growth factor 21 regulates energy metabolism by activating the AMPK-SIRT1-PGC-1alpha pathway. Proc Natl Acad Sci U S A 2010;107:12553-12558. PubMedPMC
- 22. Kim KH, Lee MS. FGF21 as a stress hormone: the roles of FGF21 in stress adaptation and the treatment of metabolic diseases. Diabetes Metab J 2014;38:245-251. ArticlePubMedPMC
- 23. Liang Q, Zhong L, Zhang J, Wang Y, Bornstein SR, Triggle CR, Ding H, Lam KS, Xu A. FGF21 maintains glucose homeostasis by mediating the cross talk between liver and brain during prolonged fasting. Diabetes 2014;63:4064-4075. ArticlePubMedPDF
- 24. Owen BM, Ding X, Morgan DA, Coate KC, Bookout AL, Rahmouni K, Kliewer SA, Mangelsdorf DJ. FGF21 acts centrally to induce sympathetic nerve activity, energy expenditure, and weight loss. Cell Metab 2014;20:670-677. ArticlePubMedPMC
- 25. Ye D, Wang Y, Li H, Jia W, Man K, Lo CM, Wang Y, Lam KS, Xu A. Fibroblast growth factor 21 protects against acetaminophen-induced hepatotoxicity by potentiating peroxisome proliferator-activated receptor coactivator protein-1alpha-mediated antioxidant capacity in mice. Hepatology 2014;60:977-989. ArticlePubMed
- 26. Kim KH, Jeong YT, Oh H, Kim SH, Cho JM, Kim YN, Kim SS, Kim do H, Hur KY, Kim HK, Ko T, Han J, Kim HL, Kim J, Back SH, Komatsu M, Chen H, Chan DC, Konishi M, Itoh N, Choi CS, Lee MS. Autophagy deficiency leads to protection from obesity and insulin resistance by inducing Fgf21 as a mitokine. Nat Med 2013;19:83-92. ArticlePubMedPDF
- 27. Chartoumpekis DV, Habeos IG, Ziros PG, Psyrogiannis AI, Kyriazopoulou VE, Papavassiliou AG. Brown adipose tissue responds to cold and adrenergic stimulation by induction of FGF21. Mol Med 2011;17:736-740. ArticlePubMedPMCPDF
- 28. Fisher FM, Kleiner S, Douris N, Fox EC, Mepani RJ, Verdeguer F, Wu J, Kharitonenkov A, Flier JS, Maratos-Flier E, Spiegelman BM. FGF21 regulates PGC-1alpha and browning of white adipose tissues in adaptive thermogenesis. Genes Dev 2012;26:271-281. ArticlePubMedPMC
- 29. Douris N, Stevanovic DM, Fisher FM, Cisu TI, Chee MJ, Nguyen NL, Zarebidaki E, Adams AC, Kharitonenkov A, Flier JS, Bartness TJ, Maratos-Flier E. Central fibroblast growth factor 21 browns white fat via sympathetic action in male mice. Endocrinology 2015;156:2470-2481. ArticlePubMedPMCPDF
- 30. Chow WS, Xu A, Woo YC, Tso AW, Cheung SC, Fong CH, Tse HF, Chau MT, Cheung BM, Lam KS. Serum fibroblast growth factor-21 levels are associated with carotid atherosclerosis independent of established cardiovascular risk factors. Arterioscler Thromb Vasc Biol 2013;33:2454-2459. ArticlePubMed
- 31. Zhang X, Hu Y, Zeng H, Li L, Zhao J, Zhao J, Liu F, Bao Y, Jia W. Serum fibroblast growth factor 21 levels is associated with lower extremity atherosclerotic disease in Chinese female diabetic patients. Cardiovasc Diabetol 2015;14:32ArticlePubMedPMCPDF
- 32. Xiao Y, Liu L, Xu A, Zhou P, Long Z, Tu Y, Chen X, Tang W, Huang G, Zhou Z. Serum fibroblast growth factor 21 levels are related to subclinical atherosclerosis in patients with type 2 diabetes. Cardiovasc Diabetol 2015;14:72ArticlePubMedPMCPDF
