- Current
- Browse
- Collections
-
For contributors
- For Authors
- Instructions to authors
- Article processing charge
- e-submission
- For Reviewers
- Instructions for reviewers
- How to become a reviewer
- Best reviewers
- For Readers
- Readership
- Subscription
- Permission guidelines
- About
- Editorial policy
Articles
- Page Path
- HOME > Diabetes Metab J > Volume 38(4); 2014 > Article
-
ReviewPathophysiology A Novel Therapeutic Agent for Type 2 Diabetes Mellitus: SGLT2 Inhibitor
- Chang Hee Jung, Jung Eun Jang, Joong-Yeol Park
-
Diabetes & Metabolism Journal 2014;38(4):261-273.
DOI: https://doi.org/10.4093/dmj.2014.38.4.261
Published online: August 20, 2014
Department of Internal Medicine, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea.
- Corresponding author: Joong-Yeol Park. Department of Internal Medicine, Asan Medical Center, University of Ulsan College of Medicine, 88 Olympic-ro 43-gil, Songpa-gu, Seoul 138-736, Korea. jypark@amc.seoul.kr
Copyright © 2014 Korean Diabetes Association
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
ABSTRACT
- Type 2 diabetes mellitus (T2DM) is a complex endocrine and metabolic disorder, and a major public health problem that is rapidly increasing in prevalence. Although a wide range of pharmacotherapies for glycemic control is now available, management of T2DM remains complex and challenging. The kidneys contribute immensely to glucose homeostasis by reabsorbing glucose from the glomerular filtrate. Sodium-glucose cotransporter 2 (SGLT2) inhibitors, a new class of antidiabetic agents that inhibit glucose absorption from the kidney independent of insulin, offer a unique opportunity to improve the outcomes of patients with T2DM. In this review, we provide an overview of two globally-approved SGLT2 inhibitors, dapagliflozin and canagliflozin, and discuss their effects and safety. This information will help clinicians to decide whether these drugs will benefit their patients.
- Type 2 diabetes mellitus (T2DM), a complex endocrine and metabolic disorder, is a major public health problem that is rapidly increasing in prevalence worldwide [1]. Although comprehensive diabetes management is important, glycemic control is essential for effective diabetes management because lowering the level of glycated hemoglobin (HbA1c) to below or around 7% can reduce microvascular complications and, if implemented soon after the diagnosis of diabetes, is associated with long-term reduction in macrovascular disease [2,3]. A wide range of pharmacotherapies for glycemic control is now available; however, management of T2DM remains complex and challenging due to its variable pathogenesis, progressive natural history, and limiting side effects of current therapies, including weight gain, hypoglycemia, fluid retention, and gastrointestinal side effects [4]. Furthermore, our current approach to treating hyperglycemia in long-standing diabetes with either established diabetes-associated complications or multiple cardiovascular disease (CVD) risk factors, does not affect the CVD risk [5,6]. Thus, the quest to develop therapeutic agents with novel mechanisms of action, which are expected to fulfill the unmet needs of current therapies, continues.
- Although several novel therapies for T2DM are on the horizon, orally administered sodium-glucose cotransporter 2 (SGLT2) inhibitors, a new class of antidiabetic agents that inhibit glucose absorption from the kidney independent of insulin, are promising. They offer a unique opportunity to address the currently unmet therapeutic needs of T2DM patients and to improve their outcomes [7]. The approval by the U.S. Food and Drug Administration (FDA) of two SGLT2 inhibitors, canagliflozin and dapagliflozin, with several others in late-stage clinical development, represents an important step forward in the treatment of T2DM because these drugs may be effective for adults with a high risk of T2DM [8,9]. To determine which patients will benefit most from these drugs, clinicians will have to understand the link between the kidney and glucose homeostasis. This review will focus on the most clinically relevant information on SGLT2 inhibitors. We will also provide the evidence to support their safety and discuss the side effects resulting from their use so that clinicians can prescribe these drugs with confidence.
INTRODUCTION
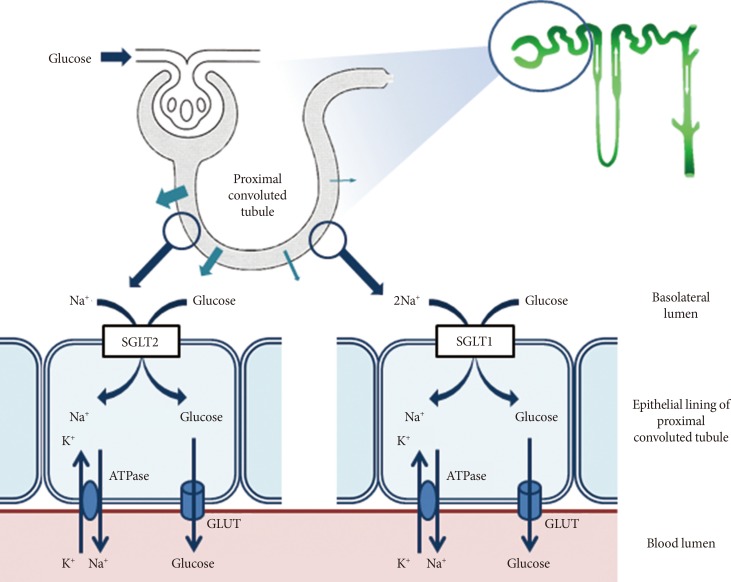
- The kidneys contribute to glucose homeostasis through several mechanisms, including gluconeogenesis, glucose use, and glucose reabsorption from the glomerular filtrate [10]. During glucose reabsorption, for example, approximately 180 L of plasma is filtered daily through the kidneys, which is equivalent to approximately 180 g of glucose, if the average plasma glucose concentration is 100 mg/dL [11]. Under normal physiological conditions, this filtered glucose is almost completely reabsorbed by renal tubular epithelial cells; thus, there is no glucose in urine [11,12]. The transport of glucose into renal tubular epithelial cells is mediated by active cotransporters, the SGLT, a family of ATP-dependent proteins involved in the transport of glucose against a concentration gradient with simultaneous transport of Na+ down a concentration gradient [12]. Although six different SGLT genes have been identified in humans, only SGLT1 and SGLT2 have been well characterized, and their roles in glucose transport in the gut and kidney, respectively, have been defined [12,13]. As shown in Fig. 1, the majority of the filtered glucose is reabsorbed through SGLT2, a low-affinity high-capacity transporter located predominantly in the S1 segment of the renal proximal tubule [7,12]. The remainder is reabsorbed through SGLT1, a high-affinity low-capacity transporter located in the S2 and S3 segments of the renal proximal tubule [7,12]. SGLT1 is also involved in glucose absorption from the gastrointestinal tract [7,12]. Table 1 presents the anatomical locations and biochemical characteristics of SGLT1 and SGLT2. Once the filtered glucose is reabsorbed from the renal proximal tubule via SGLT2 and SGLT1, it is then transported passively into the interstitium through the action of facilitative glucose transporters (GLUTs), such as GLUT1 and GLUT2, at the basolateral membrane of epithelial cells lining the proximal tubule (Fig. 1) [7,12].
RENAL GLUCOSE HANDLING: SGLT1 VERSUS SGLT2
- Perceiving the kidney as a potential treatment target and 'ally' in reducing hyperglycemia represents a fundamental change in the perspective for an organ that has been historically thought to be a 'victim' (nephropathy) in diabetes [14]. However, an improved understanding of the kidney's role in glucose homeostasis has prompted the development of novel drugs that reduce the renal reabsorption of glucose, thereby combating hyperglycemia.
- The filtered glucose load is the product of the plasma glucose concentration and the glomerular filtration rate (GFR). Therefore, as the plasma glucose concentration increases, the filtered glucose load also increases in a linear manner [7,12]. When the reabsorption capacity of the proximal tubule is surpassed, as occurs during hyperglycemia, glucosuria starts to appear [7,12]. This maximum reabsorption capacity is called as 'the maximum transport rate (Tm)' [7,12]. In healthy individuals without diabetes, this Tm for glucose is reached at blood glucose concentrations of approximately 200 mg/dL [15]. However, studies in humans with diabetes and experimental animal models of diabetes have consistently reported an increase in the rate of glucose reabsorption in the proximal tubule (i.e., an increase in Tm for glucose) [16,17]. The mean Tm for glucose has been reported to be higher by 20% or more in diabetic patients compared with healthy individuals [18]. Furthermore, an increase in SGLT2 gene expression has been shown to be one of the molecular mechanisms responsible for this increase in Tm for glucose during hyperglycemia [16,19]. During hyperglycemia, the kidney excretes the excess filtered glucose load to restore normoglycemia. By contrast, the diabetic kidney increases the Tm for glucose, thereby exacerbating hyperglycemia by curtailing glucosuria. This attenuated glucosuria, which results from an increase in Tm for glucose in patients with diabetes, might represent a maladaptation to hyperglycemia and underlie the pathogenesis of hyperglycemia [20]. In light of these pathophysiological findings, the abrogation of hyperglycemia and the reduction of renal glucose absorption through the inhibition of SLGTs is a reasonable approach to treat T2DM. Fig. 2 shows the renal glucose handling before and after SGLT2 inhibition [21].
RATIONALE FOR THE INHIBITION OF RENAL GLUCOSE UPTAKE
- Phlorizin: proof of concept
- Inhibition of SGLT2 has long been regarded as a potential treatment approach for hyperglycemia during T2DM [22]. Phlorizin, a member of the chalcone class of organic compounds, is the prototype SGLT2 inhibitor [22]. It is a naturally-occurring glucoside found in various plants such as in the root bark of the apple tree. It was first isolated in 1835 by a French chemist and subsequently used to treat fever and infectious diseases, particularly malaria [22]. Following the observation that phlorizin induces glucosuria, it became useful in the study of renal function in humans, as the ability of phlorizin to induce glucosuria is indicative of a 'Sound kidney' [22].
- Studies from the 1950s have shown that phlorizin blocks glucose transport in several tissues, including the kidney and small intestine, by inhibiting SGLT proteins [23]. Treatment of partially pancreatectomized diabetic rats with phlorizin induces glucosuria and lowers the blood glucose level [24]. Interestingly, the insulin resistance in these animals also improved [24]. Taken collectively, these findings support the important proof of concept that diabetic patients can benefit from phlorizin treatment.
- Although phlorizin inhibits the increase in the blood glucose level in mice treated with a glucose solution, it was not further developed as a therapeutic for T2DM due to several reasons [22]. The reasons include its low selectivity for SGLT2 versus SGLT1, the presence of an active metabolite, and its low oral bioavailability due to poor intestinal absorption [22]. Inhibition of SGLT1 can also result in adverse gastrointestinal effects such as severe diarrhea, dehydration, and malabsorption [25]. Phloretin, the active metabolite of phlorizin, may also produce adverse effects by inhibiting other GLUTs such as GLUT1 and GLUT2 [26]. Due to these limitations, other compounds with greater oral bioavailability and higher selectivity for SGLT2 have been developed. Fortunately, the benign phenomenon observed in subjects with familial renal glucosuria, the genetic disorder caused by mutations in the SGLT2 gene, indicates that the selective inhibition of SGLT2 may be safe [27]. Currently, there are two SGLT2 inhibitors, dapagliflozin and canagliflozin, that are approved globally. Besides these two inhibitors, other SGLT2 inhibitors are currently under development or undergoing clinical trials. In this review, we will discuss the phase III clinical data of dapagliflozin and canagliflozin, with emphasis on their safety and efficacy.
