
- Current
- Browse
- Collections
-
For contributors
- For Authors
- Instructions to authors
- Article processing charge
- e-submission
- For Reviewers
- Instructions for reviewers
- How to become a reviewer
- Best reviewers
- For Readers
- Readership
- Subscription
- Permission guidelines
- About
- Editorial policy
Articles
- Page Path
- HOME > Diabetes Metab J > Volume 40(5); 2016 > Article
-
ReviewClinical Care/Education Gemigliptin: An Update of Its Clinical Use in the Management of Type 2 Diabetes Mellitus
-
Sung-Ho Kim1, Jung-Hwa Yoo1, Woo Je Lee2, Cheol-Young Park3

-
Diabetes & Metabolism Journal 2016;40(5):339-353.
DOI: https://doi.org/10.4093/dmj.2016.40.5.339
Published online: September 12, 2016
1LG Life Sciences Ltd., R&D Park, Daejeon, Korea.
2Department of Internal Medicine, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea.
3Division of Endocrinology and Metabolism, Department of Internal Medicine, Kangbuk Samsung Hospital, Sungkyunkwan University School of Medicine, Seoul, Korea.
- Corresponding author: Cheol-Young Park. Division of Endocrinology and Metabolism, Department of Internal Medicine, Kangbuk Samsung Hospital, Sungkyunkwan University School of Medicine, 29 Saemunan-ro, Jongno-gu, Seoul 03181, Korea. cydoctor@chol.com
• Received: June 22, 2016 • Accepted: July 28, 2016
Copyright © 2016 Korean Diabetes Association
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/4.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
ABSTRACT
- Dipeptidyl peptidase-4 (DPP-4) inhibitors are a new class of oral antidiabetic agent for the treatment of type 2 diabetes mellitus. They increase endogenous levels of incretin hormones, which stimulate glucose-dependent insulin secretion, decrease glucagon secretion, and contribute to reducing postprandial hyperglycemia. Although DPP-4 inhibitors have similar benefits, they can be differentiated in terms of their chemical structure, pharmacology, efficacy and safety profiles, and clinical considerations. Gemigliptin (brand name: Zemiglo), developed by LG Life Sciences, is a potent, selective, competitive, and long acting DPP-4 inhibitor. Various studies have shown that gemigliptin is an optimized DPP-4 inhibitor in terms of efficacy, safety, and patient compliance for treatment of type 2 diabetes mellitus. In this review, we summarize the characteristics of gemigliptin and discuss its potential benefits in clinical practice.
- Type 2 diabetes mellitus (T2DM) is a complex and progressive disease requiring continuous medical care, with multifactorial risk reduction strategies including glycemic control [1]. Therefore, many factors such as adverse events (AEs), disease duration, life expectancy, important comorbidities, and established vascular complications should be taken into account to achieve a patient's individualized goal. Current treatment guidelines recommend the use of dipeptidyl peptidase-4 (DPP-4) inhibitors as first-line therapy or second-line therapy after metformin due to their ease of use, low risk of hypoglycemia, weight neutrality, and favorable tolerability [23]. Currently, there are nine approved DPP-4 inhibitors in Korea including gemigliptin. Although they have some benefits in common through the incretin-based mechanism, DPP-4 inhibitors can be differentiated in terms of pharmacology, efficacy and safety profiles, and clinical considerations along with their different chemical structure [4].
- The development program for DPP-4 inhibitors by LG Life Sciences (Seoul, Korea) was initiated in the early 2000s, and gemigliptin was approved by the Ministry of Food and Drug Safety for treatment of T2DM in June 2012 [5]. LG Life Sciences signed a licensing agreement with multinational pharmaceutical companies such as Sanofi (Paris, France) and Stendhal (Mexico City, Mexico) for 104 countries. Currently, gemigliptin has been approved in 11 countries such as India, Columbia, Costa Rica, Panama, and Ecuador, and several clinical studies are in progress in Russia, Mexico, and Thailand.
- Gemigliptin is a potent, selective, competitive, and long-acting DPP-4 inhibitor. Various studies have shown that gemigliptin is an optimized DPP-4 inhibitor in terms of efficacy, safety, and patient compliance for treatment of T2DM. In this article, we will review the recent clinical studies of gemigliptin and compare it to the other DPP-4 inhibitors with similarly designed clinical studies. This review includes the recent updates presented in PubMed, presents data for drug approvals by the U.S. Food and Drug Administration and European Medicines Agency, and summarizes the product characteristics and clinical data presented as oral and poster presentations at the American Diabetes Association, European Association for the Study of Diabetes, and International Diabetes Federation congresses.
INTRODUCTION
- Rational of drug design
- LG Life Sciences aimed to develop a new DPP-4 inhibitor with potent efficacy, high selectivity, and better compliance with once a day (qd) dosing compared to its competitors. At that time, the new compound could be designed based on the analysis of the internally known DPP-4 crystal structure, and the lead compound could be rapidly optimized through structure-based drug design. In particular, the interaction of the active part of the S2 extensive subsite of the DPP-4 enzyme with CF3 groups on the pyrimidine piperidine resulted in an increase of the selectivity and potency of gemigliptin compared with its competitors.
- Chemistry
- Zemiglo (LG Life Sciences, Seoul, Korea) is the brand name of gemigliptin, which is a novel structure with pyrimidino piperidine derivatives. The co-crystal structure of gemigliptin with the DPP-4 enzyme is shown in Fig. 1. Gemigliptin binds to the S1, S2, and S2 extensive subsites of the DPP-4 enzyme. The piperidinone group of gemigliptin binds to the S1 subsite, where the upside F atom on the piperidin ring forms a hydrogen bond with the side chain of Tyr631 and the downside F atom makes a hydrophobic interaction with the side chain of Tyr666 and Tyr662. In addition, the key interaction occurs between the CF3 groups on the pyrimidino piperidine and the S2 extensive subsite of the DPP-4 substrate, which enhances the potency of the drug and increases its selectivity as well.