- 33. Lin Z, Pan X, Wu F, Ye D, Zhang Y, Wang Y, Jin L, Lian Q, Huang Y, Ding H, Triggle C, Wang K, Li X, Xu A. Fibroblast growth factor 21 prevents atherosclerosis by suppression of hepatic sterol regulatory element-binding protein-2 and induction of adiponectin in mice. Circulation 2015;131:1861-1871. ArticlePubMedPMC
- 34. Zhu W, Cheng KK, Vanhoutte PM, Lam KS, Xu A. Vascular effects of adiponectin: molecular mechanisms and potential therapeutic intervention. Clin Sci (Lond) 2008;114:361-374. ArticlePubMedPDF
- 35. Arita Y, Kihara S, Ouchi N, Takahashi M, Maeda K, Miyagawa J, Hotta K, Shimomura I, Nakamura T, Miyaoka K, Kuriyama H, Nishida M, Yamashita S, Okubo K, Matsubara K, Muraguchi M, Ohmoto Y, Funahashi T, Matsuzawa Y. Paradoxical decrease of an adipose-specific protein, adiponectin, in obesity. Biochem Biophys Res Commun 1999;257:79-83. ArticlePubMed
- 36. Koenig W, Khuseyinova N, Baumert J, Meisinger C, Lowel H. Serum concentrations of adiponectin and risk of type 2 diabetes mellitus and coronary heart disease in apparently healthy middle-aged men: results from the 18-year follow-up of a large cohort from southern Germany. J Am Coll Cardiol 2006;48:1369-1377. PubMed
- 37. Hui X, Lam KS, Vanhoutte PM, Xu A. Adiponectin and cardiovascular health: an update. Br J Pharmacol 2012;165:574-590. ArticlePubMedPMC
- 38. Xu A, Vanhoutte PM. Adiponectin and adipocyte fatty acid binding protein in the pathogenesis of cardiovascular disease. Am J Physiol Heart Circ Physiol 2012;302:H1231-H1240. ArticlePubMed
- 39. Cheng KK, Lam KS, Wang Y, Huang Y, Carling D, Wu D, Wong C, Xu A. Adiponectin-induced endothelial nitric oxide synthase activation and nitric oxide production are mediated by APPL1 in endothelial cells. Diabetes 2007;56:1387-1394. ArticlePubMedPDF
- 40. Ouedraogo R, Wu X, Xu SQ, Fuchsel L, Motoshima H, Mahadev K, Hough K, Scalia R, Goldstein BJ. Adiponectin suppression of high-glucose-induced reactive oxygen species in vascular endothelial cells: evidence for involvement of a cAMP signaling pathway. Diabetes 2006;55:1840-1846. PubMed
- 41. Ouchi N, Kihara S, Arita Y, Nishida M, Matsuyama A, Okamoto Y, Ishigami M, Kuriyama H, Kishida K, Nishizawa H, Hotta K, Muraguchi M, Ohmoto Y, Yamashita S, Funahashi T, Matsuzawa Y. Adipocyte-derived plasma protein, adiponectin, suppresses lipid accumulation and class A scavenger receptor expression in human monocyte-derived macrophages. Circulation 2001;103:1057-1063. ArticlePubMed
- 42. Wang Y, Lam KS, Xu JY, Lu G, Xu LY, Cooper GJ, Xu A. Adiponectin inhibits cell proliferation by interacting with several growth factors in an oligomerization-dependent manner. J Biol Chem 2005;280:18341-18347. ArticlePubMed
- 43. Chang J, Li Y, Huang Y, Lam KS, Hoo RL, Wong WT, Cheng KK, Wang Y, Vanhoutte PM, Xu A. Adiponectin prevents diabetic premature senescence of endothelial progenitor cells and promotes endothelial repair by suppressing the p38 MAP kinase/p16INK4A signaling pathway. Diabetes 2010;59:2949-2959. ArticlePubMedPMCPDF