- Dapagliflozin
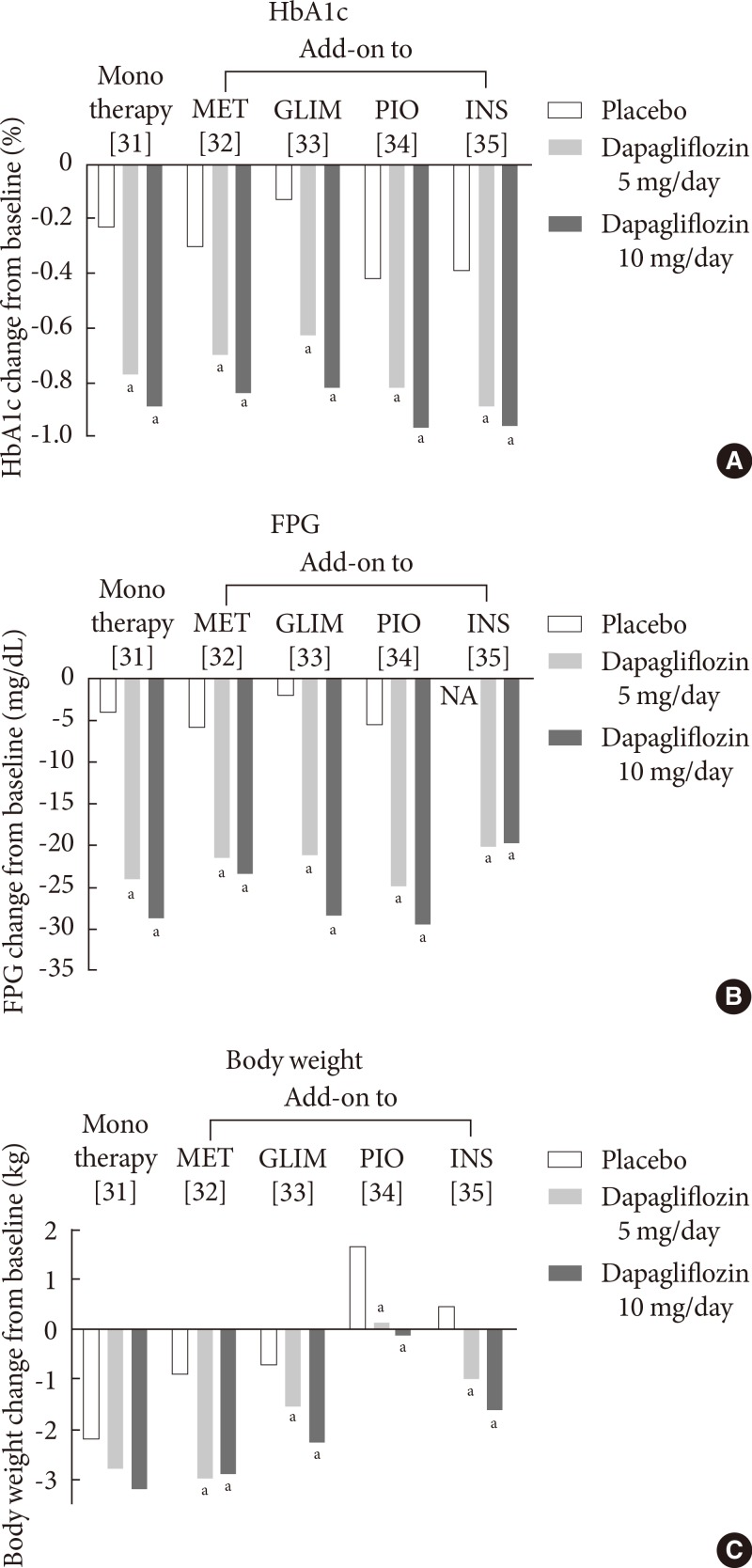
- Dapagliflozin, developed by Bristol-Myers Squibb (New York, NY, USA) and AstraZeneca (Wilmington, DE, USA), is a potent and highly selective SGLT2 inhibitor [28]. Dapagliflozin is rapidly absorbed after oral administration, with a peak plasma concentration within 2 hours of administration [28]. Dapagliflozin has been approved by the European Medicines Agency (EMA) as an adjunct to diet and exercise, in combination with other antidiabetic agents, such as insulin, and as a monotherapy in metformin-intolerant patients in November 2012 [29]. The FDA has also approved dapagliflozin for use in adult patients with T2DM in January 2014 [30]. The efficacy of dapagliflozin in phase III clinical trials with durations of more than 24 weeks is summarized in Fig. 3.
- In clinical studies, treatment with oral dapagliflozin either as monotherapy [31], in combination with other oral antidiabetic agents [32,33,34], or as an insulin-based therapy [35] can significantly improve glycemic control and reduce the fasting plasma glucose (FPG) level (Fig. 3), with longer-term extension studies (≥100 weeks) supporting its extended efficacy [36]. The placebo-corrected reduction in the HbA1c level after treatment with dapagliflozin at a dose of 10 mg/day, in combination with one of the above-mentioned agents, ranges from -0.5% to -0.7% (Fig. 3). Furthermore, dapagliflozin decreases body weight by up to 3 kg over a 24-week treatment period [31,32,33,35]. Although body weight increases when dapagliflozin is co-administered with pioglitazone, the increase is smaller than that of the placebo plus pioglitazone groups (0.69 to 1.35 kg vs. 2.99 kg, respectively) (Fig. 3) [34]. The weight reduction in dapagliflozin-treated patients has been reported to be predominantly attributable to reductions in body fat mass [37].
- In a double-blind trial using an active comparator, dapagliflozin at a dose of 10 mg/day is as effective as extended-release metformin (titrated to 2,000 mg once daily) in reducing the HbA1c level (-1.45% with dapagliflozin at 10 mg/day vs. -1.4% with metformin) and superior to metformin in reducing the FPG level (-46.4 mg/dL with dapagliflozin at 10 mg/day vs. -34 mg/dL with metformin) in treatment-naïve patients with T2DM [38].
- Dapagliflozin has also been studied in initial combination with metformin (titrated to 2,000 mg once daily) in treatment-naïve patients with T2DM. This combination was more effective than either drug alone in reducing the HbA1c level (-1.5% with dapagliflozin at 10 mg/day and -1.4% with metformin vs. -2.0% with dapagliflozin at 10 mg/day and metformin) and the FPG level (-46 mg/dL with dapagliflozin at 10 mg/day and -35 mg/dL with metformin vs. -60 mg/dL with dapagliflozin at 10mg/day and metformin) [38].
- Dapagliflozin was also compared with sulfonylurea (glipizide) in patients whose glycemic control was inadequate on metformin. Despite a similar 52-week glycemic efficacy (-0.52% with dapagliflozin at 10 mg/day vs. -0.52% with glipizide titrated to 20 mg/day), dapagliflozin results in a more reduction in body weight and in a lower level of hypoglycemia than glipizide [39]
- Canagliflozin
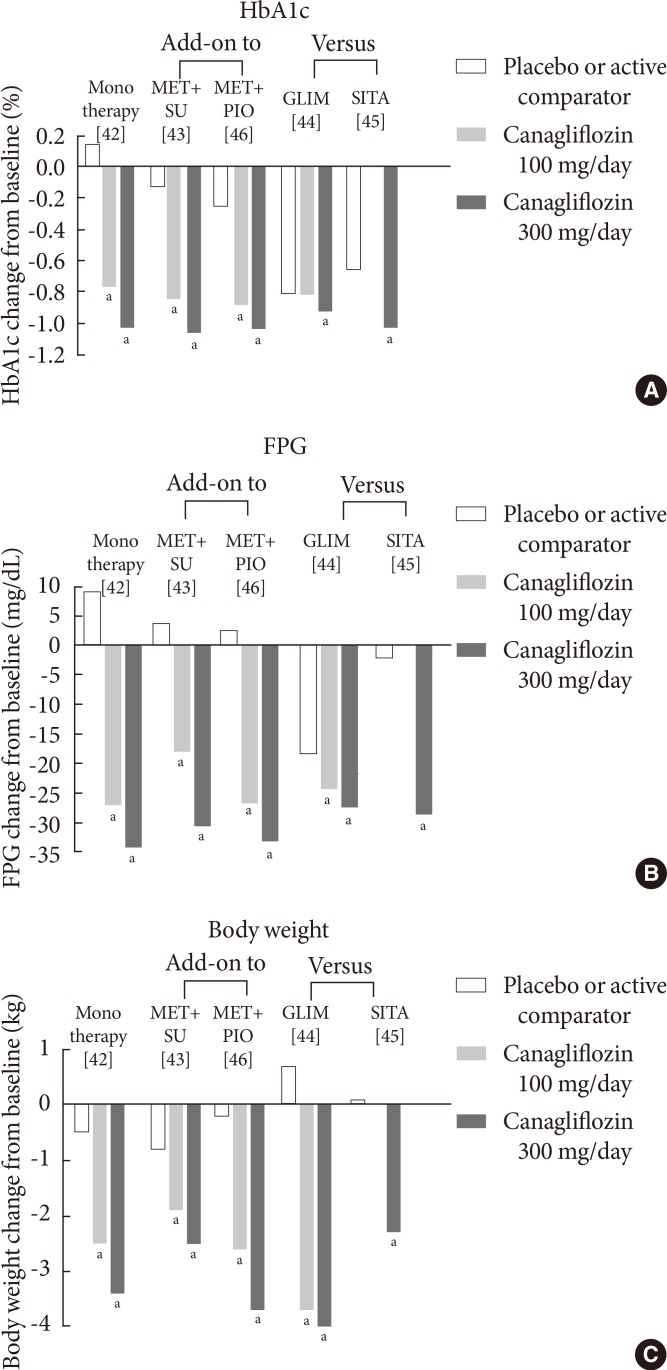
- Canagliflozin (developed by Johnson & Johnson, New Brunswick, NJ, USA) is another SGLT2 inhibitor which became the first SGLT2 inhibitor to be approved in the US in March 2013 [40]. Recently, it was similarly approved by EMA [41]. The efficacy of canagliflozin in phase III clinical trials with durations of more than 24 weeks is summarized in Fig. 4.
- Clinical studies have shown that treatment with oral canagliflozin as a monotherapy [42] or in combination with other oral antidiabetic agents [43,44,45,46] significantly improved glycemic control and reduced FPG levels (Fig. 4). After 26 weeks of canagliflozin treatment, there is a significant placebo-corrected reduction in the HbA1c level in patients inadequately controlled with diet and exercise (-0.91% with canagliflozin at 100 mg/day and -1.17% with canagliflozin at 300 mg/day) (Fig. 4A) [42]. Significant reductions in the FPG level and body weight have also been reported (Fig. 4B and C) [42]. In this study, canagliflozin also significantly reduces postprandial glycemic parameters [42], which may be mediated through a delay in glucose absorption via transient SGLT1 inhibition and an increase in urinary glucose excretion via SGLT2 inhibition [47].
- In clinical studies, which investigate the efficacy of canagliflozin as an add-on treatment to metformin, canagliflozin is compared with the placebo or sitagliptin at a dose of 100 mg/day [48]. After 26 weeks of treatment with canagliflozin at a dose of 100 or 300 mg/day, there is a reduction of the HbA1c level compared with the placebo (-0.79% with canagliflozin at 100 mg/day and -0.94% with canagliflozin at 300 mg/day vs. -0.17% with the placebo) [48]. After 52 weeks of treatment, canagliflozin at a dose of 100 mg/day or 300 mg/day demonstrates noninferiority, and canagliflozin at a dose of 300 mg/day shows superiority to sitagliptin in lowering the HbA1c level (-0.73% with canagliflozin at 100 mg/day and -0.88% with canagliflozin at 300 mg/day vs. -0.73% with sitagliptin) [48]. In addition, canagliflozin at these doses reduces body weight compared with the placebo and sitagliptin [48].
