- Current
- Browse
- Collections
-
For contributors
- For Authors
- Instructions to authors
- Article processing charge
- e-submission
- For Reviewers
- Instructions for reviewers
- How to become a reviewer
- Best reviewers
- For Readers
- Readership
- Subscription
- Permission guidelines
- About
- Editorial policy
Articles
- Page Path
- HOME > Diabetes Metab J > Volume 46(3); 2022 > Article
-
Original ArticleCOVID-19 Association of Metabolic Syndrome with COVID-19 in the Republic of Korea
-
Woo-Hwi Jeon1
 , Jeong-Yeon Seon1, So-Youn Park2
, Jeong-Yeon Seon1, So-Youn Park2 , In-Hwan Oh1
, In-Hwan Oh1
-
Diabetes & Metabolism Journal 2022;46(3):427-438.
DOI: https://doi.org/10.4093/dmj.2021.0105
Published online: November 26, 2021
1Department of Preventive Medicine, College of Medicine, Kyung Hee University, Seoul, Korea
2Department of Medical Education and Humanities, College of Medicine, Kyung Hee University, Seoul, Korea
-
Corresponding authors: So-Youn Park
 Department of Medical Education and Humanities, College of Medicine, Kyung Hee University, 23 Kyungheedae-ro, Dongdaemun-gu, Seoul 02447, Korea E-mail: ukii77@gmail.com
Department of Medical Education and Humanities, College of Medicine, Kyung Hee University, 23 Kyungheedae-ro, Dongdaemun-gu, Seoul 02447, Korea E-mail: ukii77@gmail.com -
In-Hwan Oh
 Department of Preventive Medicine, College of Medicine, Kyung Hee University, 23 Kyungheedae-ro, Dongdaemun-gu, Seoul 02447, Korea E-mail: parenchyme@gmail.com
Department of Preventive Medicine, College of Medicine, Kyung Hee University, 23 Kyungheedae-ro, Dongdaemun-gu, Seoul 02447, Korea E-mail: parenchyme@gmail.com
Copyright © 2022 Korean Diabetes Association
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/4.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
ABSTRACT
-
Background
- Metabolic syndrome (MetS) is reportedly a crucial risk factor for coronavirus disease 2019 (COVID-19). Since the epidemiological studies that examine this association are few and include small samples, we investigated the relationship between MetS and COVID-19 severity and death using a larger sample in the Republic of Korea.
-
Methods
- We analyzed 66,321 patients, 4,066 of whom had COVID-19. We used chi-square tests to examine patients’ characteristics. We performed logistic regression analysis to analyze differences in COVID-19 infection and clinical outcomes according to the presence of MetS.
-
Results
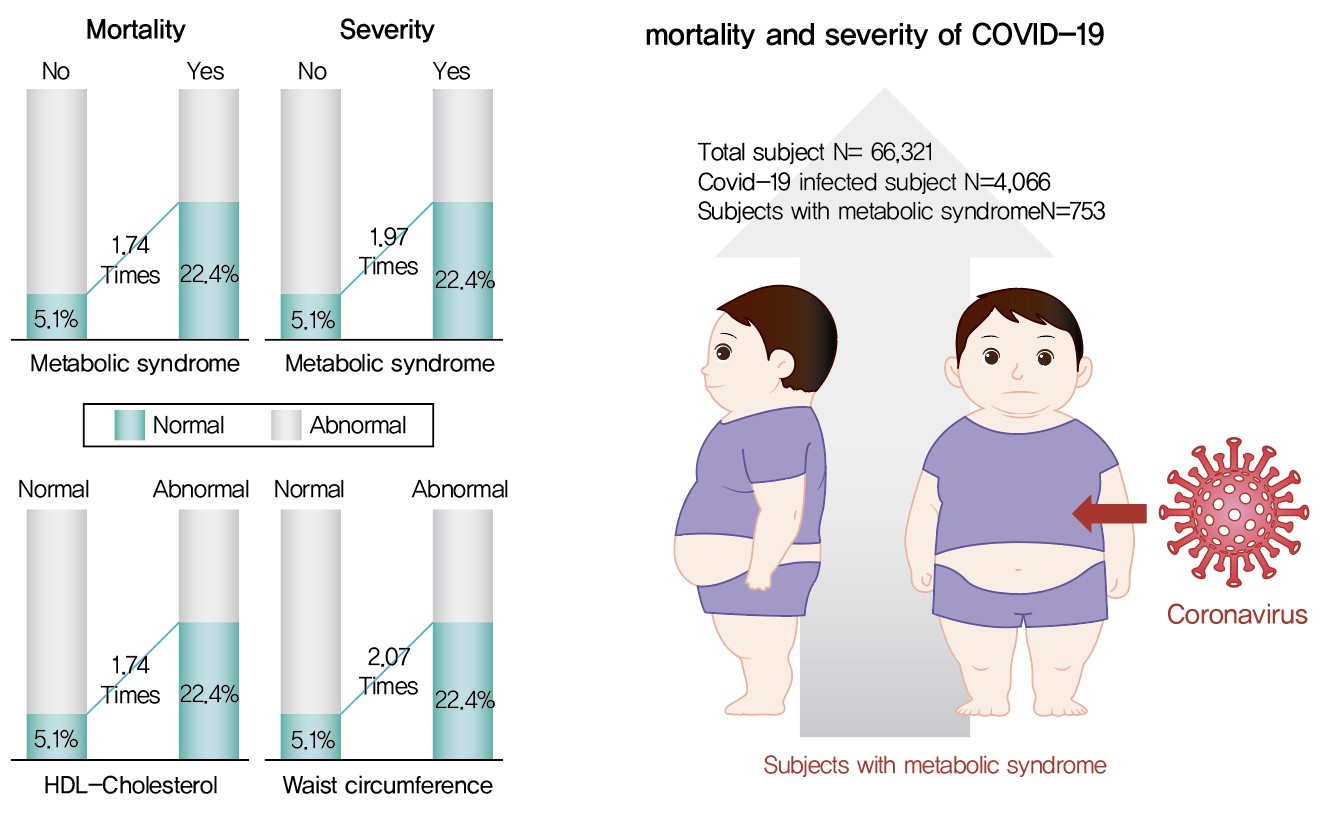
- Although MetS was not significantly associated with COVID-19 risk, acquiring MetS was significantly associated with the risk of severe COVID-19 outcomes (odds ratio [OR], 1.97; 95% confidence interval [CI], 1.34 to 2.91; P=0.001). The mortality risk was significantly higher in COVID-19 patients with MetS (OR, 1.74; 95% CI, 1.17 to 2.59; P=0.006). Patients with abnormal waist circumference were approximately 2.07 times more likely to develop severe COVID-19 (P<0.001), and high-density lipoprotein cholesterol (HDL-C) levels were significantly associated with COVID-19; the mortality risk due to COVID-19 was 1.74 times higher in men with an HDL-C level of <40 mg/dL and in women with an HDL-C level of <50 mg/dL (P=0.012).