- In vitro and in vivo pharmacology
- Gemigliptin showed the type of reversible and competitive inhibitor with a Ki value of 7.25±0.67 nM. In a kinetic study of interactions between the DPP-4 enzyme and DPP-4 inhibitors, gemigliptin was characterized by a fast association and a slow dissociation, indicating that gemigliptin is a long-acting DPP-4 inhibitor. In addition, gemigliptin showed at least >23,000-fold selectivity for various proteases and peptidase including DPP-8, DPP-9, and fibroblast activation protein-α (Table 1) [67891011121314]. Gemigliptin sustainably inhibited DPP-4 activity in a dose-dependent manner and exerted a more potent DPP-4 inhibitory effect for 24 hours than that of sitagliptin at the same dose in rats, dogs, and monkeys. In in vivo studies, gemigliptin prevents the degradation of active glucagon-like peptide-1 (GLP-1) by DPP-4 inhibition, which results in improvements of glucose tolerance by increasing insulin secretion and reducing glucagon secretion during oral glucose tolerance test. It also decreased in a dose-dependent manner glycosylated hemoglobin (HbA1c) and ameliorated β-cell damage in high-fat diet/streptozotocin-induced diabetic mice [6]. These results suggest that gemigliptin is a potent, selective, and long-acting DPP-4 inhibitor with strong binding to the DPP-4 enzyme.
- Recent studies showed that DPP-4 inhibitors exerted pleiotropic effects independent of their glucose-lowering properties [1516]. The potential pleiotropic effects of gemigliptin have also been demonstrated in various disease models. Gemigliptin prevented diabetes-induced podocyte apoptosis and reduced albuminuria in db/db mice [17]. It also prevented the thickening of the diabetes-induced glomerular basement membrane and reduced renal fibrosis in streptozotocin-induced type 1 diabetic mice [18]. In a renal fibrosis model, gemigliptin protected the renal injury via several mechanisms related to fibrosis, inflammation, and oxidative damage [19]. In addition, gemigliptin ameliorated the retinal vascular leakage and loss of tight junction protein in db/db mice and reduced retinal neovascularization in oxygen-induced retinopathy (OIR) mice. These effects were associated with attenuated overexpression of plasminogen activator inhibitor-1, monocyte chemoattractant protein-1, and vascular endothelial growth factor in the retinas of diabetic db/db mice and OIR mice [20]. Gemigliptin also exhibited a potent anti-glycation effect and cardiovascular protective effect in vivo and in vitro studies [2122232425]. Further clinical studies will be needed to elucidate the potential role of gemigliptin for micro- and macrovascular complications.
- Pharmacokinetics
- The pharmacokinetics of gemigliptin has been extensively characterized in healthy subjects and in patients with T2DM. After oral administration of a 50 mg dose to healthy subjects, gemigliptin was rapidly absorbed, with the maximum plasma concentrations (Cmax) attained at about 1.8 hours. The Cmax and area under the curve (AUC) values were increased in a dose-proportional manner [7]. Following a single oral dose of gemigliptin 50 mg to healthy subjects, the mean plasma AUC of gemigliptin was 743.1 ng/hr/mL, Cmax was 62.7 ng/mL, and apparent terminal half-life was 17.1 hours [8]. Key pharmacokinetic parameters for gemigliptin are summarized in Table 2. In addition, pharmacokinetic studies indicate that gemigliptin does not accumulate with multiple dosing and can be administered with or without food [726].
- The elimination of drugs from the body involves the process of metabolism and excretion, and the main routes of excretion generally are via urine and feces. If the clearance mechanism excessively relies onto one of the routes of elimination, or the route is impaired, the plasma concentration can be extremely changed. Therefore, pharmaceutical companies are generally looking for a drug candidate to be eliminated by multiple routes. In a mass balance and excretion study of 14C-gemigliptin in rats, the administered radioactivity was recovered equally in urine (41.2%) and feces (43.6%) [27]. In humans, 90.5% of administered radioactivity of 14C-gemigliptin was recovered: 63.4% from the urine and 27.1% from the feces. Unchanged gemigliptin was the most abundant component in urine and feces, representing 61% (39% of dose) and 41% (11% of dose) of radioactivity in urine and feces, respectively [9]. These results indicated that the elimination route of gemigliptin is relatively balanced between metabolism and excretion through both urine and feces, while other marketed DPP-4 inhibitors are highly depended on one or two elimination pathways [10].
- In an open label, non-randomized, parallel design, a single-dose study was designed to assess the pharmacokinetic profiles of gemigliptin on subjects with renal impairment (RI). In addition, the extent of drug elimination by dialysis was examined in the end-stage renal disease (ESRD) group. The area under the curve from zero to time infinity (AUCinf) of the active moiety showed 1.10, 2.10, 1.99, and 1.98 (2.17) times higher values in mild, moderate, and severe test groups, and ESRD groups on dialysis (non-dialysis period), respectively, than values of the matching control groups. In patients with ESRD, less than 2.9% of the obtained gemigliptin was eliminated through dialysis sessions and there was no significant difference in the pharmacokinetic profiles of gemigliptin between dialysis and non-dialysis periods. Therefore, gemigliptin did not require dose adjustment during the dialysis period [28].
- In patients with mild and moderate hepatic insufficiency (according to the Child-Pugh classification), mean AUC of gemigliptin were increased approximately 50% and 80% compared to healthy subjects, respectively. These modest changes, within 2-fold in gemigliptin pharmacokinetics, were not considered to be clinically meaningful. Consequently, gemigliptin has a relatively balanced clearance mechanism, so it is practical for use in the T2DM patient with renal and hepatic impairment without dose adjustment [29].
- In vitro studies indicated that gemigliptin is not an inhibitor of cytochrome P450 (CYP) 1A2, 2A6, 2B6, 2C9, 2C19, 2D6, 2E1, or 3A4, and it is also not an inducer of CYP1A2, 2C8, 2C9, 2C19, or 3A4. Therefore, gemigliptin is unlikely to cause interactions with other drugs that utilize these metabolic pathways. In vitro studies further indicated that gemigliptin did not induce p-glycoprotein (p-gp), while it mildly inhibited p-gp-mediated transport at a high concentration. Therefore, gemigliptin is unlikely to cause significant interactions with other p-gp substrates at therapeutic concentrations.
- In several drug-drug interaction studies, gemigliptin did not meaningfully alter pharmacokinetics of co-medications frequently used to treat T2DM, such as antidiabetic agents (metformin, pioglitazone, and glimepiride), and antihypertensive and lipid-lowering agents (irbesartan and rosuvastatin) [30313233]. Although co-administration of ketoconazole, a potent inhibitors of CYP3A4, resulted in a moderate increase in gemigliptin exposure (1.9-fold as total active moiety of gemigliptin), no dosage adjustment should be required when gemigliptin is co-administered with ketoconazole. In addition, gemigliptin exposure may be reduced when co-administered with rifampicin, a strong CYP3A4 inducer [34].