- As secondary agents that are added-on to metformin, canagliflozin at doses of 100 mg/day or 300 mg/day has also been compared with glimepiride (titrated to 8 mg/day) in patients inadequately controlled with metformin [44]. The reduction in the HbA1c level with canagliflozin at a dose of 100 mg/day is noninferior to that of glimepiride (-0.82% with canagliflozin at 100 mg/day vs. -0.81% with glimepiride) [44]. Furthermore, the reduction in the HbA1c level with canagliflozin at a dose of 300 mg/day is superior to that of glimepiride (-0.93% with canagliflozin at 300 mg/day vs. -0.81% with glimepiride) over a 52-week treatment period in patients on background metformin [44]. Canagliflozin at both doses is superior to glimepiride in reducing body weight (-3.7 kg with canagliflozin at 100 mg/day and -4.0 kg with canagliflozinat 300 mg/day vs. 0.7 kg with glimepiride) [44].
- The efficacy of canagliflozin is maintained when it is used as a tertiary agent in patients with T2DM who are inadequately controlled with a dual combination such as metformin and sulfonylurea or metformin and pioglitazone [43,46]. Similar to the superiority of canagliflozin 300 mg/day to sitagliptin (100 mg/day) when added as a secondary agent [48], the change in HbA1c with canagliflozin 300 mg/day was superior to that with sitagliptin when they were added in patients inadequately controlled with metformin plus sulfonylurea (-1.03% with canagliflozin 300 mg/day vs. -0.66% with sitagliptin) [45].
- In patients inadequately controlled with insulin, with or without additional oral agents, canagliflozin at a dose of 100 mg/day reduces the HbA1c level (-0.73%) over a 28-day treatment period compared with the placebo (-0.19%) [49]. The FPG level (-38.0 mg/dL vs. 8.65 mg/dL) and body weight (-0.73 kg vs. 0.03 kg) are also reduced in patients treated with canagliflozin [49].
- Other investigational SGLT2 inhibitors
- As shown in Table 2, several other SGLT2 inhibitors have completed phase III clinical trials or are currently in phase III clinical trials, including empagliflozin (BI 10773) [50], ipragliflozin (ASP 1941) [51], tofogliflozin (CSG 452) [52], luseogliflozin (TS 071) [53], and ertugliflozin (PF 04971729) [54]. Besides these agents, several other SGLT2 inhibitors are currently in phase I/II clinical trials or undergoing preclinical testing.
- Empagliflozin, developed by Boehringer Ingelheim (Ingelheim am Rhein, Germany) and Eli Lilly Pharmaceuticals (Indianapolis, IN, USA), is a potent and selective SGLT2 inhibitor, and a New Drug Application has been submitted to the FDA. It has been studied as a monotherapy, and as an add-on to metformin, two oral agents, and insulin [50].
- Ipragliflozin, codeveloped by Astellas (Tokyo, Japan) and Kotobuki Pharmaceuticals (Nagano, Japan), is another selective SGLT2 inhibitor that is used in Japan for the treatment of T2DM. It is beneficial as a monotherapy, and as an add-on to metformin or other antihyperglycemic agents such as sulfonylurea or pioglitazone [51].
PHARMACOLOGICAL INHIBITORS OF RENAL GLUCOSE UPTAKE
- Phase III clinical trials of dapagliflozin or canagliflozin have shown both agents to be generally well tolerated [31,32,33,34,35,38,39,42,43,44,45,46]. Hypoglycemia is a potential side effect of all hypoglycemic agents. However, hypoglycemia is not anticipated in patients receiving SGLT2 inhibitors because these agents decrease the plasma glucose concentration without augmenting insulin secretion and without inhibiting the counterregulatory response [12]. Indeed, the incidence of hypoglycemia in patients receiving dapagliflozin or canagliflozin was infrequent, occurring at a frequency that is similar to patients receiving a placebo [31,32,33,34,42,43,46]. However, hypoglycemia is more frequent when dapagliflozin or canagliflozin is used in combination with insulin or insulin secretagogue therapy [35,39,44]. Therefore, physicians should reduce the dose of insulin or insulin secretagogue at the time that these SGLT2 inhibitors are initiated. However, in active comparator trials, hypoglycemia has been shown to be less frequent in patients receiving dapagliflozin or canagliflozin compared with those receiving insulin secretagogue therapy [39,44].
- SGLT2 inhibitors pose a risk for urinary tract infection (UTI) because they promote glucosuria [12]. As glucosuria is present in diabetic patients, it should be determined whether an aggravation of glucosuria can promote bacterial growth [12]. Indeed, symptoms suggestive of a genital infection and lower UTI are common adverse events in patients receiving dapagliflozin or canagliflozin, and they are reported more frequently in these patients compared with those receiving a placebo or an active comparator [55,56,57]. A pooled safety analysis (n=4,545) shows that genital infections and UTIs are more common in patients receiving dapagliflozin compared with those receiving a placebo, and between-group difference sare less marked for UTIs (genital infection: 4.1% to 5.7% with dapagliflozin vs. 0.9% with the placebo; UTIs: 3.6% to 5.7% with dapagliflozin vs. 3.7% with the placebo) [55,56]. Similar to dapagliflozin, genital infections and UTIs in patients receiving canagliflozin have also been assessed in a pooled analysis of four 24-week phase III studies (n=2,313) [57]. It has been reported that genital infections are more common in the canagliflozin group than in the placebo group, occurring in 11% of women and 4% of men, compared with 3% and 1% in the placebo group, respectively [57]. These events are generally of mild to moderate intensity, and most patients respond to an initial course of standard treatment and rarely discontinue treatment [57]. In this pooled analysis, canagliflozin has been shown to associate with a moderate increase in the incidence of UTIs, with no increase in serious or upper UTIs [58].
- An increase in urine volume (up to 400 mL) has been observed in clinical studies with SGLT2 inhibitors [59]. Owing to their mild diuretic effect, several adverse events relating to the reduction in intravascular volume have been observed in clinical trials of dapagliflozin or canagliflozin. These adverse events include orthostatic hypotension, electrolyte imbalance, and a deterioration of renal function [59]. This modest diuretic action of SGLT2 inhibitors is likely to explain the moderate increase in hematocrit (1% to 2%) and the plasma urea nitrogen to creatinine ratio [12]. However, this small increase in urine volume does not associate with an electrolyte imbalance, an acid-base disorder, or nocturia, and it did not affect patient quality of life [60,61].
- With regard to the effects of SGLT2 inhibitors on blood pressure (BP), dapagliflozin reduces systolic BP by up to 5 mm Hg, with no significant increase in the heart rate or the occurrence of orthostatic hypotension [31,32,33,34,35,37,39]. The rates of hypotension, dehydration, and hypovolemia are similar in patients receiving dapagliflozin (1% to 2%) compared with those receiving a placebo or an active comparator (0% to 1%) [31,35]. Dapagliflozin treatment does not associate with an increased risk of acute renal toxicity or deterioration of renal function [62]. However, dapagliflozin is not recommended for T2DM patients with moderate to severe renal impairment, end-stage renal disease, or those on dialysis as the mean reduction in the HbA1c level is not significantly different between dapagliflozin (-0.43%) and placebo (-0.32%) in T2DM subjects with moderate renal impairment (an estimated GFR [eGFR] between 30 and 59 mL/min/1.73 m2) [63].
- Similarly, monotherapy with canagliflozin at a dose of 100 mg/day (-3.6 mm Hg) or 300 mg/day (-5.4 mm Hg) for 26 weeks results in a statistically significant reduction in systolic BP compared with the placebo (0.4 mm Hg) [42]. The diastolic BP is also reduced with canagliflozin (-1.6 and -2.0 mm Hg, respectively) versus the placebo (-0.1 mm Hg), although statistical analysis has not been performed [63]. Owing to its mild diuretic effect, several adverse events relating to a reduction in the intravascular volume are observed in canagliflozin trials. These adverse events include pollakiruia (increased urine frequency) and polyuria (increased urine volume), with a frequency generally in the 2% to 5% range, and similar to its comparator [42,45,64,65]. Occurring less frequently, but of greater concern, are the risks of postural dizziness and orthostatic hypotension. Their prevalence has been reported to occur in a dose-dependent manner, appearing in 2.3% and 3.4% of patients receiving canagliflozin at a dose 100 or 300 mg/day, respectively, versus 1.5% of patients receiving an active comparator [42,44,45,64,65]. Factors associating with volume-related adverse effects are older age (≥75 years), concomitant use of loop diuretics, and a moderate renal impairment eGFR between 30 and 59 mL/min/1.73 m2 [66]. The small, transient, and reversible decrease in the eGFR observed with canagliflozin therapy is consistent with its hemodynamic effect [66]. The current prescription guidelines for canagliflozin recommend a maximum daily dose of 100 mg for patients with an eGFR between 46 and 59 mL/min/1.73 m2. They do not recommend it for subjects with an eGFR <45 mL/min/1.73 m2 based on findings that show these individuals to have a lower glycemic efficacy than those with normal renal function, and a higher incidence of renal-related adverse effects such as hypovolemia and hyperkalemia [66].
- Regarding the effect of SGLT2 inhibitors on cardiovascular risk factors other than body weight and BP, a pooled analysis in the dapagliflozin trials concluded that dapagliflozin had an insignificant effect on lipid levels such as small changes in high-density lipoprotein cholesterol (HDL-C; 2.1% to 9.3%), triglycerides (-0.9% to -10.6%) and low-density lipoprotein cholesterol (LDL-C; -0.5% to 9.5%) [67]. In a pooled analysis of the placebo-controlled canagliflozin trials, the placebo-corrected mean percent change from baseline for LDL-C were 4.5% and 8.0% for the canagliflozin 100 and 300 mg/day groups, respectively [64]. The mechanism responsible for this rise in LDL-C is unknown but could result from hemoconcentration or transfer of cholesterol from triglycerides to apolipoprotein B100. However, canagliflozin treatment is proven to improve other lipid parameters such as increases in HDL-C (0.8% to 6.8% with the 100 mg/day and 0.9% to 8.4% with 300 mg/day) and decreases in triglycerides, which may counterbalance the slight rise in LDL-C [64].
- Despite there being no evidence of increased teratogenicity in animal studies, there is an increase in the number of breast and bladder cancers in patients receiving dapagliflozin in clinical trials [68]. Breast cancer has been reported in 10 dapagliflozin-treated patients and three placebo-treated patients [69]. All cases of breast cancer have been detected within the first year of the study [69]. Bladder cancer has also been reported in nine dapagliflozin-treated patients and one placebo-treated patient [69]. Hematuria at baseline has been observed in the majority of patients, suggesting a possible pre-existing cancer [69]. In light of these findings, dapagliflozin is not currently recommended for patients with bladder cancer [70]. Furthermore, the FDA has rejected dapagliflozin in January 2012, in part because of breast and bladder cancer concerns. Although the FDA has recently approved dapagliflozin for use in T2DM patients, it is requiring that companies perform several post-marketing studies to determine whether dapagliflozin associates with malignancies [71].