-
Conclusion
- COVID-19 is likely associated with severity and death in patients with MetS or in patients with MetS risk factors. Therefore, patients with MetS or those with abnormal waist circumference and HDL-C levels need to be treated with caution.
- As of January 13, 2021, there were more than 90 million cases positive for coronavirus disease 2019 (COVID-19) and approximately 2 million deaths globally since the first reported case in December 2019 in Wuhan, China [1]. Although COVID-19 appeared to be under control in the Republic of Korea, the number of cases showed an upward trend from November 2020, resulting in a cumulative total of 70,212 cases and 1,185 deaths as of January 13, 2021 [2]. During the prolonged pandemic period spanning over a year, several studies reported that old age, chronic disease, and poor lifestyle are risk factors associated with increased severity of COVID-19. Of these risk factors, chronic disease has been identified as a major cause of high mortality in COVID-19 patients [3]. According to a study conducted by the University of Pennsylvania, mortality was higher among COVID-19 patients with comorbidities such as cardiovascular disease, hypertension, diabetes mellitus, kidney disease, and cancer than among other COVID-19 patients [4]. Furthermore, the Global Burden of Disease Study led by the Institute for Health Metrics and Evaluation, recently published in The Lancet, states that chronic diseases that cause a high disease burden, such as ischemic heart disease, diabetes mellitus, and stroke, have significantly contributed to the high mortality of COVID-19 globally. Particularly, they reported that the high disease burden of chronic diseases is caused by the escalating prevalence of metabolic syndrome (MetS) [5].
- Although the exact etiology of MetS is unknown, lifestyle factors such as irregular and poor diet, smoking, drinking, overwork, sleep deprivation, and stress and insulin resistance are the major causes of MetS. In general, an individual is diagnosed with MetS if three of the following five symptoms are present: abdominal obesity (waist circumference ≥90 cm in men, ≥85 cm in women), high blood pressure (≥130/85 mm Hg), high blood glucose levels (≥100 mg/dL), high triglyceride levels (≥150 mg/dL), and low levels of high-density lipoprotein cholesterol (HDL-C; levels of <40 mg/dL in men, <50 mg/dL in women) [6]. While there are no characteristic symptoms of MetS, there is a higher prevalence of stroke, cancer, arteriosclerosis, diabetes mellitus, and cardiovascular disease among patients with MetS than among the general population; consequently, there is an increased mortality rate among patients with MetS [7-9].