CHARACTERISTICS OF GEMIGLIPTIN
Special populations: renal and hepatic impairment
Drug interactions
- Effects on glycemic control
- The efficacy and safety of gemigliptin monotherapy were evaluated in two double-blind placebo controlled studies and one double-blind active-controlled study. A phase II study (study identifier: LG-DPCL002) of gemigliptin was conducted in a randomized, double-blind, placebo-controlled, parallel group design with three doses of 50, 100, and 200 mg qd for the purpose of finding a dose responsiveness and an optimal dose in patients with T2DM. The mean changes of HbA1c at week 12 from the baseline were –0.98%, –0.74%, –0.78% (when adjusted with placebo data, –0.92%, –0.68%, and –0.72%) at 50, 100, and 200 mg, respectively [35]. Among the effective doses obtained from the phase II study in patients with T2DM, the 50 mg dose showed a similar efficacy as the 100 and 200 mg doses, within the maximum safety margin. Similar findings were reported from two phase III studies. Patients were randomized to receive gemigliptin, either a 50 mg qd (n=90) or a placebo (n=92) for 24 weeks (study identifier: LG-DPCL005; Clinical-Trials.gov registration number: NCT01601990). The placebo-subtracted changes from baseline in HbA1c were reported to be -0.71% (95% confidence interval [CI], -1.04 to -0.37) with gemigliptin 50 mg (Table 3) [35363738394041424344454647484950515253545556575859606162636465666768697071]. In addition, a 28-week open-label extension study was designed to evaluate the long-term safety and efficacy of gemigliptin. Among 165 patients who consented to participate in the extension period of study LG-DPCL005, 158 patients (96%) completed their treatments for 52 weeks. All patients were switched to or continued their treatments only with gemigliptin 50 mg qd during the extension period. A further decrease in HbA1c was observed in the continued treatment with gemigliptin 50 mg in this extension period, and the mean change from baseline at 52 weeks (–0.87%) was still clinically and statistically significant (full analysis set analysis, P<0.0001) [72].
- In another double-blind, active-controlled, phase III trial (study identifier: LG-DPCL011), eligible patients with HbA1c greater than 7.5% were randomized to receive gemigliptin 50 mg qd with metformin slow release (SR) qd (n=141), gemigliptin 50 mg qd (n=142), or metformin SR qd (n=150) for 24 weeks. After 24 weeks, the reduction from the baseline in HbA1c was –1.24% for gemigliptin monotherapy [37].
- The combination therapy of gemigliptin with metformin showed additional effects by increasing the plasma active GLP-1 concentration and lowering the serum glucose level. The plasma glucagon level was also lower in combination therapy with metformin than in monotherapy [32].
- In this randomized, double-blind, active-controlled, phase III trial (study identifier: LG-DPCL011, INICOM study; ClinicalTrials.gov registration number: NCT01787396), eligible patients with an HbA1c greater than 7.5% were randomized to gemigliptin 50 mg qd+metformin SR qd (n=141), gemigliptin 50 mg qd (n=142), or metformin SR qd (n=150). From weeks 2 to 6, metformin SR was uptitrated incrementally from 500 to 2,000 mg/day maximum in the gemigliptin/metformin and metformin groups. The mean daily doses of metformin at week 24 were 1,699 and 1,868 mg for the gemigliptin/metformin group and the metformin group, respectively. Mean change in HbA1c from baseline was –2.06% for gemigliptin/metformin group versus –1.24% for the gemigliptin group and –1.47% for the metformin group, respectively (P<0.0001 for all comparisons of combination therapy vs. monotherapy) [37]. The differences in proportions achieving an HbA1c <7% or <6.5% were also statistically significant (P<0.0001) between the combination therapy and the respective monotherapy groups.
- A 24-week, multinational, randomized, double-blind, active-controlled study (study identifier: LG-DPCL006; ClinicalTrials.gov registration number: NCT01602003) was designed to assess the efficacy and safety of gemigliptin 50 mg compared to the active control (sitagliptin) added to ongoing metformin therapy in patients with T2DM inadequately controlled with metformin alone (HbA1c, 7% to 11%) [73]. After 24 weeks, the reduction from baseline for HbA1c was 0.81% for gemigliptin 25 mg twice a day (bid) and 0.77% for gemigliptin 50 mg qd, and the differences in the least square mean changes from baseline between groups (each group of gemigliptin-sitagliptin group) were -0.011% in gemigliptin 25 mg bid and 0.004% in gemigliptin 50 mg qd. The proportion of patients achieving an HbA1c <7% at week 24 (gemigliptin 25 mg bid group, 50%; gemigliptin 50 mg qd group, 54.07%) was comparable to the results with sitagliptin 100 mg qd (48.87%). The efficacy of lowering HbA1c in the gemigliptin group was generally consistent across the subgroups based on age (<65 or ≥65 years), gender, duration of T2DM (5, >5 to 10, or >10 years), and baseline body mass index (BMI, <25 or ≥25 kg/m2). In addition, gemigliptin groups led to a significantly greater inhibition of plasma DPP-4 compared to sitagliptin [38].
- This study was extended by 28 weeks in order to evaluate the long-term efficacy and safety of gemigliptin. About 90% of the patients who completed 24 weeks of treatment in each group consented to continue the study to receive further treatment with gemigliptin 50 mg qd for 28 weeks. The subjects who had been treated with the active control drug (sitagliptin) were also treated with gemigliptin for the long-term extension periods. All treatment groups showed clinically and statistically (P<0.0001) significant improvement in glycemic control from baseline after 52 weeks. The reduction from the baseline in HbA1c was –1.06 (95% CI, –1.28 to –0.85) in the patients who continued to receive gemigliptin 50 mg qd. When sitagliptin was switched to gemigliptin at week 24, there was additional 0.1% reduction from the baseline in HbA1c at week 52 and significantly more patients receiving gemigliptin (27.3%) than sitagliptin (6.8%) achieved an HbA1c <6.5% [74]. These results show that the switching from sitagliptin to gemigliptin provided the sustained glycemic improvements in patients with T2DM.