SAFETY AND TOLERABILITY OF SGLT2 INHIBITORS
- SGLT2 inhibition represents a particularly appealing approach to treat diabetes, in contrast to many other antidiabetic agents, because SGLT2 inhibition does not directly influence insulin secretion, indicating that it utilizes a novel mechanism of action [7,12]. Indeed, the future role of SGLT2 inhibitors in the management of type 1 diabetes mellitus cannot be ruled out, although SGLT2 inhibitors have not yet been tested for this indication. Furthermore, this class of drugs has a unique property of inducing weight loss, which could also be effective in the treatment of obesity and metabolic syndromes [72]. Although the early weight loss is due to mild osmotic diuresis, the progressive long-term reduction in body weight is attributed to a reduction of fat mass, which is attributed to the loss of energy through glucose excretion in urine [37]. Further research is warranted to prove the effects of SGLT2 inhibitors on obesity and metabolic syndromes. However, even when these putative effects of SGLT2 inhibitors are proven, dietary intervention should be enhanced as weight loss can be attenuated by compensatory hyperphagia [73].
- While recent trials of other antidiabetic agents have failed to show a definite protective effect for macrovascular outcomes, there is an obvious benefit for microvascular complications [2,5,6]. As SGLT2 inhibitors significantly improve BP, body weight, and glycemic indicators, there is an expectation that these agents will protect not only against microvascular complications but also against macrovascular diseases associated with T2DM. Although the likelihood of these beneficial effects of SGLT2 inhibitors is strong, definite evidence that reliably defines the effects of these agents on vascular events, along with a clear understanding of their safety profile, will be a prerequisite for their widespread use in clinical practice. Currently, several large-scale clinical trials "Dapagliflozin Effect on Cardiovascular Events" (DECLARE-TIMI58, NCT-01730534) and "Canagliflozin Cardiovascular Assessment Study" (CANVAS, NCT01032629) using dapagliflozin and canagliflozin, respectively] are ongoing to evaluate the effects of dapagliflozin and canagliflozin on the risk of CVD. Evidence that will define the overall balance of benefits and risks of this new drug class is anticipated within the next 5 years.
FUTURE PERSPECTIVE
- SGLT2 inhibitors prevent glucose reabsorption from renal tubules, thereby promoting urinary glucose excretion and decreasing plasma glucose levels. Current data in humans indicate that SGLT2 inhibitors represent an effective and novel strategy to control the plasma glucose concentration in patients with T2DM. The clinical trials of the most advanced SGLT2 inhibitors, dapagliflozin and canagliflozin, show therapeutic benefits in attaining glycemic control, lowering the plasma glucose level, and reducing the body weight of patients with T2DM. Furthermore, the hypoglycemic episodes associated with SGLT2 inhibitors are mostly mild in severity and not statistically significant compared with the active comparator. Other SGLT2 inhibitors in earlier stages of clinical development also show promising effects on glycemic control. Apart from these effects, several beneficial effects on BP and body weight have also been observed.
- As SGLT2 inhibitors have a unique mechanism of action that is independent of insulin secretion or the degree of insulin resistance, the efficacy of this class of drugs is anticipated not to decline with progressive β-cell failure or in the presence of severe insulin resistance [12]. However, as the efficacy of these agents is dependent on glomerular filtration, these therapeutic benefits are limited to a subset of diabetic patients with normal renal function or mild renal dysfunction [74]. Although SGLT2 inhibitors appear to be well tolerated, increased risks of genital infections, and in some studies, UTIs have been reported [55,56,57,58]. Although long-term safety data are required to determine the significance of these observations, time will tell whether this increase in frequency of genitourinary infections will be tolerated by diabetic patients.
- The magnitude of the global diabetes problem and the unmet needs of current antidiabetic agents are the primary drivers behind the effort to identify new treatment modalities. Although currently available data indicate that SGLT2 inhibitors fulfill these unmet needs to some extent, larger studies with longer follow-up periods are warranted to establish the long-term safety and efficacy of SGLT2 inhibitors.
CONCLUSIONS
- 1. Guariguata L. By the numbers: new estimates from the IDF Diabetes Atlas Update for 2012. Diabetes Res Clin Pract 2012;98:524-525. ArticlePubMed
- 2. UK Prospective Diabetes Study (UKPDS) Group. Intensive blood-glucose control with sulphonylureas or insulin compared with conventional treatment and risk of complications in patients with type 2 diabetes (UKPDS 33). Lancet 1998;352:837-853. ArticlePubMed
- 3. Holman RR, Paul SK, Bethel MA, Matthews DR, Neil HA. 10-year follow-up of intensive glucose control in type 2 diabetes. N Engl J Med 2008;359:1577-1589. ArticlePubMed
- 4. Inzucchi SE. Oral antihyperglycemic therapy for type 2 diabetes: scientific review. JAMA 2002;287:360-372. ArticlePubMed
- 5. Duckworth W, Abraira C, Moritz T, Reda D, Emanuele N, Reaven PD, Zieve FJ, Marks J, Davis SN, Hayward R, Warren SR, Goldman S, McCarren M, Vitek ME, Henderson WG, Huang GD. VADT Investigators. Glucose control and vascular complications in veterans with type 2 diabetes. N Engl J Med 2009;360:129-139. ArticlePubMed
- 6. Action to Control Cardiovascular Risk in Diabetes Study Group. Gerstein HC, Miller ME, Byington RP, Goff DC Jr, Bigger JT, Buse JB, Cushman WC, Genuth S, Ismail-Beigi F, Grimm RH Jr, Probstfield JL, Simons-Morton DG, Friedewald WT. Effects of intensive glucose lowering in type 2 diabetes. N Engl J Med 2008;358:2545-2559. ArticlePubMedPMC
- 7. Chao EC, Henry RR. SGLT2 inhibition: a novel strategy for diabetes treatment. Nat Rev Drug Discov 2010;9:551-559. ArticlePubMedPDF
- 8. Hasan FM, Alsahli M, Gerich JE. SGLT2 inhibitors in the treatment of type 2 diabetes. Diabetes Res Clin Pract 2014;104:297-322. ArticlePubMed
- 9. Raskin P. Sodium-glucose cotransporter inhibition: therapeutic potential for the treatment of type 2 diabetes mellitus. Diabetes Metab Res Rev 2013;29:347-356. ArticlePubMed
- 10. Mather A, Pollock C. Glucose handling by the kidney. Kidney Int Suppl 2011;(120):S1-S6. ArticlePMC
- 11. Gerich JE. Role of the kidney in normal glucose homeostasis and in the hyperglycaemia of diabetes mellitus: therapeutic implications. Diabet Med 2010;27:136-142. ArticlePubMedPMC
- 12. Abdul-Ghani MA, Norton L, Defronzo RA. Role of sodium-glucose cotransporter 2 (SGLT 2) inhibitors in the treatment of type 2 diabetes. Endocr Rev 2011;32:515-531. ArticlePubMedPDF
- 13. Wright EM, Hirayama BA, Loo DF. Active sugar transport in health and disease. J Intern Med 2007;261:32-43. ArticlePubMed
- 14. Bays H. From victim to ally: the kidney as an emerging target for the treatment of diabetes mellitus. Curr Med Res Opin 2009;25:671-681. ArticlePubMed
- 15. Gerich JE, Meyer C, Woerle HJ, Stumvoll M. Renal gluconeogenesis: its importance in human glucose homeostasis. Diabetes Care 2001;24:382-391. PubMed
- 16. Rahmoune H, Thompson PW, Ward JM, Smith CD, Hong G, Brown J. Glucose transporters in human renal proximal tubular cells isolated from the urine of patients with non-insulin-dependent diabetes. Diabetes 2005;54:3427-3434. ArticlePubMedPDF
- 17. Kamran M, Peterson RG, Dominguez JH. Overexpression of GLUT2 gene in renal proximal tubules of diabetic Zucker rats. J Am Soc Nephrol 1997;8:943-948. ArticlePubMed
- 18. Mogensen CE. Maximum tubular reabsorption capacity for glucose and renal hemodynamcis during rapid hypertonic glucose infusion in normal and diabetic subjects. Scand J Clin Lab Invest 1971;28:101-109. PubMed
- 19. Freitas HS, Anhe GF, Melo KF, Okamoto MM, Oliveira-Souza M, Bordin S, Machado UF. Na(+) -glucose transporter-2 messenger ribonucleic acid expression in kidney of diabetic rats correlates with glycemic levels: involvement of hepatocyte nuclear factor-1alpha expression and activity. Endocrinology 2008;149:717-724. PubMed
- 20. Defronzo RA. Banting Lecture. From the triumvirate to the ominous octet: a new paradigm for the treatment of type 2 diabetes mellitus. Diabetes 2009;58:773-795. PubMedPMC
- 21. DeFronzo RA, Davidson JA, Del Prato S. The role of the kidneys in glucose homeostasis: a new path towards normalizing glycaemia. Diabetes Obes Metab 2012;14:5-14. ArticlePubMed
- 22. Ehrenkranz JR, Lewis NG, Kahn CR, Roth J. Phlorizin: a review. Diabetes Metab Res Rev 2005;21:31-38. ArticlePubMed
- 23. Alvarado F, Crane RK. Phlorizin as a competitive inhibitor of the active transport of sugars by hamster small intestine, in vitro. Biochim Biophys Acta 1962;56:170-172. ArticlePubMed
- 24. Rossetti L, Smith D, Shulman GI, Papachristou D, DeFronzo RA. Correction of hyperglycemia with phlorizin normalizes tissue sensitivity to insulin in diabetic rats. J Clin Invest 1987;79:1510-1515. ArticlePubMedPMC
- 25. Wright EM, Loo DD, Hirayama BA. Biology of human sodium glucose transporters. Physiol Rev 2011;91:733-794. ArticlePubMed
- 26. Thorens B, Mueckler M. Glucose transporters in the 21st Century. Am J Physiol Endocrinol Metab 2010;298:E141-E145. ArticlePubMed
- 27. Elsas LJ, Rosenberg LE. Familial renal glycosuria: a genetic reappraisal of hexose transport by kidney and intestine. J Clin Invest 1969;48:1845-1854. ArticlePubMedPMC
- 28. Plosker GL. Dapagliflozin: a review of its use in type 2 diabetes mellitus. Drugs 2012;72:2289-2312. ArticlePubMed
- 29. European Medicines Agency. European Medicines Agency authorizing the use of dapagliflozin in November 2012 updated 2014 Jul 16. Available from: http://www.ema.europa.eu/ema/index.jsp?curl=pages/medicines/human/medicines/002322/human_med_001546.jsp&mid=WC0b01ac058001d124.
- 30. U.S. Food and Drug Administration. FDA approves Farxiga to treat type 2 diabetes updated 2014 Jan 9. Available from: http://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm380829.htm.