- Recent studies have reported that MetS, a major cause of chronic diseases, is associated with the severity of COVID-19. A research team at Tulane University School of Medicine published a study showing that individuals with MetS who have a diagnosis of COVID-19 are more likely to have a poor prognosis. Furthermore, according to an analysis on 287 COVID-19 patients admitted to two hospitals in New Orleans, the odds of death due to COVID-19 was 3.4 times higher in patients with MetS than in others, while the odds for intensive care unit (ICU) treatment, ventilator support, or acute respiratory distress syndrome was approximately 5 times higher in COVID-19 patients with MetS than in those without MetS [10]. Researchers believe that vascular dysfunction and impaired adipocyte regulation caused by COVID-19 in patients with MetS elevate the risk of increased severity of COVID-19 [11,12].
- The Republic of Korea is one of the countries with a high and consistently increasing prevalence of MetS. As of 2015, MetS prevalence in the Republic of Korea was 22.4% [13]. A study that analyzed changes in the prevalence of MetS based on the data from the Korean National Health and Nutrition Examination Survey from 2008 to 2017 observed a marked elevation in MetS prevalence in men from 24.5% in 2008 to 28.1% in 2017, and a slight reduction in MetS prevalence in women, though still high, from 20.5% in 2008 to 18.7% in 2017 [6]. Although MetS prevalence is high and may contribute toward increased mortality from COVID-19 [12], only a few epidemiological studies have analyzed the association between MetS and COVID-19 in the Republic of Korea, and the majority of studies conducted abroad have been small-sample studies. Hence, it is necessary to investigate the effects of MetS on COVID-19 using a national database. Therefore, this study aimed to analyze the association between MetS and COVID-19 severity and mortality using a national COVID-19 database.
INTRODUCTION
- Ethics statement
- This study conformed to the Guidelines on De-identification of Personal Data of Korea and was approved by the Kyung Hee University’s Institutional Review Board (IRB No. KHSIRB-20-301[EA]) as a review exemption study. As the study used de-identified data, the requirement for informed consent was waived by the board.
- Data and study participants
- The associations among COVID-19, clinical outcome, and MetS were analyzed using the National Health Insurance Service COVID Database, which contains medical records from January 2015 to July 2020, including those of 129,120 patients who visited hospitals in the Republic of Korea between January 1, 2020, and May 30, 2020 [14]. Patients in the database were categorized as COVID-19 and non-COVID-19 patients, and stratified random sampling was performed at a 1:15 ratio. The stratifying factors used were sex, age, and area of residence. In this study, patients aged <20 years and those with missing essential health examination-related data (waist circumference, blood pressure, triglyceride levels, HDL-C levels, and fasting blood glucose levels) were excluded, resulting in a total of 66,321 patients in the final analysis. Of the total, 62,255 were non-COVID-19 patients, while 4,066 were COVID-19 patients (Fig. 1).
- Clinical factors and outcomes
- Clinical outcomes include ventilator support and death in COVID-19 patients, while general characteristics included sex, age, area of residence, health insurance premium, Charlson Comorbidity Index (CCI), normal levels of MetS components (waist circumference, triglyceride level, HDL-C level, blood pressure, and fasting glucose level), number of MetS components in the abnormal range, and MetS diagnosis. Health insurance premiums were used as indicators of economic status as the premiums in the Republic of Korea are set proportionate to income. Medical aid recipients do not pay health insurance premiums, and health insurance subscribers who pay a premium were classified into the first quintile (lowest), second quintile, third quintile, fourth quintile, and fifth quintile (highest). The CCI was weighted using the updated criteria proposed by Quan et al. [15] based on the 2019 main diagnosis. The most up-to-date clinical information was collected from the 2015 to 2018 health examination data for the diagnosis of MetS. The normal ranges of the five components of MetS were determined based on the following clinical criteria: waist circumference (men ≥90 cm, women ≥85 cm), triglyceride level (≥150 mg/dL), HDL-C level (male <40 mg/dL, female <50 mg/dL), blood pressure (≥130/85 mm Hg or a diagnosis of hypertension in 2019), and blood glucose level (≥100 mg/L or a diagnosis of diabetes mellitus in 2019). Additionally, the number of the aforementioned clinical factors in the abnormal range was calculated, and patients with three or more factors in the abnormal range were defined as MetS patients.
- Statistical analyses
- The risk factors for COVID-19 and clinical outcomes were analyzed in relation to the participants’ general characteristics using chi-square tests. Logistic regression was used to analyze the association between COVID-19 or clinical outcomes (ventilator support or death) and MetS, MetS criteria, and other patient characteristics. Sex, age, area of residence, and health insurance premium were adjusted in all logistic regression models. Because MetS is a complex syndrome of several adult diseases, the diagnosis of the condition was deemed to include information about various underlying diseases; thus, CCI was not adjusted for in the logistic regression models.