- 44. Lin Z, Tian H, Lam KS, Lin S, Hoo RC, Konishi M, Itoh N, Wang Y, Bornstein SR, Xu A, Li X. Adiponectin mediates the metabolic effects of FGF21 on glucose homeostasis and insulin sensitivity in mice. Cell Metab 2013;17:779-789. ArticlePubMed
- 45. Unger RH, Scherer PE, Holland WL. Dichotomous roles of leptin and adiponectin as enforcers against lipotoxicity during feast and famine. Mol Biol Cell 2013;24:3011-3015. ArticlePubMedPMC
- 46. Wang Y, Lam KS, Xu A. Adiponectin as a negative regulator in obesity-related mammary carcinogenesis. Cell Res 2007;17:280-282. ArticlePubMedPDF
- 47. Iwaki M, Matsuda M, Maeda N, Funahashi T, Matsuzawa Y, Makishima M, Shimomura I. Induction of adiponectin, a fat-derived antidiabetic and antiatherogenic factor, by nuclear receptors. Diabetes 2003;52:1655-1663. ArticlePubMedPDF
- 48. Wang ZV, Schraw TD, Kim JY, Khan T, Rajala MW, Follenzi A, Scherer PE. Secretion of the adipocyte-specific secretory protein adiponectin critically depends on thiol-mediated protein retention. Mol Cell Biol 2007;27:3716-3731. ArticlePubMedPMCPDF
- 49. Liu M, Zhou L, Xu A, Lam KS, Wetzel MD, Xiang R, Zhang J, Xin X, Dong LQ, Liu F. A disulfide-bond A oxidoreductase-like protein (DsbA-L) regulates adiponectin multimerization. Proc Natl Acad Sci U S A 2008;105:18302-18307. ArticlePubMedPMC
- 50. Gaich G, Chien JY, Fu H, Glass LC, Deeg MA, Holland WL, Kharitonenkov A, Bumol T, Schilske HK, Moller DE. The effects of LY2405319, an FGF21 analog, in obese human subjects with type 2 diabetes. Cell Metab 2013;18:333-340. ArticlePubMed
- 51. Fruebis J, Tsao TS, Javorschi S, Ebbets-Reed D, Erickson MR, Yen FT, Bihain BE, Lodish HF. Proteolytic cleavage product of 30-kDa adipocyte complement-related protein increases fatty acid oxidation in muscle and causes weight loss in mice. Proc Natl Acad Sci U S A 2001;98:2005-2010. ArticlePubMedPMC
- 52. Xu J, Lloyd DJ, Hale C, Stanislaus S, Chen M, Sivits G, Vonderfecht S, Hecht R, Li YS, Lindberg RA, Chen JL, Jung DY, Zhang Z, Ko HJ, Kim JK, Veniant MM. Fibroblast growth factor 21 reverses hepatic steatosis, increases energy expenditure, and improves insulin sensitivity in diet-induced obese mice. Diabetes 2009;58:250-259. ArticlePubMedPMCPDF
- 53. Veniant MM, Komorowski R, Chen P, Stanislaus S, Winters K, Hager T, Zhou L, Wada R, Hecht R, Xu J. Long-acting FGF21 has enhanced efficacy in diet-induced obese mice and in obese rhesus monkeys. Endocrinology 2012;153:4192-4203. ArticlePubMedPDF
- 54. Hua X, Yokoyama C, Wu J, Briggs MR, Brown MS, Goldstein JL, Wang X. SREBP-2, a second basic-helix-loop-helix-leucine zipper protein that stimulates transcription by binding to a sterol regulatory element. Proc Natl Acad Sci U S A 1993;90:11603-11607. ArticlePubMedPMC
- 55. Horton JD, Goldstein JL, Brown MS. SREBPs: activators of the complete program of cholesterol and fatty acid synthesis in the liver. J Clin Invest 2002;109:1125-1131. ArticlePubMedPMC