- 31. Ferrannini E, Ramos SJ, Salsali A, Tang W, List JF. Dapagliflozin monotherapy in type 2 diabetic patients with inadequate glycemic control by diet and exercise: a randomized, double-blind, placebo-controlled, phase 3 trial. Diabetes Care 2010;33:2217-2224. PubMedPMC
- 32. Bailey CJ, Gross JL, Pieters A, Bastien A, List JF. Effect of dapagliflozin in patients with type 2 diabetes who have inadequate glycaemic control with metformin: a randomised, double-blind, placebo-controlled trial. Lancet 2010;375:2223-2233. ArticlePubMed
- 33. Strojek K, Yoon KH, Hruba V, Elze M, Langkilde AM, Parikh S. Effect of dapagliflozin in patients with type 2 diabetes who have inadequate glycaemic control with glimepiride: a randomized, 24-week, double-blind, placebo-controlled trial. Diabetes Obes Metab 2011;13:928-938. ArticlePubMed
- 34. Rosenstock J, Vico M, Wei L, Salsali A, List JF. Effects of dapagliflozin, an SGLT2 inhibitor, on HbA(1c), body weight, and hypoglycemia risk in patients with type 2 diabetes inadequately controlled on pioglitazone monotherapy. Diabetes Care 2012;35:1473-1478. ArticlePubMedPMCPDF
- 35. Wilding JP, Woo V, Soler NG, Pahor A, Sugg J, Rohwedder K, Parikh S. Dapagliflozin 006 Study Group. Long-term efficacy of dapagliflozin in patients with type 2 diabetes mellitus receiving high doses of insulin: a randomized trial. Ann Intern Med 2012;156:405-415. ArticlePubMed
- 36. Wilding JP, Woo V, Rohwedder K, Sugg J, Parikh S. for the Dapagliflozin 006 Study Group. Dapagliflozin in patients with type 2 diabetes receiving high doses of insulin: efficacy and safety over 2 years. Diabetes Obes Metab 2013 98:524-525. Epub 2013 Aug 1. DOI: http://dx.doi.org/10.1111/dom.12187.Article
- 37. Bolinder J, Ljunggren O, Kullberg J, Johansson L, Wilding J, Langkilde AM, Sugg J, Parikh S. Effects of dapagliflozin on body weight, total fat mass, and regional adipose tissue distribution in patients with type 2 diabetes mellitus with inadequate glycemic control on metformin. J Clin Endocrinol Metab 2012;97:1020-1031. ArticlePubMed
- 38. Henry RR, Murray AV, Marmolejo MH, Hennicken D, Ptaszynska A, List JF. Dapagliflozin, metformin XR, or both: initial pharmacotherapy for type 2 diabetes, a randomised controlled trial. Int J Clin Pract 2012;66:446-456. ArticlePubMed
- 39. Nauck MA, Del Prato S, Meier JJ, Duran-Garcia S, Rohwedder K, Elze M, Parikh SJ. Dapagliflozin versus glipizide as add-on therapy in patients with type 2 diabetes who have inadequate glycemic control with metformin: a randomized, 52-week, double-blind, active-controlled noninferiority trial. Diabetes Care 2011;34:2015-2022. PubMedPMC
- 40. U.S. Food and Drug Administration. FDA approves Invokana to treat type 2 diabetes updated 2013 Mar 29. Available from: http://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm345848.htm.
- 41. European Medicines Agency. European Medicines Agency authorizing the use of canagliflozin in November 2013 updated 2013 Nov 30. Available from: http://www.ema.europa.eu/ema/index.jsp?curl=pages/medicines/human/medicines/002649/human_med_001707.jsp&mid=WC0b01ac058001d124.
- 42. Stenlof K, Cefalu WT, Kim KA, Alba M, Usiskin K, Tong C, Canovatchel W, Meininger G. Efficacy and safety of canagliflozin monotherapy in subjects with type 2 diabetes mellitus inadequately controlled with diet and exercise. Diabetes Obes Metab 2013;15:372-382. ArticlePubMedPMCPDF
- 43. Wilding JP, Charpentier G, Hollander P, Gonzalez-Galvez G, Mathieu C, Vercruysse F, Usiskin K, Law G, Black S, Canovatchel W, Meininger G. Efficacy and safety of canagliflozin in patients with type 2 diabetes mellitus inadequately controlled with metformin and sulphonylurea: a randomised trial. Int J Clin Pract 2013;67:1267-1282. ArticlePubMedPMCPDF
- 44. Cefalu WT, Leiter LA, Yoon KH, Arias P, Niskanen L, Xie J, Balis DA, Canovatchel W, Meininger G. Efficacy and safety of canagliflozin versus glimepiride in patients with type 2 diabetes inadequately controlled with metformin (CANTATA-SU): 52 week results from a randomised, double-blind, phase 3 non-inferiority trial. Lancet 2013;382:941-950. ArticlePubMed
- 45. Schernthaner G, Gross JL, Rosenstock J, Guarisco M, Fu M, Yee J, Kawaguchi M, Canovatchel W, Meininger G. Canagliflozin compared with sitagliptin for patients with type 2 diabetes who do not have adequate glycemic control with metformin plus sulfonylurea: a 52-week randomized trial. Diabetes Care 2013;36:2508-2515. PubMedPMC
- 46. Forst T, Guthrie R, Goldenberg R, Yee J, Vijapurkar U, Meininger G, Stein P. Efficacy and safety of canagliflozin over 52 weeks in patients with type 2 diabetes on background metformin and pioglitazone. Diabetes Obes Metab 2014;16:467-477. ArticlePubMedPMCPDF
- 47. Polidori D, Sha S, Mudaliar S, Ciaraldi TP, Ghosh A, Vaccaro N, Farrell K, Rothenberg P, Henry RR. Canagliflozin lowers postprandial glucose and insulin by delaying intestinal glucose absorption in addition to increasing urinary glucose excretion: results of a randomized, placebo-controlled study. Diabetes Care 2013;36:2154-2161. PubMedPMC
- 48. Lavalle-Gonzalez FJ, Januszewicz A, Davidson J, Tong C, Qiu R, Canovatchel W, Meininger G. Efficacy and safety of canagliflozin compared with placebo and sitagliptin in patients with type 2 diabetes on background metformin monotherapy: a randomised trial. Diabetologia 2013;56:2582-2592. ArticlePubMedPMCPDF
- 49. Devineni D, Morrow L, Hompesch M, Skee D, Vandebosch A, Murphy J, Ways K, Schwartz S. Canagliflozin improves glycaemic control over 28 days in subjects with type 2 diabetes not optimally controlled on insulin. Diabetes Obes Metab 2012;14:539-545. ArticlePubMed
- 50. Liakos A, Karagiannis T, Athanasiadou E, Sarigianni M, Mainou M, Papatheodorou K, Bekiari E, Tsapas A. Efficacy and safety of empagliflozin for type 2 diabetes: a systematic review and meta-analysis. Diabetes Obes Metab Epub 2014 Apr 26. DOI: http://dx.doi.org/10.1111/dom.12307.Article
- 51. Poole RM, Dungo RT. Ipragliflozin: first global approval. Drugs 2014;74:611-617. ArticlePubMedPDF
- 52. Pafili K, Papanas N. Tofogliflozin: the road goes ever on. Expert Opin Pharmacother 2014;15:1197-1201. ArticlePubMed
- 53. Seino Y, Sasaki T, Fukatsu A, Ubukata M, Sakai S, Samukawa Y. Efficacy and safety of luseogliflozin as monotherapy in Japanese patients with type 2 diabetes mellitus: a randomized, double-blind, placebo-controlled, phase 3 study. Curr Med Res Opin 2014.Article
- 54. Miao Z, Nucci G, Amin N, Sharma R, Mascitti V, Tugnait M, Vaz AD, Callegari E, Kalgutkar AS. Pharmacokinetics, metabolism, and excretion of the antidiabetic agent ertugliflozin (PF-04971729) in healthy male subjects. Drug Metab Dispos 2013;41:445-456. ArticlePubMed
- 55. Johnsson KM, Ptaszynska A, Schmitz B, Sugg J, Parikh SJ, List JF. Vulvovaginitis and balanitis in patients with diabetes treated with dapagliflozin. J Diabetes Complications 2013;27:479-484. ArticlePubMed
- 56. Johnsson KM, Ptaszynska A, Schmitz B, Sugg J, Parikh SJ, List JF. Urinary tract infections in patients with diabetes treated with dapagliflozin. J Diabetes Complications 2013;27:473-478. ArticlePubMed
- 57. Nyirjesy P, Sobel JD, Fung A, Mayer C, Capuano G, Ways K, Usiskin K. Genital mycotic infections with canagliflozin, a sodium glucose co-transporter 2 inhibitor, in patients with type 2 diabetes mellitus: a pooled analysis of clinical studies. Curr Med Res Opin 2014;30:1109-1119. ArticlePubMed
- 58. Nicolle LE, Capuano G, Fung A, Usiskin K. Urinary tract infection in randomized phase III studies of canagliflozin, a sodium glucose co-transporter 2 inhibitor. Postgrad Med 2014;126:7-17. Article
- 59. Nair S, Wilding JP. Sodium glucose cotransporter 2 inhibitors as a new treatment for diabetes mellitus. J Clin Endocrinol Metab 2010;95:34-42. ArticlePubMed
- 60. Wilding JP, Norwood P, T'Joen C, Bastien A, List JF, Fiedorek FT. A study of dapagliflozin in patients with type 2 diabetes receiving high doses of insulin plus insulin sensitizers: applicability of a novel insulin-independent treatment. Diabetes Care 2009;32:1656-1662. PubMedPMC
- 61. Zhang L, Feng Y, List J, Kasichayanula S, Pfister M. Dapagliflozin treatment in patients with different stages of type 2 diabetes mellitus: effects on glycaemic control and body weight. Diabetes Obes Metab 2010;12:510-516. ArticlePubMed
- 62. Ptaszynska A, Chalamandaris AG, Sugg JE, Johnsson KM, Parikh S, List JL. Effect of dapagliflozin on renal function. Diabetes 2012;61(Suppl 1):A283.
- 63. Kohan DE, Fioretto P, Tang W, List JF. Long-term study of patients with type 2 diabetes and moderate renal impairment shows that dapagliflozin reduces weight and blood pressure but does not improve glycemic control. Kidney Int 2014;85:962-971. ArticlePubMed
- 64. Rosenstock J, Aggarwal N, Polidori D, Zhao Y, Arbit D, Usiskin K, Capuano G, Canovatchel W. Canagliflozin DIA 2001 Study Group. Dose-ranging effects of canagliflozin, a sodium-glucose cotransporter 2 inhibitor, as add-on to metformin in subjects with type 2 diabetes. Diabetes Care 2012;35:1232-1238. ArticlePubMedPMCPDF
- 65. Yale JF, Bakris G, Cariou B, Yue D, David-Neto E, Xi L, Figueroa K, Wajs E, Usiskin K, Meininger G. Efficacy and safety of canagliflozin in subjects with type 2 diabetes and chronic kidney disease. Diabetes Obes Metab 2013;15:463-473. ArticlePubMedPMCPDF
- 66. Janssen Research and Development LCC. Canagliflozin slides presented at the Food and Drug Administration (FDA) Endocrinologic and Metabolic Drugs Advisory Committee (EMDAC) meeting, January 10, 2013 updated 2013 Jan 18. Available from: http://www.fda.gov/AdvisoryCommittees/CommitteesMeetingMaterials/Drugs/EndocrinologicandMetabolicDrugsAdvisoryCommittee/ucm336233.htm.
- 67. Ptaszynska A, Hardy E, Johnsson E, Parikh S, List J. Effects of dapagliflozin on cardiovascular risk factors. Postgrad Med 2013;125:181-189. Article
- 68. Burki TK. FDA rejects novel diabetes drug over safety fears. Lancet 2012;379:507ArticlePubMed
- 69. Ptaszynska A, Johnsson KM, Apanovitch AM. Safety of dapagliflozin in clinical trials for T2DM. Diabetes 2012;61(Suppl 1):A258.
- 70. FORXIGA. Summary of product characteristics. Wilmington: Bristol-Myers Squibb/AstraZeneca EEIG; 2013.
- 71. AstraZeneca Global. AstraZeneca and Bristol-Myers Squibb receive complete response letter from US Food and Drug Administration for dapagliflozin updated 2012 Jan 19. Available from: http://www.astrazeneca.com/Media/Press-releases/Article/19012012--AstraZeneca-Bristol-Myers-Squibb-receive-CRL-for-dapagliflozin.