- We established logistic regression models while considering the adjustment variables and the diagnosis of MetS to examine the risk of COVID-19 in patients with MetS. Ventilator support and death were set as the clinical outcome variables for COVID-19 in patients with MetS. The clinical MetS components were individually considered to identify specific risk factors for MetS that were associated with COVID-19 and mortality. Statistical analyses were performed using SAS version 9.4 (SAS Institute, Cary, NC, USA), and the level of significance was set at 5% for all statistical analyses.
METHODS
- Analysis of target patient characteristics
- The baseline characteristics of COVID-19 patients are shown in Table 1. Among the various characteristics of participants, age, health insurance premium, CCI, waist circumference, HDL-C level, and fasting glucose levels were significantly associated with COVID-19. COVID-19 rate ranged from 4.1% to 6.6% across age groups, with the lowest rate in the 20 to 29 years group and the highest rate in the 50 to 59 years group. Regarding economic status, the infection rate was highest (12.2%) among medical aid recipients, who are considered to have the lowest economic status. The COVID-19 rate tended to increase with increasing CCI. Furthermore, the COVID-19 rate was high among patients with abnormal waist circumference (men ≥90 cm, women ≥85 cm, 6.5%), HDL-C level (male <40 mg/dL, female <50 mg/dL, 6.7%), and fasting glucose level (6.4%).
- The association of COVID-19 patient characteristics with ventilator support and death was analyzed using chi-square tests (Table 1). The use of ventilator support indicated severe COVID-19 symptoms. The severity of COVID-19 symptoms differed based on sex, age, area of residence, CCI, waist circumference, triglyceride levels, blood pressure, fasting glucose levels, number of MetS components, and MetS diagnosis. The proportion of patients with severe symptoms was approximately three times higher among men (5.2%) than among women and tended to increase with advancing age. The proportion of severe COVID-19 patients was highest in the North Gyeongsang Province region (5.9%) when compared with that in all other regions, and it increased with increasing CCI. The proportion of severe COVID-19 patients was higher among those with MetS (6.5%) than among those without MetS and was the lowest among patients without any MetS components (1.1%). Moreover, the proportion of severe COVID-19 patients was higher among those with abnormal waist circumference (6.2%), triglyceride levels (4.8%), and fasting glucose levels (4.6%) than among COVID-19 patients with normal corresponding parameters.
- The mortality rate was more than threefold higher among male COVID-19 patients (6.1%) than among female patients. No deaths were observed in COVID-19 patients in their 20s and 30s, while the mortality rate was highest among patients aged ≥80 years (28.6%). Similar to the proportion of severe COVID-19 patients, the mortality rate was highest in the North Gyeongsang Province region (6.7%), and this increased with increasing CCI, indicating the severity of underlying diseases. The COVID-19 mortality rate was approximately threefold higher among those with MetS than among those without MetS and tended to increase with an increasing number of MetS components. Additionally, the mortality rate was higher among patients with abnormal waist circumference (5.8%), HDL-C levels (5.4%), blood pressure (5.2%), and fasting glucose levels (5.3%).
- Odds ratio for the risk of COVID-19 and clinical outcomes
- Multivariate logistic regression analysis was performed to analyze the difference between COVID-19 infection and clinical outcome according to the presence of MetS, and all input variables were adjusted (Table 2). In the model with COVID-19 infection as an outcome variable, age and health insurance premiums were found to have a significant effect on COVID-19 infection (P<0.001), but while MetS did not show this significant association.
- The associations between MetS and the clinical outcomes of ventilator support and death were analyzed using logistic regression (Table 2). The results showed that sex, age, and MetS were significantly associated with ventilator support. Female COVID-19 patients were less likely to require ventilator support than their male counterparts (odds ratio [OR], 0.36; 95% confidence interval [CI], 0.24 to 0.52; P<0.001), and the likelihood of needing ventilator support increased with advancing age (P<0.001). Moreover, experiencing MetS was significantly associated with ventilator support (OR, 1.97; 95% CI, 1.34 to 2.91; P=0.001).
- Logistic regression analysis was performed to identify risk factors affecting mortality in COVID-19 patients. As a result of the analysis, sex, age, health insurance premium, and MetS were analyzed to be associated with COVID-19 death. There was a lower likelihood of deaths among women than among men (OR, 0.26; 95% CI, 0.17 to 0.39; P<0.001), and the likelihood of death increased with advancing age (P<0.001). Regarding health insurance premiums, patients in the first quantile (OR, 0.43; 95% CI, 0.20 to 0.91; P=0.028) or fifth quantile (OR, 0.50; 95% CI, 0.25 to 0.99; P=0.046) were less likely to succumb to COVID-19 than those receiving medical aid. Patients with MetS were more likely to die than those without MetS (OR, 1.74; 95% CI, 1.17 to 2.59; P=0.006).