- 56. Adams AC, Astapova I, Fisher FM, Badman MK, Kurgansky KE, Flier JS, Hollenberg AN, Maratos-Flier E. Thyroid hormone regulates hepatic expression of fibroblast growth factor 21 in a PPARalpha-dependent manner. J Biol Chem 2010;285:14078-14082. PubMedPMC
- 57. Li Y, Wong K, Giles A, Jiang J, Lee JW, Adams AC, Kharitonenkov A, Yang Q, Gao B, Guarente L, Zang M. Hepatic SIRT1 attenuates hepatic steatosis and controls energy balance in mice by inducing fibroblast growth factor 21. Gastroenterology 2014;146:539-549.e7. ArticlePubMed
- 58. Fisher FM, Estall JL, Adams AC, Antonellis PJ, Bina HA, Flier JS, Kharitonenkov A, Spiegelman BM, Maratos-Flier E. Integrated regulation of hepatic metabolism by fibroblast growth factor 21 (FGF21) in vivo. Endocrinology 2011;152:2996-3004. ArticlePubMedPMCPDF
- 59. Chen W, Hoo RL, Konishi M, Itoh N, Lee PC, Ye HY, Lam KS, Xu A. Growth hormone induces hepatic production of fibroblast growth factor 21 through a mechanism dependent on lipolysis in adipocytes. J Biol Chem 2011;286:34559-34566. ArticlePubMedPMC
- 60. Kikai M, Terada K, Yamamoto K, Motoyama S, Wada N, Wakana N, Yamada H. Transplantation of brown adipose tissue inhibits atherosclerosis in apoE deficient mice via activation of Fgf-21/adiponectin axis. Circulation 2015;132:A13907.
- 61. Bartelt A, Bruns OT, Reimer R, Hohenberg H, Ittrich H, Peldschus K, Kaul MG, Tromsdorf UI, Weller H, Waurisch C, Eychmuller A, Gordts PL, Rinninger F, Bruegelmann K, Freund B, Nielsen P, Merkel M, Heeren J. Brown adipose tissue activity controls triglyceride clearance. Nat Med 2011;17:200-205. ArticlePubMedPDF
- 62. Liu SQ, Roberts D, Kharitonenkov A, Zhang B, Hanson SM, Li YC, Zhang LQ, Wu YH. Endocrine protection of ischemic myocardium by FGF21 from the liver and adipose tissue. Sci Rep 2013;3:2767ArticlePubMedPMCPDF
- 63. Joki Y, Ohashi K, Yuasa D, Shibata R, Ito M, Matsuo K, Kambara T, Uemura Y, Hayakawa S, Hiramatsu-Ito M, Kanemura N, Ogawa H, Daida H, Murohara T, Ouchi N. FGF21 attenuates pathological myocardial remodeling following myocardial infarction through the adiponectin-dependent mechanism. Biochem Biophys Res Commun 2015;459:124-130. ArticlePubMed
- 64. Yan X, Chen J, Zhang C, Zhou S, Zhang Z, Chen J, Feng W, Li X, Tan Y. FGF21 deletion exacerbates diabetic cardiomyopathy by aggravating cardiac lipid accumulation. J Cell Mol Med 2015;19:1557-1568. ArticlePubMedPMCPDF
REFERENCES
Figure & Data
References
Citations

- Fibroblast Growth Factor–Based Pharmacotherapies for the Treatment of Obesity-Related Metabolic Complications
Leigang Jin, Ranyao Yang, Leiluo Geng, Aimin Xu
Annual Review of Pharmacology and Toxicology.2023; 63(1): 359. CrossRef - Efruxifermin, an investigational treatment for fibrotic or cirrhotic nonalcoholic steatohepatitis (NASH)
Tobias Puengel, Frank Tacke
Expert Opinion on Investigational Drugs.2023; 32(6): 451. CrossRef - The role of perivascular adipose tissue-secreted adipocytokines in cardiovascular disease