- In this multicenter, randomized, double-blind, phase III study (study identifier: LG-DPCL010, TROICA study; ClinicalTrials.gov registration number: NCT01990469), eligible patients with inadequate glycemic control (7%≤HbA1c≤11%) were randomized to gemigliptin 50 mg qd (n=109) or placebo (n=110). The baseline demographics were similar between groups (age, 60.9 years; BMI, 24.9 kg/m2; duration of T2DM, 12.9 years), with mean±standard deviation (SD) baseline HbA1c of 8.12%±0.82% in the gemigliptin group and 8.15%±0.89% in the placebo group. At week 24, the adjusted mean±standard error change for HbA1c with gemigliptin was –0.88%±0.17% (change with placebo –0.01%±0.18%; difference –0.87%±0.12%; 95% CI, –1.09 to –0.64; P<0.0001) [39].
- RI in T2DM limits the usable medications for lowering glucose level and requires frequent monitoring of renal function. Gemigliptin has balanced elimination between urinary/fecal excretion and hepatic metabolism; therefore, it does not require dose adjustment in patient with moderate to severe RI. This study evaluated the efficacy and safety of gemigliptin in T2DM patients with moderate to severe RI. This randomized, double-blind, parallel group, phase IIIb study (study identifier: LG-DPCL015, GUARD study; ClinicalTrials.gov registration number: NCT01968044) was composed of a 12-week, placebo controlled period, followed by a 40-week, double-blind active controlled extension period (placebo switched to linagliptin). A total of 132 patients with moderate or severe RI were randomized to receive gemigliptin (n=66) or placebo (n=66). Insulin was used as predominant background therapy (63.1%). At week 12, the placebo-adjusted mean change in HbA1c from the baseline was –1.20% (95% CI, –1.53 to –0.87; P<0.0001) [40]. A similar profile was also observed in other glycemic control parameters (fasting plasma glucose, glycated albumin, and fructosamine).
- The efficacy and safety of gemigliptin was directly compared with other DPP-4 inhibitors in the three multicenter, randomized, active-controlled studies. Two head-to-head studies between gemigliptin and sitagliptin were conducted in patients with T2DM as an add-on to metformin and initial combination therapy with metformin. As mentioned above, the phase III study showed non-inferiority in HbA1c reduction for gemigliptin compared with sitagliptin in the add-on to metformin therapy [73]. The second study was performed to assess the efficacy of gemigliptin in combination with metformin for the initial treatment of drug-naïve patients with an HbA1c greater than 7.5% (study identifier: LG-DPCL012). After 12 weeks, the mean HbA1c was reduced from baseline by 2.75%, 2.24%, and 2.75% for gemigliptin 50, sitagliptin 100, and glimepiride 2 mg qd, respectively. However, there was no significant difference in HbA1c reduction between groups [75].
- The other head-to-head study (study identifier: LG-DPCL015, GUARD study; ClinicalTrials.gov registration number: NCT01968044) that compared gemigliptin and linagliptin was performed in the T2DM patients with RI. As mentioned above, the placebo group was switched to linagliptin 5 mg qd during the 40-week extension period. After 52 weeks, the adjusted mean±standard error change from baseline in HbA1c was –1.00%±0.21% and –0.65%±0.22% in the gemigliptin and placebo/linagliptin groups, respectively [76]. During the 40-week extension, AEs were reported in 68.0% and 73.1% of subjects using gemigliptin and linagliptin, respectively. The incidence of hypoglycemia was similar between treatment groups (gemigliptin, 20.0%; linagliptin, 28.8%). There were no meaningful changes from the baseline in the body weight (gemigliptin, 0.28 kg; linagliptin 0.33 kg).
- Effects on glycemic variability
- Glycemic variability and chronic sustained hyperglycemia are the main components of dysglycemia in diabetes. The previous studies suggested that different pharmacodynamic profiles between DPP-4 inhibitors have been associated with the different effects on glycemic variability [7778]. In this study, a multicenter, randomized, active-controlled, parallel group, open-label, exploratory study was designed to evaluate the efficacy on glycemic variability and safety of initial combination therapy of gemigliptin 50 mg qd versus sitagliptin 100 mg qd, or glimepiride 2 mg qd with metformin in patients with T2DM (study identifier: LG-DPCL012, STABLE study; ClinicalTrials.gov registration number: NCT01890629). The mean amplitude of glycemic excursions (MAGE) and SD of glucose were used for assessing glucose fluctuations from the baseline after 12 weeks of treatment. At 12 weeks, MAGE was significantly lower in the DPP-4 inhibitor groups (gemigliptin and sitagliptin) than in the glimepiride group (–43.1, –38.3, and –21.7 mg/dL, respectively). Furthermore, the SD of mean glucose was significantly lower in patients with gemigliptin when compared with sitagliptin (P=0.023) and glimepiride (P=0.0058) [75].
- Beyond glucose control
- In addition to improvements in glycemic control, gemigliptin as mono- and combination therapy slightly decreased mean total cholesterol, low density lipoprotein cholesterol, and triglycerides. The result was summarized in Table 4.
- The GUARD study also evaluated whether gemigliptin had renoprotective effects in type 2 diabetic patients with moderate to severe RI. The urinary albumin creatinine ratio mean changes from baseline were –339 mg/g Cr with gemigliptin compared with 172 mg/g Cr with the placebo (P<0.0001) after 12 weeks. This reduction in albuminuria of the gemigliptin group was observed regardless of glucose lowering effect, blood pressure change, and the use of a renin-angiotensin-aldosterone system blocking agent. Furthermore, urinary nephrin in the gemigliptin group were also significantly reduced compared to the placebo group [76].
- Ongoing studies
- Several clinical studies in LG Life Sciences are actively underway to assess the efficacy and safety as an add-on combination therapy with insulin (with or without metformin) (ClinicalTrials.gov registration number: NCT02831361), to evaluate the efficacy and safety of gemigliptin-rosuvastatin fixed-dose combination in patients with T2DM and dyslipidemia in phase III clinical trials (ClinicalTrials.gov registration number: NCT02126358), and to evaluate the efficacy and safety of gemigliptin compared with vildagliptin in Russian patients with T2DM (ClinicalTrials.gov registration number: NCT02343926).