- In the logistic regression model, which set whether to wear a entilator as an outcome variable, it was found that patients with MetS were more likely to wear a ventilator. Logistic regression analyses were used to identify the specific MetS components that were associated with the clinical outcomes of COVID-19 (Table 3). Consistent with previous models, we adjusted for factors including sex, age, area of residence, and health insurance premium. Patients with abnormal waist circumference were more likely to require ventilator support (OR, 2.07; 95% CI, 1.41 to 3.02; P<0.001). Patients with an abnormal HDL-C level were more likely to expire (OR, 1.74; 95% CI, 1.13 to 2.66; P=0.012).
RESULTS
- This study analyzed the association between MetS and COVID-19 severity and mortality in 66,321 patients in the Republic of Korea between January 1, 2020, and August 14, 2020; among them, 4,066 were diagnosed with COVID-19.
- First, regarding the susceptibility of MetS patients to COVID-19, the results were not statistically significant, suggesting that MetS is not associated with COVID-19. However, the odds of progressing toward severe COVID-19 were 1.97 times higher among those with MetS than among those without MetS (P=0.0001), while the odds of death were 1.74 times higher among those with MetS than among those without MetS (P=0.006).
- A previous study in the USA reported that MetS predicts infection severity and the associated mortality in COVID-19. A study in New Orleans, USA, on 287 COVID-19 in-patients examined the correlation of MetS and its components (hypertension, obesity, diabetes mellitus, triglyceride level, and HDL-C level) with the severity of COVID-19 and found that patients with MetS were more likely to progress toward severe COVID-19 and die due to COVID-19 than patients without MetS [10]. While hypertension, obesity, diabetes mellitus, and low HDL-C predicted the severity of COVID-19, they were not associated with mortality rate. Another recent study conducted in the USA analyzed the correlation between COVID-19 and the risk factors for MetS based on electronic medical records from 1999 to 2019 and revealed that the odds of death from COVID-19 and progression to severe COVID-19 were higher among COVID-19 patients with MetS than among those without MetS [16]. Regarding the MetS components, dyslipidemia, diabetes mellitus, and obesity predicted the severity of COVID-19 and the associated death.
- There are a few possible reasons underlying the higher risk of severe COVID-19 and death due to COVID-19 in patients with MetS. First, as previously stated, MetS is a risk factor for several diseases such as diabetes mellitus, obesity, ischemic heart disease, and stroke. As patients with MetS concurrently present with at least three of five components (abdominal obesity, hypertension, glucose impairment, high triglyceride level, and low HDL-C level), the presence of chronic diseases is highly likely. Many studies reported that metabolic disorders such as diabetes mellitus and obesity increase COVID-19-related mortality [4,17]. Particularly, COVID-19 was found to be 57% more fatal in patients with diabetes mellitus than in others [18]. Thus, comorbidities induced by MetS may have significantly contributed to the severity of and death due to COVID-19.
- Second, one of the causes of severe COVID-19 is the excessive inflammatory response triggered by cytokines, such as interferon, which exerts antiviral activity [12,19]. Pathologically, although patients with MetS have elevated levels of inflammatory cytokines such as tumor necrosis factor-α and interferon-6 owing to abnormal adipokine production [20], they have relatively low adiponectin levels [21]. Owing to this chronic inflammatory state, patients with MetS develop more severe inflammatory responses on contracting COVID-19 [22], which increases the severity and mortality rate of COVID-19 in patients with MetS.
- Infection severity and mortality in COVID-19 were found to be affected by MetS. Hence, to examine the association of each MetS component (waist circumference, triglyceride level, HDL-C level, blood pressure, and fasting glucose level) with severity and mortality in COVID-19, we conducted regression analyses with each component.
- Regarding COVID-19 severity, patients with abnormal waist circumference, men with a waist circumference of ≥90 cm and women with a waist circumference of ≥85 cm, were approximately 2.07 times more likely to develop severe COVID-19 (P<0.001). Waist circumference is the most typical indicator of obesity [23], and having a large waist circumference indicates that the individual has serious visceral and abdominal obesity. Persistent visceral obesity hinders the normal insulin mechanism in the body, thereby increasing levels of inflammatory factors and inducing the development of various diseases such as diabetes mellitus and lung disease [24]. Moreover, an abnormal waist circumference may be fatal not only for diseases such as influenza, pneumonia, and coronary heart disease but also for COVID-19. A British study that analyzed obesity and COVID-19-related mortality by sex using the biobank data reported that male and female patients with a large waist circumference were 1.36 and 1.66 times more likely to die due to COVID-19, respectively [25]. Moreover, an analysis of the association between visceral obesity and COVID-19 severity using the same data source and visceral obesity criteria of ≥102 cm for men and ≥88 cm for women found that patients with visceral obesity were 1.84 times more likely to progress toward severe COVID-19 (P<0.001) [26]. Another study analyzed the association between obesity and COVID-19 using waist circumference as an indicator of obesity along with body mass index. According to a meta-analysis of 75 studies in 10 countries in Asia, North America, and Europe, obese patients were 74% more likely to be admitted to the ICU due to COVID-19 and 48% more likely to die due to COVID-19 than their normal-weight counterparts [27]. The reason for the detrimental effect of waist circumference on COVID-19 may be the same as that for the fatality of obesity on COVID-19—as mentioned earlier, waist circumference is an indicator of obesity. This may be because the accumulation of fat tissue in the abdomen elevates the diaphragm, and the muscles inferior to the thoracic cage compress the lungs, thus hindering airflow within the lungs [28].