- 72. Ghosh RK, Ghosh SM, Chawla S, Jasdanwala SA. SGLT2 inhibitors: a new emerging therapeutic class in the treatment of type 2 diabetes mellitus. J Clin Pharmacol 2012;52:457-463. ArticlePubMed
- 73. Devenny JJ, Godonis HE, Harvey SJ, Rooney S, Cullen MJ, Pelleymounter MA. Weight loss induced by chronic dapagliflozin treatment is attenuated by compensatory hyperphagia in diet-induced obese (DIO) rats. Obesity (Silver Spring) 2012;20:1645-1652. ArticlePubMedPDF
- 74. Misra M. SGLT2 inhibitors: a promising new therapeutic option for treatment of type 2 diabetes mellitus. J Pharm Pharmacol 2013;65:317-327. ArticlePubMedPDF
REFERENCES
Fig. 1Glucose reabsorption in the renal proximal tubule. Under normal physiological conditions, the kidney reabsorbs all of the filtered glucose. This occurs via the actions of sodium-glucose cotransporter 2 (SGLT2) in the early proximal tubule, which reabsorbs most of the filtered glucose load, and SGLT1 in the more distal regions of the tubule, which absorbs the remaining glucose. These cotransporters are located on the luminal epithelium. Glucose transporter 2 (GLUT2) and GLUT1 facilitate glucose transport across the basolateral membrane in the early and the more distal regions of the proximal tubule, respectively.
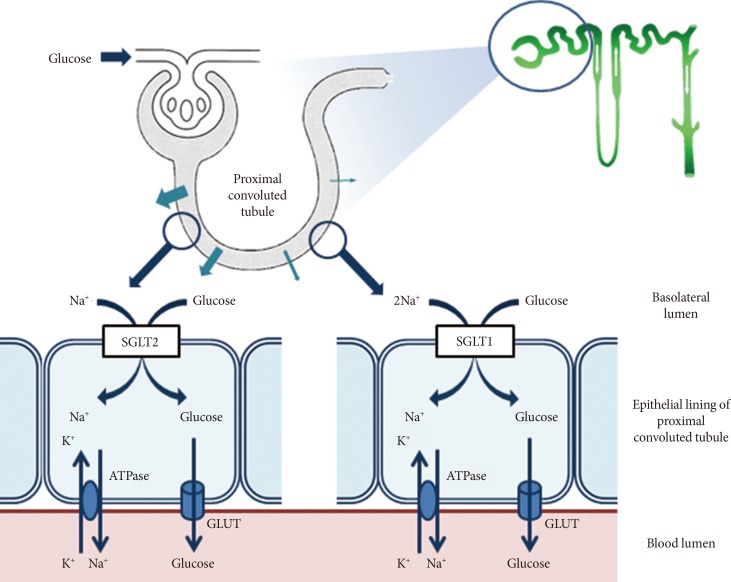

Fig. 2Renal glucose handling before and after sodium-glucose cotransporter 2 (SGLT2) inhibition. SGLT2 inhibition reduces the maximum transport rate (Tm) of glucose. This reduced Tm for glucose through SGLT2 inhibition results in a decrease in glucose reabsorption in the renal proximal tubule and lowers the renal threshold so that glucosuria occurs at a lower plasma glucose concentration.
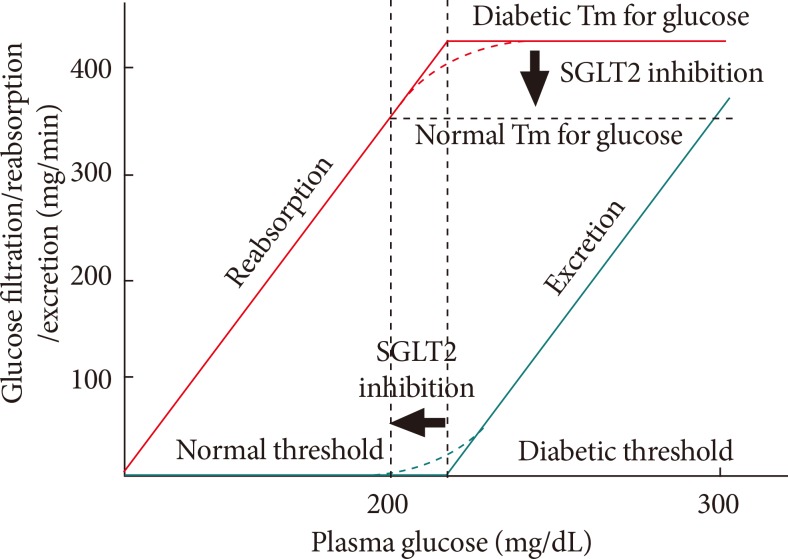

Fig. 3Mean changes in (A) the glycated hemoglobin (HbA1c) level, (B) the fasting plasma glucose (FPG) level, and (C) body weight in clinical trials with dapagliflozin at a dose of 5 or 10 mg/day. Data are adjusted for baseline values. MET, metformin; GLIM, glimepiride; PIO, pioglitazone; INS, insulin; NA, not available. aP<0.05 vs. the placebo.
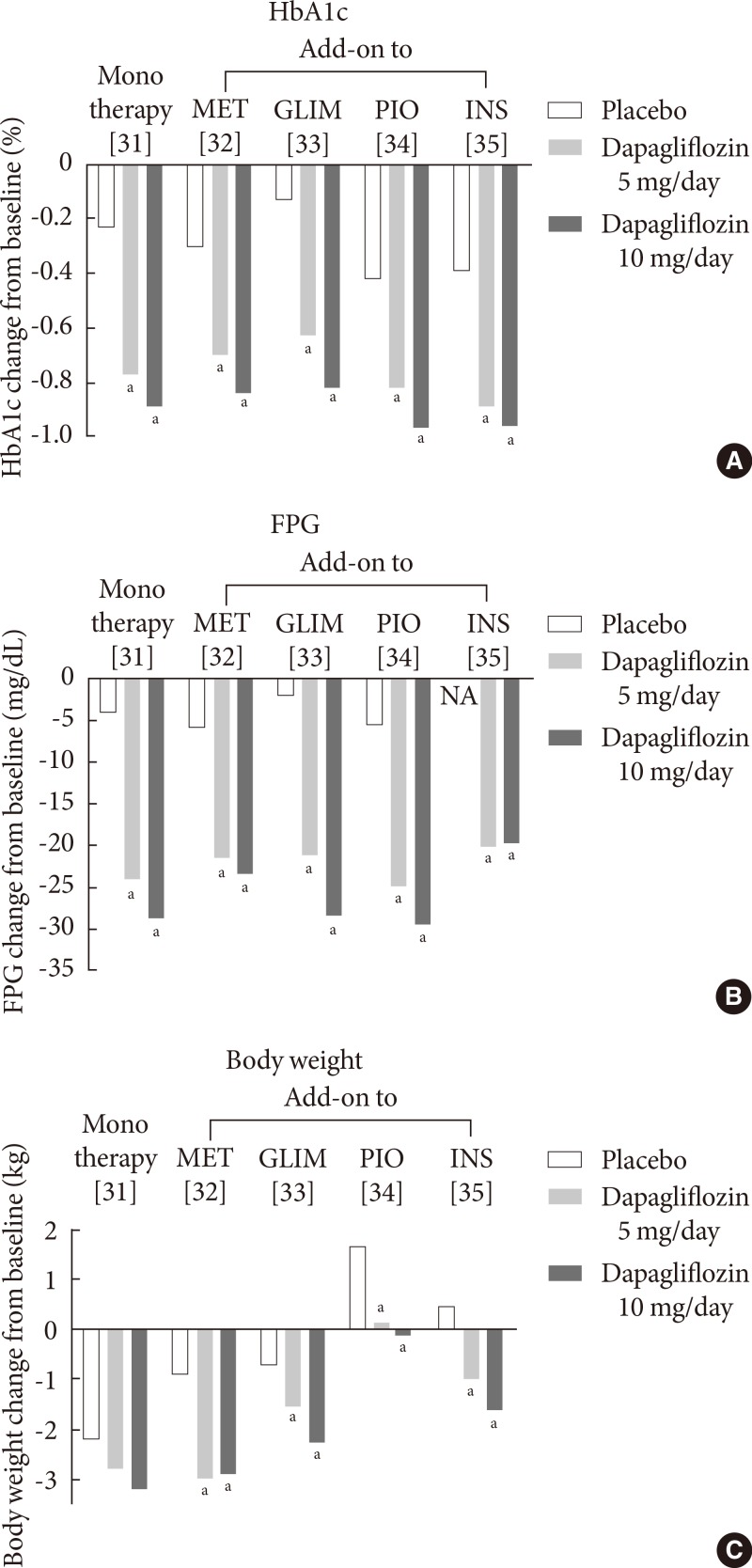

Fig. 4Mean changes in (A) the glycated hemoglobin (HbA1c) level, (B) the fasting plasma glucose (FPG) level, and (C) body weight in clinical trials with canagliflozin at a dose of 100 or 300 mg/day. Data are adjusted for baseline values. MET, metformin; SU, sulfonylurea; PIO, pioglitazone; GLIM, glimepiride; SIT, sitagliptin. aP<0.05 vs. the placebo or active comparator.