- Safety and tolerability of gemigliptin
- A pooled safety analysis was performed with subjects who had received the investigational product at least once in studies LG-DPCL002, LG-DPCL005, LG-DPCL006, LG-DPCL010, LG-DPCL011, and LG-DPCL012. In the safety analysis, 1,080 patients received an oral dose of gemigliptin 50 mg in the comparative clinical studies, and 446 patients received other oral antidiabetic agents (OADs). The most commonly reported AEs were upper respiratory tract infection, urinary tract infection, nasopharyngitis, headache, diarrhea, arthralgia, hypertension, and cough. Nasopharyngitis was the only AE reported in more than 5.0% of subjects, and its incidence rate was similar between the two groups, with 6.7% and 6.3% in gemigliptin and other OADs groups, respectively. Table 5 shows the frequency of AEs and discontinuation of treatment due to AEs from clinical trials of gemigliptin. The incidence of hypoglycemia was similar to placebo or active control groups when gemigliptin was administered as monotherapy or combination therapy. Most events were mild to moderate in nature and did not require an additional treatment or discontinuation. In addition, treatment with gemigliptin does not increase body weight (Table 6).
- Overall, gemigliptin was generally well tolerated in controlled clinical studies in both monotherapy and combination therapy. The incidences of AEs were generally similar to incidences in the placebo and active control groups.
CLINICAL EFFICACY OF GEMIGLIPTIN
Monotherapy
Initial combination therapy with metformin
Add-on to metformin
Add-on to metformin and glimepiride
Add-on therapy in patients with renal impairment
Head-to-head comparisons between gemigliptin and other DPP-4 inhibitors
Effect on lipid profiles
Effect on albuminuria
- Based on the findings from the studies in this review, Table 7 summarizes key points contained in the article. Gemigliptin is a potent, selective, and long-acting DPP-4 inhibitor and has been shown to be effective and well tolerated as monotherapy and combination therapy in patients with T2DM. In addition, gemigliptin is more effective in reducing glycemic variability than glimepiride and sitagliptin, and lowers albuminuria independent of its glucose lowering effect. Therefore, gemigliptin may provide important benefits for special populations, especially patients with renal insufficiency and/or the elderly. The results from ongoing clinical studies will also provide additional evidence of the clinical benefits of gemigliptin in the management of T2DM.
CONCLUSIONS
- 1. American Diabetes Association. Standards of medical care in diabetes: 2014. Diabetes Care 2014;37(Suppl 1):S14-S80. ArticlePubMedPDF
- 2. Garber AJ, Abrahamson MJ, Barzilay JI, Blonde L, Bloomgarden ZT, Bush MA, Dagogo-Jack S, DeFronzo RA, Einhorn D, Fonseca VA, Garber JR, Garvey WT, Grunberger G, Handelsman Y, Henry RR, Hirsch IB, Jellinger PS, McGill JB, Mechanick JI, Rosenblit PD, Umpierrez GE. Consensus statement by the American Association of Clinical Endocrinologists and American College of Endocrinology on the comprehensive type 2 diabetes management algorithm: 2016 executive summary. Endocr Pract 2016;22:84-113. ArticlePubMed
- 3. Inzucchi SE, Bergenstal RM, Buse JB, Diamant M, Ferrannini E, Nauck M, Peters AL, Tsapas A, Wender R, Matthews DR. American Diabetes Association (ADA). European Association for the Study of Diabetes (EASD). Management of hyperglycemia in type 2 diabetes: a patient-centered approach. Position statement of the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD). Diabetes Care 2012;35:1364-1379. ArticlePubMedPMCPDF
- 4. Deacon CF. Dipeptidyl peptidase-4 inhibitors in the treatment of type 2 diabetes: a comparative review. Diabetes Obes Metab 2011;13:7-18. ArticlePubMed
- 5. Kim SH, Lee SH, Yim HJ. Gemigliptin, a novel dipeptidyl peptidase 4 inhibitor: first new anti-diabetic drug in the history of Korean pharmaceutical industry. Arch Pharm Res 2013;36:1185-1188. ArticlePubMedPDF
- 6. Kim SH, Jung E, Yoon MK, Kwon OH, Hwang DM, Kim DW, Kim J, Lee SM, Yim HJ. Pharmacological profiles of gemigliptin (LC15-0444), a novel dipeptidyl peptidase-4 inhibitor, in vitro and in vivo. Eur J Pharmacol 2016;788:54-64. ArticlePubMed
- 7. Lim KS, Cho JY, Kim BH, Kim JR, Kim HS, Kim DK, Kim SH, Yim HJ, Lee SH, Shin SG, Jang IJ, Yu KS. Pharmacokinetics and pharmacodynamics of LC15-0444, a novel dipeptidyl peptidase IV inhibitor, after multiple dosing in healthy volunteers. Br J Clin Pharmacol 2009;68:883-890. ArticlePubMedPMC
- 8. Lim KS, Kim JR, Choi YJ, Shin KH, Kim KP, Hong JH, Cho JY, Shin HS, Yu KS, Shin SG, Kwon OH, Hwang DM, Kim JA, Jang IJ. Pharmacokinetics, pharmacodynamics, and tolerability of the dipeptidyl peptidase IV inhibitor LC15-0444 in healthy Korean men: a dose-block-randomized, double-blind, placebo-controlled, ascending single-dose, phase I study. Clin Ther 2008;30:1817-1830. ArticlePubMed