- Regarding the risk of death from COVID-19 in relation to each MetS component, HDL-C alone was significantly associated, wherein the risk for death due to COVID-19 was 1.74 times higher in men with HDL-C <40 mg/dL and in women with HDL-C <50 mg/dL (P=0.012). A strong correlation between HDL-C levels and COVID-19 severity and mortality has been reported in several studies. A Chinese study on the correlation between HDL-C and COVID-19 in 228 COVID-19 inpatients of the Public Health Treatment Center of Changsha from January to March 2020 reported that the risk of severe COVID-19 was 2.8 times higher among patients with low HDL-C levels than among those with normal HDL-C levels [29]. Furthermore, an analysis of the COVID-19 clearance rate and HDL-C levels performed at the Beijing University School of Medicine identified that the time until the elimination of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) from the body was more than three-fold longer, indicating that recovery takes longer in patients with low HDL-C levels [30]. Moreover, a low HDL-C concentration impacts COVID-19-related mortality. A study that examined lipid profile and its association with COVID-19-related mortality in 99 COVID-19 patients between February and April 2020 found that the odds of progression to severe infection and death were higher among patients with low HDL-C levels [31]. HDL influences immune cells involved in innate immune responses, and it coordinates inflammatory responses and the antigen-presenting functions of macrophages. Furthermore, it activates T-cells and regulates the functions of macrophages and lymphocytes upon the onset of inflammatory disease. A low concentration of HDL-C—which plays a pivotal role in immune responses—may increase the risk of death or progression to severe infection upon exposure to SARS-CoV-2 because of impaired regulation of the innate immune response [32,33].
- This study has a few limitations. The 2015 to 2018 health examination records were used to collect data on the diagnosis of MetS; although the latest test results were used, relatively older data were used for patients without recent diagnostic records. Additionally, the varying years in which the diagnosis was provided may potentially lead to errors. Finally, exposure to SARS-CoV-2 or contact with affected individuals are an important factor in the outcome of the COVID-19 incidence. But these variables are really hard to control. It is a limitation that this part has not been corrected in our study. Nevertheless, this study is significant for using an adequate sample size with a national database to shed light on the association between COVID-19 and MetS, a risk factor for COVID-19, and for presenting reliable epidemiological data and patient management criteria.
- This study showed that COVID-19 is likely to have a fatal outcome in patients diagnosed with MetS or with risk factors related to MetS. Therefore, it is important for patients with MetS or patients who have abnormal waist circumference and HDL-C levels, which were the identified risk factors in this study, to take all possible measures to prevent the development of COVID-19. If patients with MetS contract COVID-19, then intensive treatment is necessary to prevent disease progression. Although it was not examined as a major outcome, the severity and mortality of COVID-19 were higher in males and in older patients. Thus, in addition to the management of individual risk factors confirmed in this study, older male patients with MetS should be given priority during treatment. Finally, as MetS is caused by poor lifestyle behaviors, such as a poor diet, smoking, drinking, overwork, and stress, along with environmental factors, it is crucial to prevent MetS through lifestyle changes. Detecting MetS through periodic health examinations and national campaigns to encourage proper exercise and diet would not only promote public health but also further protect people from contracting chronic diseases and infectious diseases such as COVID-19.
DISCUSSION
-
CONFLICTS OF INTEREST
No potential conflict of interest relevant to this article was reported.
-
AUTHOR CONTRIBUTIONS
Conception or design: W.H.J.
Acquisition, analysis, or interpretation of data: J.Y.S.
Drafting the work or revising: W.H.J., S.Y.P., I.H.O.
Final approval of the manuscript: W.H.J., J.Y.S., S.Y.P., I.H.O.