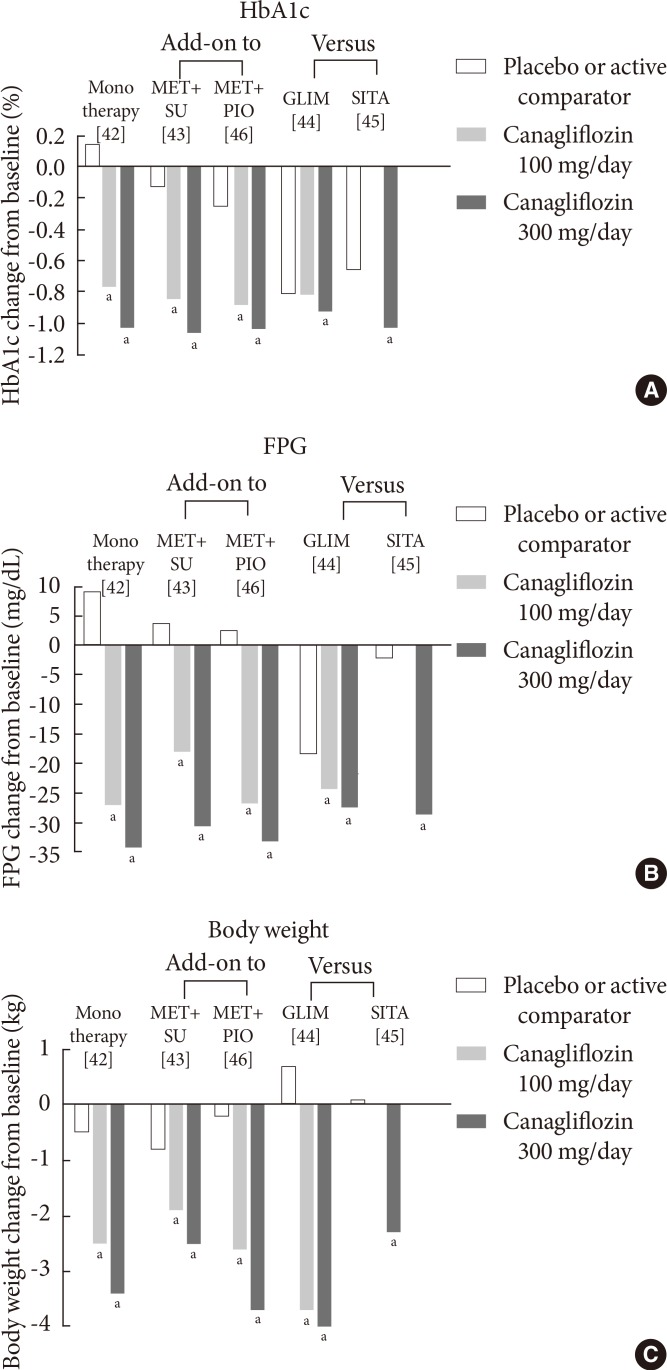

Figure & Data
References
Citations
Citations to this article as recorded by 

- Clinical outcomes with the use of sodium-glucose cotransporter-2 inhibitors in patients with atrial fibrillation and type 2 diabetes mellitus: a multi-centre, real-world cohort study
Jaehyuk Jang, Soyoon Park, Soohyun Kim, Sung-Hwan Kim, Yong-Seog Oh, Young Kyoung Sa, Youmi Hwang, Sung-Won Jang, Sang-Hyun Ihm, Young Choi
European Journal of Preventive Cardiology.2024; 31(3): 320. CrossRef - Quantitative analysis of low-content impurity crystal forms in canagliflozin tablets by NIR solid-state analysis technique
Mingdi Liu, Jichao Liu, Qiuhong Wang, Ping Song, Haichao Li, Zan Sun, Chenglong Shi, Weibing Dong
Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy.2024; 311: 124000. CrossRef - A Randomized Crossover Trial of Mixed Meal Tolerance Test Response in People with Type 1 Diabetes on Insulin Pump Therapy and YG1699 or Dapagliflozin
Pablo Lapuerta, Silvia Urbina, Jiaojuan He, Alyssa Wittle, Chenghai Li, Tong Li, Helen Wang, Marcus Hompesch
Clinical Pharmacology & Therapeutics.2024;[Epub] CrossRef - Canagliflozin independently reduced plasma volume from conventional diuretics in patients with type 2 diabetes and chronic heart failure: a subanalysis of the CANDLE trial
Kazuki Shiina, Hirofumi Tomiyama, Atsushi Tanaka, Takumi Imai, Itaru Hisauchi, Isao Taguchi, Akira Sezai, Shigeru Toyoda, Kaoru Dohi, Haruo Kamiya, Keisuke Kida, Toshihisa Anzai, Taishiro Chikamori, Koichi Node, Masayoshi Ajioka, Junya Ako, Rie Amano, Mit
Hypertension Research.2023; 46(2): 495. CrossRef - Ecofriendly appraisal of stability‐indicating high‐performance chromatographic assay of canagliflozin and metformin with their toxic impurities; in silico toxicity prediction
Raghda A. Emam, Aml A. Emam
Journal of Separation Science.2023;[Epub] CrossRef - Effect of sodium‐glucose cotransporter 2 inhibitors on the rate of decline in kidney function: A systematic review and meta‐analysis
Yanbei Duo, Junxiang Gao, Tao Yuan, Weigang Zhao
Journal of Diabetes.2023; 15(1): 58. CrossRef - Quantitative analysis of low content polymorphic impurities in canagliflozin tablets by PXRD, NIR, ATR-FITR and Raman solid-state analysis techniques combined with stoichiometry
Mingdi Liu, Jichao Liu, Qiuhong Wang, Ping Song, Haichao Li, Songgu Wu, Junbo Gong
Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy.2023; 293: 122458. CrossRef - SGLT2 Inhibitors vs. GLP-1 Agonists to Treat the Heart, the Kidneys and the Brain
Bartosz Rolek, Mateusz Haber, Magdalena Gajewska, Sylwester Rogula, Arkadiusz Pietrasik, Aleksandra Gąsecka
Journal of Cardiovascular Development and Disease.2023; 10(8): 322. CrossRef - Cardiorenal Protections of SGLT2 Inhibitors in the Treatment of Type 2
Diabetes
Somayeh Nazari, Hossein Mirkhani
Current Diabetes Reviews.2023;[Epub] CrossRef - Efficacy and Safety of Evogliptin Add-on Therapy to Dapagliflozin/Metformin Combinations in Patients with Poorly Controlled Type 2 Diabetes Mellitus: A 24-Week Multicenter Randomized Placebo-Controlled Parallel-Design Phase-3 Trial with a 28-Week Extensio
Jun Sung Moon, Il Rae Park, Hae Jin Kim, Choon Hee Chung, Kyu Chang Won, Kyung Ah Han, Cheol-Young Park, Jong Chul Won, Dong Jun Kim, Gwan Pyo Koh, Eun Sook Kim, Jae Myung Yu, Eun-Gyoung Hong, Chang Beom Lee, Kun-Ho Yoon
Diabetes & Metabolism Journal.2023; 47(6): 808. CrossRef - Novel perspective on cardiovascular adverse event profiles due to treatment with sodium-glucose transport protein 2 (SGLT2) inhibitors in the Japanese Adverse Drug Event Report (JADER) database
Koji Suematsu
Personalized Medicine Universe.2023; 12: 23. CrossRef - The fate of flavonoids after oral administration: a comprehensive overview of its bioavailability
Abid Naeem, Yang Ming, Hu Pengyi, Kang Yong Jie, Liu Yali, Zhang Haiyan, Xiao Shuai, Li Wenjing, Wu Ling, Zhang Ming Xia, Liu Shan Shan, Zheng Qin
Critical Reviews in Food Science and Nutrition.2022; 62(22): 6169. CrossRef - Add-on therapy with dapagliflozin in routine outpatient care of type 2 diabetes patients from Turkey: a retrospective cohort study on HbA1c, body weight, and blood pressure outcomes
Derun Taner Ertugrul, Erdal Kan, Cigdem Bahadir Tura, Haci Bayram Tugtekin, Hayati Ayakta, Mehmet Celebioglu, Ceren Yılmaz, Onur Utebay, Ilhan Yetkin, Eren Gurkan, Kerem Sezer, Ramazan Gen, Suleyman Ozcaylak, Yildiz Okuturlar, Mehmet Coskun, Nilgun Govec
International Journal of Diabetes in Developing Countries.2022; 42(1): 147. CrossRef - Sodium–glucose cotransporter 2 inhibitors do not increase the risk of fractures in real‐world clinical practice in Korea: A national observational cohort study
Kyoung Hwa Ha, Dae Jung Kim, Yong Jun Choi
Journal of Diabetes Investigation.2022; 13(6): 986. CrossRef - Luseogliflozin preserves the pancreatic beta-cell mass and function in db/db mice by improving mitochondrial function
Yuki Yamauchi, Akinobu Nakamura, Takashi Yokota, Kiyohiko Takahashi, Shinichiro Kawata, Kazuhisa Tsuchida, Kazuno Omori, Hiroshi Nomoto, Hiraku Kameda, Kyu Yong Cho, Toshihisa Anzai, Shinya Tanaka, Yasuo Terauchi, Hideaki Miyoshi, Tatsuya Atsumi
Scientific Reports.2022;[Epub] CrossRef - Blood glucose levels and bodyweight change after dapagliflozin administration
Hyunah Kim, Seung‐Hwan Lee, Hyunyong Lee, Hyeon Woo Yim, Jae‐Hyoung Cho, Kun‐Ho Yoon, Hun‐Sung Kim
Journal of Diabetes Investigation.2021; 12(9): 1594. CrossRef - Dapagliflozin Improves Cardiac Hemodynamics and Mitigates Arrhythmogenesis in Mitral Regurgitation‐Induced Myocardial Dysfunction
Yu‐Wen Lin, Chin‐Yu Chen, Jhih‐Yuan Shih, Bor‐Chih Cheng, Ching‐Ping Chang, Mao‐Tsun Lin, Chung‐Han Ho, Zhih‐Cherng Chen, Sudeshna Fisch, Wei‐Ting Chang
Journal of the American Heart Association.2021;[Epub] CrossRef - Cardiovascular Safety of Sodium Glucose Cotransporter 2 Inhibitors as Add-on to Metformin Monotherapy in Patients with Type 2 Diabetes Mellitus
Ja Young Jeon, Kyoung Hwa Ha, Dae Jung Kim
Diabetes & Metabolism Journal.2021; 45(4): 505. CrossRef - Ipragliflozin, an SGLT2 Inhibitor, Ameliorates High-Fat Diet-Induced Metabolic Changes by Upregulating Energy Expenditure through Activation of the AMPK/ SIRT1 Pathway
Ji-Yeon Lee, Minyoung Lee, Ji Young Lee, Jaehyun Bae, Eugene Shin, Yong-ho Lee, Byung-Wan Lee, Eun Seok Kang, Bong-Soo Cha
Diabetes & Metabolism Journal.2021; 45(6): 921. CrossRef - SGLT2i: beyond the glucose-lowering effect
Lihua Ni, Cheng Yuan, Guopeng Chen, Changjiang Zhang, Xiaoyan Wu
Cardiovascular Diabetology.2020;[Epub] CrossRef - Ipragliflozin Additively Ameliorates Non-Alcoholic Fatty Liver Disease in Patients with Type 2 Diabetes Controlled with Metformin and Pioglitazone: A 24-Week Randomized Controlled Trial
Eugene Han, Yong-ho Lee, Byung-Wan Lee, Eun Seok Kang, Bong-Soo Cha
Journal of Clinical Medicine.2020; 9(1): 259. CrossRef - Stability Indicating, pH and pKa Dependent HPLC–DAD Method for the Simultaneous Determination of Weakly Ionizable Empagliflozin, Dapagliflozin and Canagliflozin in Pharmaceutical Formulations
Shahzad Sharif, Rashida Bashir, Ahmad Adnan, Sabiha Mansoor, Izaz Ahmad, Ayoub Rashid Ch, Muhammad Saqlain Tahir
Chromatographia.2020; 83(12): 1453. CrossRef - Cardiorenal protection with SGLT2: Lessons from the cardiovascular outcome trials
José Silva‐Cardoso, Omar Sheikh, Mouhamed Nashawi, Son Pham, Kelly M. Gallegos, Laith R. Dinkha, Robert J. Chilton
Journal of Diabetes.2020; 12(4): 279. CrossRef - Effect of the Sodium-Glucose Cotransporter 2 Inhibitor, Dapagliflozin, on Genitourinary Infection in an Animal Model of Type 2 Diabetes
Jin Bong Choi, Je Mo Yoo, Ye-Jee Lee, Jae Woong Kim, Seung-Ju Lee, Hee Youn Kim, Dong Sup Lee, Seung-Hyun Ko, Hyun-Sop Choe
International Neurourology Journal.2020; 24(1): 21. CrossRef - Sodium‐glucose cotransporter 2 inhibitors regulate ketone body metabolism via inter‐organ crosstalk
Jin Hee Kim, Minyoung Lee, Soo Hyun Kim, So Ra Kim, Byung‐Wan Lee, Eun Seok Kang, Bong‐Soo Cha, Jin Won Cho, Yong‐ho Lee
Diabetes, Obesity and Metabolism.2019; 21(4): 801. CrossRef - Effects of Sodium-Glucose Cotransporter 2 Inhibitors on Renal Outcomes in Patients with Type 2 Diabetes: A Systematic Review and Meta-Analysis of Randomized Controlled Trials
Jae Hyun Bae, Eun-Gee Park, Sunhee Kim, Sin Gon Kim, Seokyung Hahn, Nam Hoon Kim
Scientific Reports.2019;[Epub] CrossRef - SGLT2 inhibition with empagliflozin attenuates myocardial oxidative stress and fibrosis in diabetic mice heart
Chenguang Li, Jie Zhang, Mei Xue, Xiaoyu Li, Fei Han, Xiangyang Liu, Linxin Xu, Yunhong Lu, Ying Cheng, Ting Li, Xiaochen Yu, Bei Sun, Liming Chen
Cardiovascular Diabetology.2019;[Epub] CrossRef - An update of SGLT1 and SGLT2 inhibitors in early phase diabetes-type 2 clinical trials