Meichao Cai, Dongsheng Zhao, Xiao Han, Shuang Han, Wenxin Zhang, Zhennan Zang, Chenchen Gai, Rong Rong, Tian Gao
Frontiers in Immunology.2023;[Epub] CrossRef - Pharmacological treatment with FGF21 strongly improves plasma cholesterol metabolism to reduce atherosclerosis
Cong Liu, Milena Schönke, Enchen Zhou, Zhuang Li, Sander Kooijman, Mariëtte R Boon, Mikael Larsson, Kristina Wallenius, Niek Dekker, Louise Barlind, Xiao-Rong Peng, Yanan Wang, Patrick C N Rensen
Cardiovascular Research.2022; 118(2): 489. CrossRef - Relationship between Serum FGF21 and vWF Expression and Carotid Atherosclerosis in Elderly Patients with Hypertension
Jing Bian, Lairong Chen, Qin Li, Yunfeng Zhao, Delu Yin, Shanhong Sun, Bhagyaveni M.A
Journal of Healthcare Engineering.2022; 2022: 1. CrossRef - Serum FGF21 Levels Predict the MACE in Patients With Myocardial Infarction After Coronary Artery Bypass Graft Surgery
Wei Xie, Dan Li, Yaru Shi, Ning Yu, Yu Yan, Yingchao Zhang, Qiongli Yu, Yulin Li, Jie Du, Zhuofeng Lin, Fan Wu
Frontiers in Cardiovascular Medicine.2022;[Epub] CrossRef - Adipokines, adiposity, and atherosclerosis
Longhua Liu, Zunhan Shi, Xiaohui Ji, Wenqian Zhang, Jinwen Luan, Tarik Zahr, Li Qiang
Cellular and Molecular Life Sciences.2022;[Epub] CrossRef - Current understanding and controversies on the clinical implications of fibroblast growth factor 21
Yasaman Badakhshi, Tianru Jin
Critical Reviews in Clinical Laboratory Sciences.2021; 58(5): 311. CrossRef - The potential role of plasma fibroblast growth factor 21 as a diagnostic biomarker for abdominal aortic aneurysm presence and development
Ting Xie, Liangying Yin, Dan Guo, Zixin Zhang, Yuexin Chen, Bao Liu, Wei Wang, Yuehong Zheng
Life Sciences.2021; 274: 119346. CrossRef - Ultrasound-assisted C3F8-filled PLGA nanobubbles for enhanced FGF21 delivery and improved prophylactic treatment of diabetic cardiomyopathy
Jiameng Gao, Jingjing Liu, Zheying Meng, Yanming Li, Yuping Hong, Lirui Wang, Lan He, Bing Hu, Yuanyi Zheng, Tianliang Li, Daxiang Cui, E. Shen
Acta Biomaterialia.2021; 130: 395. CrossRef - Effect of Fibroblast Growth Factor 21 on the Development of Atheromatous Plaque and Lipid Metabolic Profiles in an Atherosclerosis-Prone Mouse Model
Hyo Jin Maeng, Gha Young Lee, Jae Hyun Bae, Soo Lim
International Journal of Molecular Sciences.2020; 21(18): 6836. CrossRef - The therapeutic potential of FGF21 in metabolic diseases: from bench to clinic
Leiluo Geng, Karen S. L. Lam, Aimin Xu
Nature Reviews Endocrinology.2020; 16(11): 654. CrossRef - Two-hundred-liter scale fermentation, purification of recombinant human fibroblast growth factor-21, and its anti-diabetic effects on ob/ob mice
Qi Hui, Zhen Huang, Shucai Pang, Xuanxin Yang, Jinghang Li, Bingjie Yu, Lu Tang, Xiaokun Li, Xiaojie Wang
Applied Microbiology and Biotechnology.2019; 103(2): 719. CrossRef - Fibroblast growth factor 21: A role in cardiometabolic disorders and cardiovascular risk prediction?