-
FUNDING
This research was supported by a grant of the Korea Health Technology R&D Project through the Korea Health Industry Development Institute (KHIDI), funded by the Ministry of Health & Welfare, Republic of Korea (grant number: HI20C1068)
NOTES
-
Acknowledgements
- None
- 1. World Health Organization: Coronavirus disease (COVID-19): situation report 2020. Available from: https://www.who.int/emergencies/diseases/novel-coronavirus-2019/situation-reports (cited 2021 Oct 27).
- 2. Korea Centers for Disease Control & Prevention: The updates on COVID-19 in Korea as of 13 January. Available from: http://ncov.mohw.go.kr/tcmBoardView.do?brdId=3&brdGubun=31&dataGubun=&ncvContSeq=4620&contSeq=4620&board_id=312&gubun=ALL#/ (cited 2021 Oct 27).
- 3. Aggarwal G, Cheruiyot I, Aggarwal S, Wong J, Lippi G, Lavie CJ, et al. Association of cardiovascular disease with coronavirus disease 2019 (COVID-19) severity: a meta-analysis. Curr Probl Cardiol 2020;45:100617.ArticlePubMedPMC
- 4. Ssentongo P, Ssentongo AE, Heilbrunn ES, Ba DM, Chinchilli VM. Association of cardiovascular disease and 10 other preexisting comorbidities with COVID-19 mortality: a systematic review and meta-analysis. PLoS One 2020;15:e0238215.ArticlePubMedPMC
- 5. GBD 2019 Viewpoint Collaborators. Five insights from the Global Burden of Disease Study 2019. Lancet 2020;396:1135-59.PubMedPMC
- 6. Kim MH, Lee SH, Shin KS, Son DY, Kim SH, Joe H, et al. The change of metabolic syndrome prevalence and its risk factors in Korean adults for decade: Korea National Health and Nutrition Examination Survey for 2008-2017. Korean J Fam Pract 2020;10:44-52.Article
- 7. Galassi A, Reynolds K, He J. Metabolic syndrome and risk of cardiovascular disease: a meta-analysis. Am J Med 2006;119:812-9.ArticlePubMed
- 8. Malik S, Wong ND, Franklin SS, Kamath TV, L’Italien GJ, Pio JR, et al. Impact of the metabolic syndrome on mortality from coronary heart disease, cardiovascular disease, and all causes in United States adults. Circulation 2004;110:1245-50.ArticlePubMed
- 9. Ford ES. Risks for all-cause mortality, cardiovascular disease, and diabetes associated with the metabolic syndrome: a summary of the evidence. Diabetes Care 2005;28:1769-78.PubMed
- 10. Xie J, Zu Y, Alkhatib A, Pham TT, Gill F, Jang A, et al. Metabolic syndrome and COVID-19 mortality among adult black patients in New Orleans. Diabetes Care 2020;44:188-93.ArticlePubMedPMCPDF
- 11. Yanai H. Metabolic syndrome and COVID-19. Cardiol Res 2020;11:360-5.ArticlePubMedPMC
- 12. Enkhtur A, Yoon JS, Lee CW. Factors increasing the risk of mortality and morbidity due to coronavirus infection in patients with metabolic syndrome. Precis Future Med 2020;4:83-90.ArticlePDF
- 13. Huh JH, Kang DR, Jang JY, Shin JH, Kim JY, Choi S, et al. Metabolic syndrome epidemic among Korean adults: Korean survey of cardiometabolic syndrome (2018). Atherosclerosis 2018;277:47-52.ArticlePubMed
- 14. National Health Insurance: National Health Insurance Sharing Service 2020. Available from: https://nhiss.nhis.or.kr/bd/ay/bdaya001iv.do (cited 2021 Oct 27).
- 15. Quan H, Li B, Couris CM, Fushimi K, Graham P, Hider P, et al. Updating and validating the Charlson comorbidity index and score for risk adjustment in hospital discharge abstracts using data from 6 countries. Am J Epidemiol 2011;173:676-82.ArticlePubMed
- 16. Ghoneim S, Butt MU, Hamid O, Shah A, Asaad I. The incidence of COVID-19 in patients with metabolic syndrome and non-alcoholic steatohepatitis: a population-based study. Metabol Open 2020;8:100057.ArticlePubMedPMC
- 17. Onder G, Rezza G, Brusaferro S. Case-fatality rate and characteristics of patients dying in relation to COVID-19 in Italy. JAMA 2020;323:1775-6.ArticlePubMed
- 18. Bhatraju PK, Ghassemieh BJ, Nichols M, Kim R, Jerome KR, Nalla AK, et al. COVID-19 in critically ill patients in the Seattle region: case series. N Engl J Med 2020;382:2012-22.ArticlePubMedPMC
- 19. Lee JS, Park S, Jeong HW, Ahn JY, Choi SJ, Lee H, et al. Immunophenotyping of COVID-19 and influenza highlights the role of type I interferons in development of severe COVID-19. Sci Immunol 2020 5:e. abd1554.ArticlePubMedPMC
- 20. Ridker PM. High-sensitivity C-reactive protein: potential adjunct for global risk assessment in the primary prevention of cardiovascular disease. Circulation 2001;103:1813-8.ArticlePubMed
- 21. An YB. Association between serum adiponectin concentration and the onset of metabolic syndrome (Korean Endocrine Society 19: 492–500, 2004, Park SS and 12 others). Endocrinol Metab 2005;20:103-4.