- 9. Kim N, Patrick L, Mair S, Stevens L, Ford G, Birks V, Lee SH. Absorption, metabolism and excretion of [14C]gemigliptin, a novel dipeptidyl peptidase 4 inhibitor, in humans. Xenobiotica 2014;44:522-530. ArticlePubMed
- 10. Filippatos TD, Athyros VG, Elisaf MS. The pharmacokinetic considerations and adverse effects of DPP-4 inhibitors [corrected]. Expert Opin Drug Metab Toxicol 2014;10:787-812. PubMed
- 11. Deacon CF, Lebovitz HE. Comparative review of dipeptidyl peptidase-4 inhibitors and sulphonylureas. Diabetes Obes Metab 2016;18:333-347. ArticlePubMedPDF
- 12. Baetta R, Corsini A. Pharmacology of dipeptidyl peptidase-4 inhibitors: similarities and differences. Drugs 2011;71:1441-1467. ArticlePubMed
- 13. Nabeno M, Akahoshi F, Kishida H, Miyaguchi I, Tanaka Y, Ishii S, Kadowaki T. A comparative study of the binding modes of recently launched dipeptidyl peptidase IV inhibitors in the active site. Biochem Biophys Res Commun 2013;434:191-196. ArticlePubMed
- 14. Morishita R, Nakagami H. Teneligliptin: expectations for its pleiotropic action. Expert Opin Pharmacother 2015;16:417-426. ArticlePubMed
- 15. Omar B, Ahren B. Pleiotropic mechanisms for the glucose-lowering action of DPP-4 inhibitors. Diabetes 2014;63:2196-2202. ArticlePubMedPDF
- 16. Aroor AR, Sowers JR, Jia G, DeMarco VG. Pleiotropic effects of the dipeptidylpeptidase-4 inhibitors on the cardiovascular system. Am J Physiol Heart Circ Physiol 2014;307:H477-H492. ArticlePubMedPMC
- 17. Jung E, Kim J, Ho Kim S, Kim S, Cho MH. Gemigliptin improves renal function and attenuates podocyte injury in mice with diabetic nephropathy. Eur J Pharmacol 2015;761:116-124. ArticlePubMed
- 18. Jung GS, Jeon JH, Choe MS, Kim SW, Lee IK, Kim MK, Park KG. Renoprotective effect of gemigliptin, a dipeptidyl peptidase-4 inhibitor, in streptozotocin-induced type 1 diabetic mice. Diabetes Metab J 2016;40:211-221. ArticlePubMedPMCPDF
- 19. Min HS, Kim JE, Lee MH, Song HK, Kang YS, Lee MJ, Lee JE, Kim HW, Cha JJ, Chung YY, Hyun YY, Han JY, Cha DR. Dipeptidyl peptidase IV inhibitor protects against renal interstitial fibrosis in a mouse model of ureteral obstruction. Lab Invest 2014;94:598-607. ArticlePubMedPDF
- 20. Jung E, Kim J, Kim CS, Kim SH, Cho MH. Gemigliptin, a dipeptidyl peptidase-4 inhibitor, inhibits retinal pericyte injury in db/db mice and retinal neovascularization in mice with ischemic retinopathy. Biochim Biophys Acta 2015;1852:2618-2629. ArticlePubMed
- 21. Jung E, Kim J, Kim SH, Kim S, Cho MH. Gemigliptin, a novel dipeptidyl peptidase-4 inhibitor, exhibits potent anti-glycation properties in vitro and in vivo. Eur J Pharmacol 2014;744:98-102. ArticlePubMed
- 22. Moon JY, Woo JS, Seo JW, Lee A, Kim DJ, Kim YG, Kim SY, Lee KH, Lim SJ, Cheng XW, Lee SH, Kim W. The dose-dependent organ-specific effects of a dipeptidyl peptidase-4 inhibitor on cardiovascular complications in a model of type 2 diabetes. PLoS One 2016;11:e0150745ArticlePubMedPMC
- 23. Choi SH, Park S, Oh CJ, Leem J, Park KG, Lee IK. Dipeptidyl peptidase-4 inhibition by gemigliptin prevents abnormal vascular remodeling via NF-E2-related factor 2 activation. Vascul Pharmacol 2015;73:11-19. ArticlePubMed
- 24. Hwang HJ, Chung HS, Jung TW, Ryu JY, Hong HC, Seo JA, Kim SG, Kim NH, Choi KM, Choi DS, Baik SH, Yoo HJ. The dipeptidyl peptidase-IV inhibitor inhibits the expression of vascular adhesion molecules and inflammatory cytokines in HUVECs via Akt- and AMPK-dependent mechanisms. Mol Cell Endocrinol 2015;405:25-34. ArticlePubMed
- 25. Hwang HJ, Jung TW, Ryu JY, Hong HC, Choi HY, Seo JA, Kim SG, Kim NH, Choi KM, Choi DS, Baik SH, Yoo HJ. Dipeptidyl petidase-IV inhibitor (gemigliptin) inhibits tunicamycin-induced endoplasmic reticulum stress, apoptosis and inflammation in H9c2 cardiomyocytes. Mol Cell Endocrinol 2014;392:1-7. ArticlePubMed
- 26. Choi HY, Noh YH, Kim YH, Kim MJ, Lee SH, Kim JA, Kim B, Lim HS, Bae KS. Effects of food on the pharmacokinetics of gemigliptin/metformin sustained-release 50/1,000 mg (25/500 mg x 2 tablets) fixeddose combination tablet in healthy male volunteers. Int J Clin Pharmacol Ther 2014;52:381-391. PubMed
- 27. Kim Y, Kim U, Kim IS, Lee SH, Lee J, Kim DH, Yoo HH. Absorption, distribution, metabolism and excretion of gemigliptin, a novel dipeptidyl peptidase IV inhibitor, in rats. Xenobiotica 2014;44:627-634. ArticlePubMed
- 28. Shon JH, Kim N, Park SJ, Oh MK, Kim EY, Lee SH, Kim YH, Shin JG. Effect of renal impairment and haemodialysis on the pharmacokinetics of gemigliptin (LC15-0444). Diabetes Obes Metab 2014;16:1028-1031. ArticlePubMed
- 29. Zemiglo: Ministry of Food and Drug Safety (MFDS) Package insert cited 2016 Aug 3. Available from: https://ezdrug.mfds.go.kr.