Niki Katsiki, Christos Mantzoros
Metabolism.2019; 93: iii. CrossRef - Heparin‐poloxamer hydrogel‐encapsulated rhFGF21 enhances wound healing in diabetic mice
Huan Liu, Yeli Zhao, Yuchi Zou, Wenting Huang, Liyun Zhu, Fei Liu, Dongxue Wang, Kaiming Guo, Jian Hu, Jun Chen, Lixia Ye, Xiaokun Li, Li Lin
The FASEB Journal.2019; 33(9): 9858. CrossRef - Fibroblast growth factor 21 association with subclinical atherosclerosis and arterial stiffness in type 2 diabetes
Saeed Yafei, Fathy Elsewy, Eman Youssef, Mohammed Ayman, Mohamed El-Shafei
Diabetes & Metabolic Syndrome: Clinical Research & Reviews.2019; 13(1): 882. CrossRef - Myokines in metabolic homeostasis and diabetes
Jürgen Eckel
Diabetologia.2019; 62(9): 1523. CrossRef - Inhibition of vascular neointima hyperplasia by FGF21 associated with FGFR1/Syk/NLRP3 inflammasome pathway in diabetic mice
Wei Wei, Xiao-Xue Li, Ming Xu
Atherosclerosis.2019; 289: 132. CrossRef - LY2405319, an analog of fibroblast growth factor 21 ameliorates α-smooth muscle actin production through inhibition of the succinate—G-protein couple receptor 91 (GPR91) pathway in mice
Cong Thuc Le, Giang Nguyen, So Young Park, Dae Hee Choi, Eun-Hee Cho, Partha Mukhopadhyay
PLOS ONE.2018; 13(2): e0192146. CrossRef - Relationship of Selected Adipokines with Markers of Vascular Damage in Patients with Type 2 Diabetes
Jaromíra Spurná, David Karásek, Veronika Kubíčková, Dominika Goldmannová, Ondřej Krystyník, Jan Schovánek, Josef Zadražil
Metabolic Syndrome and Related Disorders.2018; 16(5): 246. CrossRef - Serum FGF21 Is Associated with Future Cardiovascular Events in Patients with Coronary Artery Disease
Yun Shen, Xueli Zhang, Yiting Xu, Qin Xiong, Zhigang Lu, Xiaojing Ma, Yuqian Bao, Weiping Jia
Cardiology.2018; 139(4): 212. CrossRef - Relationship between Circulating FGF21 Concentrations and the Severity of Coronary Artery Damage in Subjects with Cardiovascular Disease
Sung Don Park, Kwi-Hyun Bae, Yeon-Kyung Choi, Jae-Han Jeon, Jung Beom Seo, Namkyun Kim, Chang-Yeon Kim, Sung Woo Kim, Won Kee Lee, Jung Guk Kim, In-Kyu Lee, Jang Hoon Lee, Keun-Gyu Park
Journal of Lipid and Atherosclerosis.2018; 7(1): 42. CrossRef - Fibroblast growth factor 21 regulates foam cells formation and inflammatory response in Ox-LDL-induced THP-1 macrophages