- 22. Alamdari NM, Rahimi FS, Afaghi S, Zarghi A, Qaderi S, Tarki FE, et al. The impact of metabolic syndrome on morbidity and mortality among intensive care unit admitted COVID-19 patients. Diabetes Metab Syndr 2020;14:1979-86.ArticlePubMedPMC
- 23. Janssen I, Heymsfield SB, Allison DB, Kotler DP, Ross R. Body mass index and waist circumference independently contribute to the prediction of nonabdominal, abdominal subcutaneous, and visceral fat. Am J Clin Nutr 2002;75:683-8.ArticlePubMed
- 24. Klein S, Allison DB, Heymsfield SB, Kelley DE, Leibel RL, Nonas C, et al. Waist circumference and cardiometabolic risk: a consensus statement from Shaping America’s Health. Association for Weight Management and Obesity Prevention; NAASO, The Obesity Society; the American Society for Nutrition; and the American Diabetes Association. Am J Clin Nutr 2007;85:1197-202.ArticlePubMed
- 25. Peters SAE, MacMahon S, Woodward M. Obesity as a risk factor for COVID-19 mortality in women and men in the UK biobank: comparisons with influenza/pneumonia and coronary heart disease. Diabetes Obes Metab 2021;23:258-62.PubMedPMC
- 26. Zhu Z, Hasegawa K, Ma B, Fujiogi M, Camargo CA Jr, Liang L. Association of obesity and its genetic predisposition with the risk of severe COVID-19: analysis of population-based cohort data. Metabolism 2020;112:154345.ArticlePubMedPMC
- 27. Popkin BM, Du S, Green WD, Beck MA, Algaith T, Herbst CH, et al. Individuals with obesity and COVID-19: a global perspective on the epidemiology and biological relationships. Obes Rev 2020;21:e13128.ArticlePubMedPMCPDF
- 28. Wadman M. Why obesity worsens COVID-19. Science 2020;369:1280-1.ArticlePubMed
- 29. Wang G, Zhang Q, Zhao X, Dong H, Wu C, Wu F, et al. Low high-density lipoprotein level is correlated with the severity of COVID-19 patients: an observational study. Lipids Health Dis 2020;19:204.ArticlePubMedPMCPDF
- 30. Ding X, Zhang J, Liu L, Yuan X, Zang X, Lu F, et al. High-density lipoprotein cholesterol as a factor affecting virus clearance in covid-19 patients. Respir Med 2020;175:106218.ArticlePubMedPMC
- 31. Sun JT, Chen Z, Nie P, Ge H, Shen L, Yang F, et al. Lipid profile features and their associations with disease severity and mortality in patients with COVID-19. Front Cardiovasc Med 2020;7:584987.ArticlePubMedPMC
- 32. Kaji H. High-density lipoproteins and the immune system. J Lipids 2013;2013:684903.ArticlePubMedPMCPDF
- 33. McKechnie JL, Blish CA. The innate immune system: fighting on the front lines or fanning the flames of COVID-19? Cell Host Microbe 2020;27:863-9.ArticlePubMedPMC
REFERENCES
Figure & Data
References
Citations

- Heterogeneity in familial clustering of metabolic syndrome components in the multiethnic GENNID study
Jia Y. Wan, Deborah Goodman, Sukh Makhnoon, Trina M. Norden‐Krichmar, Baolin Wu, Karen L. Edwards
Obesity.2024; 32(1): 176. CrossRef - Associated Factors with Changes of Metabolic Abnormalities among General Population in COVID-19 Pandemic
Eunjoo Kwon, Eun-Hee Nah, Suyoung Kim, Seon Cho, Hyeran Park
Korean Journal of Health Promotion.2023; 23(2): 55. CrossRef - Association between metabolic syndrome and mortality in patients with COVID-19: A nationwide cohort study
Hyo Jin Park, Jin-Hyung Jung, Kyungdo Han, Jean Shin, Yoojeong Lee, Yujin Chang, Kyeyeung Park, Yoon Jeong Cho, Youn Seon Choi, Seon Mee Kim, Ga Eun Nam
Obesity Research & Clinical Practice.2022; 16(6): 484. CrossRef

 KDA
KDA
 PubReader
PubReader ePub Link
ePub Link Cite
Cite