- 30. Choi HY, Lim HS, Kim YH, Jeon HS, Kim MJ, Lee SH, Jung JH, Lee YK, Kim HJ, Bae KS. Evaluation of the pharmacokinetics of the DPP-4 inhibitor gemigliptin when coadministered with rosuvastatin or irbesartan to healthy subjects. Curr Med Res Opin 2015;31:229-241. ArticlePubMed
- 31. Choi HY, Kim YH, Kim MJ, Lee SH, Bang K, Han S, Lim HS, Bae KS. Evaluation of pharmacokinetic drug interactions between gemigliptin (dipeptidylpeptidase-4 inhibitor) and glimepiride (sulfonylurea) in healthy volunteers. Drugs R D 2014;14:165-176. ArticlePubMedPMCPDF
- 32. Shin D, Cho YM, Lee S, Lim KS, Kim JA, Ahn JY, Cho JY, Lee H, Jang IJ, Yu KS. Pharmacokinetic and pharmacodynamic interaction between gemigliptin and metformin in healthy subjects. Clin Drug Investig 2014;34:383-393.ArticlePubMedPDF
- 33. Kim SE, Yi S, Shin KH, Kim TE, Kim MJ, Kim YH, Yoon SH, Cho JY, Shin SG, Jang IJ, Yu KS. Evaluation of the pharmacokinetic interaction between the dipeptidyl peptidase IV inhibitor LC15-0444 and pioglitazone in healthy volunteers. Int J Clin Pharmacol Ther 2012;50:17-23. ArticlePubMed
- 34. Noh YH, Lim HS, Jin SJ, Kim MJ, Kim YH, Sung HR, Choi HY, Bae KS. Effects of ketoconazole and rifampicin on the pharmacokinetics of gemigliptin, a dipeptidyl peptidase-IV inhibitor: a crossover drug-drug interaction study in healthy male Korean volunteers. Clin Ther 2012;34:1182-1194. ArticlePubMed
- 35. Rhee EJ, Lee WY, Yoon KH, Yoo SJ, Lee IK, Baik SH, Kim YK, Lee MK, Park KS, Park JY, Cha BS, Lee HW, Min KW, Bae HY, Kim MJ, Kim JA, Kim DK, Kim SW. A multicenter, randomized, placebo-controlled, double-blind phase II trial evaluating the optimal dose, efficacy and safety of LC 15-0444 in patients with type 2 diabetes. Diabetes Obes Metab 2010;12:1113-1119. ArticlePubMed
- 36. Yang SJ, Min KW, Gupta SK, Park JY, Shivane VK, Pitale SU, Agarwal PK, Sosale A, Gandhi P, Dharmalingam M, Mohan V, Mahesh U, Kim DM, Kim YS, Kim JA, Kim PK, Baik SH. A multicentre, multinational, randomized, placebo-controlled, double-blind, phase 3 trial to evaluate the efficacy and safety of gemigliptin (LC15-0444) in patients with type 2 diabetes. Diabetes Obes Metab 2013;15:410-416. ArticlePubMed
- 37. Lim S, Min K, Yu JM, Chamnan P, Kim ES, Yoon KH, Kwon S, Moon MK, Lee KW, Kim DJ, Kim M, Wongtanate M, Kim EY, Kim SH, Lee MK. No. 109: Efficacy and safety of gemigliptin/metformin initial combination therapy versus either as monotherapy in drug-naïve patients with type 2 diabetes In: Oral presentation of 51st Annual Meeting of European Association for the Study of Diabetes; 2015 Sep 14-18; Stockholm, SZ.
- 38. Rhee EJ, Min KW, Jang HC, Nam-Goong IS, Chung CH, Park JY, Bae HY, Kim DM, Baik SH, Lee MK, Kim BJ, Chang SA, Ahn CW, Kim YS, Yoon KH, Park KS, Kim HJ, Shivane VK, Sosale AR, Dharmalingam M, Gandhi P, Gupta SK, Pitale SU, Agarwal PK, Rais N, Mohan V, Mahesh U, Kim JA, Kim PK, Kim SW. No. 1128: Efficacy and safety of gemigliptin compared with sitagliptin added to ongoing metformin therapy in patients with type 2 diabetes inadequately controlled with metformin alone In: Poster presentation of American Diabetes Association 72nd Scientific sessions; 2012 Jun 8-12; Philadelphia, PA.
- 39. Kim SH, Yu J, Jang H, Song Y, Ahn K, Oh T, Lee H, Lee D, Kim J, Park TS, Jeong CH, Kim BJ, Han K, Park K. No. 1169: Efficacy and safety of gemigliptin as add-on therapy in patients with type 2 diabetes inadequately controlled on metformin and glimepiride In: Poster presentation of American Diabetes Association 76th Scientific sessions; 2016 Jun 10-14; New Orleans, LA.
- 40. Yoon S, Han B, Kim S, Han S, Jo YI, Jeong K, Oh KH, Park H, Park SH, Kang SW, Na KR, Jang Y, Kim SH, Cha D. GUARD Study. No. 804: Efficacy and safety of gemigliptin in type 2 diabetes patients with moderate to severe renal impairment In: Poster presentation of 51st Annual Meeting of European Association for the Study of Diabetes; 2015 Sep 14-18; Stockholm, SZ.
- 41. Raz I, Hanefeld M, Xu L, Caria C, Williams-Herman D, Khatami H. Sitagliptin Study 023 Group. Efficacy and safety of the dipeptidyl peptidase-4 inhibitor sitagliptin as monotherapy in patients with type 2 diabetes mellitus. Diabetologia 2006;49:2564-2571. ArticlePubMedPDF
- 42. Dejager S, Razac S, Foley JE, Schweizer A. Vildagliptin in drug-naïve patients with type 2 diabetes: a 24-week, double-blind, randomized, placebo-controlled, multiple-dose study. Horm Metab Res 2007;39:218-223. ArticlePubMed
- 43. Rosenstock J, Sankoh S, List JF. Glucose-lowering activity of the dipeptidyl peptidase-4 inhibitor saxagliptin in drug-naive patients with type 2 diabetes. Diabetes Obes Metab 2008;10:376-386. ArticlePubMed
- 44. Barnett AH, Patel S, Harper R, Toorawa R, Thiemann S, von Eynatten M, Woerle HJ. Linagliptin monotherapy in type 2 diabetes patients for whom metformin is inappropriate: an 18-week randomized, double-blind, placebo-controlled phase III trial with a 34-week active-controlled extension. Diabetes Obes Metab 2012;14:1145-1154. ArticlePubMed
- 45. Kadowaki T, Kondo K. Efficacy, safety and dose-response relationship of teneligliptin, a dipeptidyl peptidase-4 inhibitor, in Japanese patients with type 2 diabetes mellitus. Diabetes Obes Metab 2013;15:810-818. ArticlePubMed
- 46. Aschner P, Kipnes MS, Lunceford JK, Sanchez M, Mickel C, Williams-Herman DE. Sitagliptin Study 021 Group. Effect of the dipeptidyl peptidase-4 inhibitor sitagliptin as monotherapy on glycemic control in patients with type 2 diabetes. Diabetes Care 2006;29:2632-2637. ArticlePubMedPDF