Ernest Adeghate, Sahar Mohsin, Faisal Adi, Fares Ahmed, Ali Yahya, Huba Kalász, Kornelia Tekes, Ernest A. Adeghate
Expert Opinion on Investigational Drugs.2019; 28(9): 811. CrossRef - Stability‐indicating chromatographic and chemometric methods for environmentally benign determination of canagliflozin and its major degradation product; A comparative study and greenness assessment
Aml A. Emam, Nada S. Abdelwahab
Biomedical Chromatography.2019;[Epub] CrossRef - A Lower Baseline Urinary Glucose Excretion Predicts a Better Response to the Sodium Glucose Cotransporter 2 Inhibitor
You-Cheol Hwang, Jae Hyeon Kim, Byung-Wan Lee, Woo Je Lee
Diabetes & Metabolism Journal.2019; 43(6): 898. CrossRef - Predictors of the Therapeutic Efficacy and Consideration of the Best Combination Therapy of Sodium-Glucose Co-transporter 2 Inhibitors
Ji-Yeon Lee, Yongin Cho, Minyoung Lee, You Jin Kim, Yong-ho Lee, Byung-Wan Lee, Bong-Soo Cha, Eun Seok Kang
Diabetes & Metabolism Journal.2019; 43(2): 158. CrossRef - Comparative Efficacy and Safety Among Sodium-glucose Cotransporter-2 Inhibitors in Type 2 Diabetes – Results from a Retrospective Single-centre Study
Manash P Baruah, Sanjay Kalra
European Endocrinology.2019; 15(2): 113. CrossRef - Canagliflozin stability study and ecofriendly chromatographic determination of its degradation product: A comparative study
Aml A. Emam
Journal of Separation Science.2018; 41(4): 822. CrossRef - Efficacy and tolerability of novel triple combination therapy in drug-naïve patients with type 2 diabetes from the TRIPLE-AXEL trial: protocol for an open-label randomised controlled trial
Nam Hoon Kim, Soo Lim, Soo Heon Kwak, Min Kyong Moon, Jun Sung Moon, Yong-ho Lee, Ho Chan Cho, Juneyoung Lee, Sin Gon Kim
BMJ Open.2018; 8(9): e022448. CrossRef - Short-term outcomes of patients with Type 2 diabetes mellitus treated with canagliflozin compared with sitagliptin in a real-world setting
YL Shao, KH Yee, SK, Koh, YF Wong, LY Yeoh, S Low, CF Sum
Singapore Medical Journal.2018; 59(5): 251. CrossRef - Clinical implications of current cardiovascular outcome trials with sodium glucose cotransporter-2 (SGLT2) inhibitors
Soo Lim, Robert H. Eckel, Kwang Kon Koh
Atherosclerosis.2018; 272: 33. CrossRef - Effect of Empagliflozin, a Selective Sodium-Glucose Cotransporter 2 Inhibitor, on Kidney and Peripheral Nerves in Streptozotocin-Induced Diabetic Rats
Kyung Ae Lee, Heung Yong Jin, Na Young Lee, Yu Ji Kim, Tae Sun Park
Diabetes & Metabolism Journal.2018; 42(4): 338. CrossRef - The effect of Ramadan fasting and continuing sodium-glucose co-transporter-2 (SGLT2) inhibitor use on ketonemia, blood pressure and renal function in Muslim patients with type 2 diabetes
Yanli Shao, Gwyneth Joy Lim, Chin Lian Chua, Yip Fong Wong, Ester Chai Kheng Yeoh, Serena Kiat Mun Low, Chee Fang Sum
Diabetes Research and Clinical Practice.2018; 142: 85. CrossRef - Effect of Dapagliflozin on Alanine Aminotransferase Improvement in Type 2 Diabetes Mellitus with Non-alcoholic Fatty Liver Disease
Dug-Hyun Choi, Chan-Hee Jung, Ji-Oh Mok, Chul-Hee Kim, Sung-Koo Kang, Bo-Yeon Kim
Endocrinology and Metabolism.2018; 33(3): 387. CrossRef - Comparison between sodium–glucose cotransporter 2 inhibitors and pioglitazone as additions to insulin therapy in type 2 diabetes patients: A systematic review with an indirect comparison meta‐analysis
Yun Kyung Cho, Ye‐Jee Kim, Yu Mi Kang, Seung Eun Lee, Joong‐Yeol Park, Woo Je Lee, Chang Hee Jung
Journal of Diabetes Investigation.2018; 9(4): 882. CrossRef - Characteristics of Dapagliflozin Responders: A Longitudinal, Prospective, Nationwide Dapagliflozin Surveillance Study in Korea
Eugene Han, Ari Kim, Sung Jae Lee, Je-Yon Kim, Jae Hyeon Kim, Woo Je Lee, Byung-Wan Lee
Diabetes Therapy.2018; 9(4): 1689. CrossRef - Combining SGLT2 Inhibition With a Thiazolidinedione Additively Attenuate the Very Early Phase of Diabetic Nephropathy Progression in Type 2 Diabetes Mellitus
Eugene Han, Eugene Shin, Gyuri Kim, Ji-Yeon Lee, Yong-ho Lee, Byung-Wan Lee, Eun Seok Kang, Bong-Soo Cha
Frontiers in Endocrinology.2018;[Epub] CrossRef - Clinical parameters affecting dapagliflozin response in patients with type 2 diabetes
J.-Y. Lee, G. Kim, S.R. Kim, Y.-H. Lee, B.-W. Lee, B.-S. Cha, E.S. Kang
Diabetes & Metabolism.2017; 43(2): 191. CrossRef - Glucagon and glucagon-like peptide-1 as novel anti-inflammatory and immunomodulatory compounds
Daniella B.R. Insuela, Vinicius F. Carvalho
European Journal of Pharmacology.2017; 812: 64. CrossRef - Efficacy and safety of tofogliflozin in Japanese patients with type 2 diabetes mellitus with inadequate glycaemic control on insulin therapy (J‐STEP/INS): Results of a 16‐week randomized, double‐blind, placebo‐controlled multicentre trial
Yasuo Terauchi, Masahiro Tamura, Masayuki Senda, Ryoji Gunji, Kohei Kaku
Diabetes, Obesity and Metabolism.2017; 19(10): 1397. CrossRef - A comparison of effects of DPP-4 inhibitor and SGLT2 inhibitor on lipid profile in patients with type 2 diabetes
Seon-Ah Cha, Yong-Moon Park, Jae-Seung Yun, Tae-Seok Lim, Ki-Ho Song, Ki-Dong Yoo, Yu-Bae Ahn, Seung-Hyun Ko
Lipids in Health and Disease.2017;[Epub] CrossRef - Morning Spot Urine Glucose-to-Creatinine Ratios Predict Overnight Urinary Glucose Excretion in Patients With Type 2 Diabetes
So Ra Kim, Yong-ho Lee, Sang-Guk Lee, Sun Hee Lee, Eun Seok Kang, Bong-Soo Cha, Hyun Chul Lee, Jeong-Ho Kim, Byung-Wan Lee
Annals of Laboratory Medicine.2017; 37(1): 9. CrossRef - The Relationship between Increases in Morning Spot Urinary Glucose Excretion and Decreases in HbA1C in Patients with Type 2 Diabetes After Taking an SGLT2 Inhibitor: A Retrospective, Longitudinal Study
So Ra Kim, Yong-ho Lee, Eun Seok Kang, Bong-Soo Cha, Byung-Wan Lee
Diabetes Therapy.2017; 8(3): 601. CrossRef - Clinical and Genetic Features of Patients With Type 2 Diabetes and Renal Glycosuria
Siqian Gong, Jiandong Guo, Xueyao Han, Meng Li, Lingli Zhou, Xiaoling Cai, Yu Zhu, Yingying Luo, Simin Zhang, Xianghai Zhou, Yumin Ma, Linong Ji
The Journal of Clinical Endocrinology & Metabolism.2017; 102(5): 1548. CrossRef - Effect of Sodium-Glucose Co-Transporter 2 Inhibitor, Dapagliflozin, on Renal Renin-Angiotensin System in an Animal Model of Type 2 Diabetes
Seok Joon Shin, Sungjin Chung, Soo Jung Kim, Eun-Mi Lee, Young-Hye Yoo, Ji-Won Kim, Yu-Bae Ahn, Eun-Sook Kim, Sung-Dae Moon, Myung-Jun Kim, Seung-Hyun Ko, Kwang-Hyun Baek
PLOS ONE.2016; 11(11): e0165703. CrossRef - Sodium–glucose cotransporter 2 inhibitors: an evidence-based practice approach to their use in the natural history of type 2 diabetes
Stanley S. Schwartz, Intekhab Ahmed
Current Medical Research and Opinion.2016; 32(5): 907. CrossRef - Insulin resistance, role of metformin and other non-insulin therapies in pediatric type 1 diabetes
Fida Bacha, Sara Klinepeter Bartz
Pediatric Diabetes.2016; 17(8): 545. CrossRef - Renal threshold for glucose reabsorption predicts diabetes improvement by sodium‐glucose cotransporter 2 inhibitor therapy
Aya Osaki, Shuichi Okada, Tsugumichi Saito, Eijiro Yamada, Kumeo Ono, Yawara Niijima, Hiroto Hoshi, Masanobu Yamada
Journal of Diabetes Investigation.2016; 7(5): 751. CrossRef - Sodium-glucose co-transporter inhibition in the treatment of diabetes: Sweetening the pot
Carlos A Alvarez, Ian J Neeland, Darren K McGuire
Diabetes and Vascular Disease Research.2015; 12(2): 74. CrossRef - A simple and sensitive high performance liquid chromatography assay with a fluorescence detector for determination of canagliflozin in human plasma
Muzaffar Iqbal, Nasr Y. Khalil, Amer M. Alanazi, Khalid A. Al-Rashood
Analytical Methods.2015; 7(7): 3028. CrossRef - Efficacy and safety of sodium–glucose cotransporter 2 inhibitors in type 2 diabetes: a meta-analysis of randomized controlled trials for 1 to 2 years
Xiang-Yang Liu, Ning Zhang, Rui Chen, Jia-Guo Zhao, Pei Yu
Journal of Diabetes and its Complications.2015; 29(8): 1295. CrossRef - Severe hypercalcemia and hypernatremia in a patient treated with canagliflozin
Arshpreet Kaur, Stephen J Winters
Endocrinology, Diabetes & Metabolism Case Reports.2015;[Epub] CrossRef - Pharmacological treatment and therapeutic perspectives of metabolic syndrome
Soo Lim, Robert H. Eckel
Reviews in Endocrine and Metabolic Disorders.2014; 15(4): 329. CrossRef
- Figure
- Related articles
-
- Blood Pressure Target in Type 2 Diabetes Mellitus
- Not Control but Conquest: Strategies for the Remission of Type 2 Diabetes Mellitus
- Association between Type 2 Diabetes Mellitus and Brain Atrophy: A Meta-Analysis
- Real-World Analysis of Therapeutic Outcome in Type 1 Diabetes Mellitus at a Tertiary Care Center

 KDA
KDA
 PubReader
PubReader Cite
Cite