Nan Wang, Jun-yan Li, Shuai Li, Xiao-chen Guo, Tong Wu, Wen-fei Wang, De-shan Li
Biomedicine & Pharmacotherapy.2018; 108: 1825. CrossRef - Chip‐based high resolution tandem mass spectrometric determination of fibroblast growth factor—chondroitin sulfate disaccharides noncovalent interaction
Adrian C. Robu, Laurentiu Popescu, Daniela G. Seidler, Alina D. Zamfir
Journal of Mass Spectrometry.2018; 53(7): 624. CrossRef - Conditioned medium from contracting skeletal muscle cells reverses insulin resistance and dysfunction of endothelial cells
Yihe Zhao, Nana Li, Zhu Li, Da Zhang, Liming Chen, Zhi Yao, Wenyan Niu
Metabolism.2018; 82: 36. CrossRef - Dietary n-3 long-chain polyunsaturated fatty acids upregulate energy dissipating metabolic pathways conveying anti-obesogenic effects in mice
Stefanie Worsch, Mathias Heikenwalder, Hans Hauner, Bernhard L. Bader
Nutrition & Metabolism.2018;[Epub] CrossRef - Metabolic Values of Fibroblasts Growth Factor 21 in Patients with Coronary Artery Disease and Obesity
L. M. Pasiyeshvili, K. V. Ivanova
Ukraïnsʹkij žurnal medicini, bìologìï ta sportu.2018; 3(4): 110. CrossRef - FGF21 activates AMPK signaling: impact on metabolic regulation and the aging process
Antero Salminen, Anu Kauppinen, Kai Kaarniranta
Journal of Molecular Medicine.2017; 95(2): 123. CrossRef - Regulation of longevity by FGF21: Interaction between energy metabolism and stress responses
Antero Salminen, Kai Kaarniranta, Anu Kauppinen
Ageing Research Reviews.2017; 37: 79. CrossRef - Contribution of serum FGF21 level to the identification of left ventricular systolic dysfunction and cardiac death
Yun Shen, Xueli Zhang, Xiaoping Pan, Yiting Xu, Qin Xiong, Zhigang Lu, Xiaojing Ma, Yuqian Bao, Weiping Jia
Cardiovascular Diabetology.2017;[Epub] CrossRef - Fibroblast growth factor 21 – a key player in cardiovascular disorders?
Monika Lenart-Lipińska, Dariusz Duma, Magdalena Hałabiś, Marcin Dziedzic, Janusz Solski
Hormone Molecular Biology and Clinical Investigation.2017;[Epub] CrossRef - The role of fibroblast growth factor 21 in atherosclerosis
John Kokkinos, Shudi Tang, Kerry-Anne Rye, Kwok Leung Ong
Atherosclerosis.2017; 257: 259. CrossRef

 KDA
KDA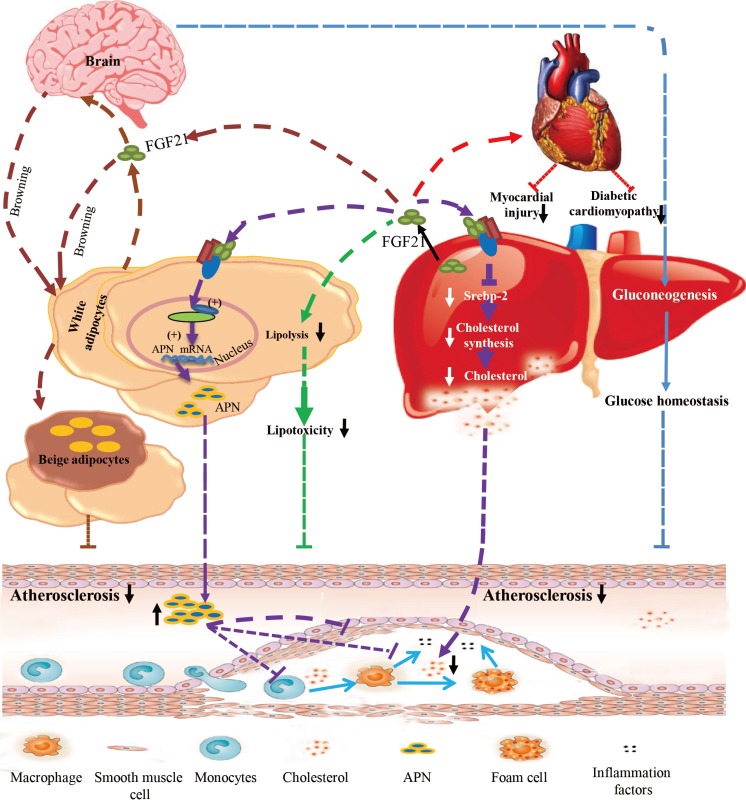
 PubReader
PubReader Cite
Cite