- 47. Pi-Sunyer FX, Schweizer A, Mills D, Dejager S. Efficacy and tolerability of vildagliptin monotherapy in drug-naïve patients with type 2 diabetes. Diabetes Res Clin Pract 2007;76:132-138. ArticlePubMed
- 48. Rosenstock J, Aguilar-Salinas C, Klein E, Nepal S, List J, Chen R. CV181-011 Study Investigators. Effect of saxagliptin monotherapy in treatment-naïve patients with type 2 diabetes. Curr Med Res Opin 2009;25:2401-2411. ArticlePubMed
- 49. Del Prato S, Barnett AH, Huisman H, Neubacher D, Woerle HJ, Dugi KA. Effect of linagliptin monotherapy on glycaemic control and markers of β-cell function in patients with inadequately controlled type 2 diabetes: a randomized controlled trial. Diabetes Obes Metab 2011;13:258-267. ArticlePubMed
- 50. DeFronzo RA, Fleck PR, Wilson CA, Mekki Q. Alogliptin Study 010 Group. Efficacy and safety of the dipeptidyl peptidase-4 inhibitor alogliptin in patients with type 2 diabetes and inadequate glycemic control: a randomized, double-blind, placebo-controlled study. Diabetes Care 2008;31:2315-2317. PubMedPMC
- 51. Hong S, Park CY, Han KA, Chung CH, Ku BJ, Jang HC, Ahn CW, Lee MK, Moon MK, Son HS, Lee CB, Cho YW, Park SW. Efficacy and safety of teneligliptin, a novel dipeptidyl peptidase-4 inhibitor, in Korean patients with type 2 diabetes mellitus: a 24-week multicentre, randomized, double-blind, placebo-controlled phase III trial. Diabetes Obes Metab 2016;18:528-532. PubMedPMC
- 52. Goldstein BJ, Feinglos MN, Lunceford JK, Johnson J, Williams-Herman DE. Sitagliptin 036 Study Group. Effect of initial combination therapy with sitagliptin, a dipeptidyl peptidase-4 inhibitor, and metformin on glycemic control in patients with type 2 diabetes. Diabetes Care 2007;30:1979-1987. ArticlePubMedPDF
- 53. Bosi E, Dotta F, Jia Y, Goodman M. Vildagliptin plus metformin combination therapy provides superior glycaemic control to individual monotherapy in treatment-naive patients with type 2 diabetes mellitus. Diabetes Obes Metab 2009;11:506-515. ArticlePubMed
- 54. Jadzinsky M, Pfutzner A, Paz-Pacheco E, Xu Z, Allen E, Chen R. CV181-039 Investigators. Saxagliptin given in combination with metformin as initial therapy improves glycaemic control in patients with type 2 diabetes compared with either monotherapy: a randomized controlled trial. Diabetes Obes Metab 2009;11:611-622. ArticlePubMed
- 55. Haak T, Meinicke T, Jones R, Weber S, von Eynatten M, Woerle HJ. Initial combination of linagliptin and metformin improves glycaemic control in type 2 diabetes: a randomized, double-blind, placebo-controlled study. Diabetes Obes Metab 2012;14:565-574. ArticlePubMed
- 56. Pratley RE, Fleck P, Wilson C. Efficacy and safety of initial combination therapy with alogliptin plus metformin versus either as monotherapy in drug-naïve patients with type 2 diabetes: a randomized, double-blind, 6-month study. Diabetes Obes Metab 2014;16:613-621. ArticlePubMed
- 57. Charbonnel B, Karasik A, Liu J, Wu M, Meininger G. Sitagliptin Study 020 Group. Efficacy and safety of the dipeptidyl peptidase-4 inhibitor sitagliptin added to ongoing metformin therapy in patients with type 2 diabetes inadequately controlled with metformin alone. Diabetes Care 2006;29:2638-2643. ArticlePubMedPDF
- 58. Bosi E, Camisasca RP, Collober C, Rochotte E, Garber AJ. Effects of vildagliptin on glucose control over 24 weeks in patients with type 2 diabetes inadequately controlled with metformin. Diabetes Care 2007;30:890-895. ArticlePubMedPDF
- 59. DeFronzo RA, Hissa MN, Garber AJ, Luiz Gross J, Yuyan Duan R, Ravichandran S, Chen RS. Saxagliptin 014 Study Group. The efficacy and safety of saxagliptin when added to metformin therapy in patients with inadequately controlled type 2 diabetes with metformin alone. Diabetes Care 2009;32:1649-1655. ArticlePubMedPMCPDF
- 60. Taskinen MR, Rosenstock J, Tamminen I, Kubiak R, Patel S, Dugi KA, Woerle HJ. Safety and efficacy of linagliptin as add-on therapy to metformin in patients with type 2 diabetes: a randomized, double-blind, placebo-controlled study. Diabetes Obes Metab 2011;13:65-74. ArticlePubMed
- 61. Nauck MA, Ellis GC, Fleck PR, Wilson CA, Mekki Q. Alogliptin Study 008 Group. Efficacy and safety of adding the dipeptidyl peptidase-4 inhibitor alogliptin to metformin therapy in patients with type 2 diabetes inadequately controlled with metformin monotherapy: a multicentre, randomised, double-blind, placebo-controlled study. Int J Clin Pract 2009;63:46-55. ArticlePubMed
- 62. Kim MK, Rhee EJ, Han KA, Woo AC, Lee MK, Ku BJ, Chung CH, Kim KA, Lee HW, Park IB, Park JY, Chul Jang HC, Park KS, Jang WI, Cha BY. Efficacy and safety of teneligliptin, a dipeptidyl peptidase-4 inhibitor, combined with metformin in Korean patients with type 2 diabetes mellitus: a 16-week, randomized, double-blind, placebo-controlled phase III trial. Diabetes Obes Metab 2015;17:309-312. ArticlePubMedPMCPDF
- 63. Hermansen K, Kipnes M, Luo E, Fanurik D, Khatami H, Stein P. Sitagliptin Study 035 Group. Efficacy and safety of the dipeptidyl peptidase-4 inhibitor, sitagliptin, in patients with type 2 diabetes mellitus inadequately controlled on glimepiride alone or on glimepiride and metformin. Diabetes Obes Metab 2007;9:733-745. ArticlePubMed
- 64. Lukashevich V, Del Prato S, Araga M, Kothny W. Efficacy and safety of vildagliptin in patients with type 2 diabetes mellitus inadequately controlled with dual combination of metformin and sulphonylurea. Diabetes Obes Metab 2014;16:403-409. ArticlePubMedPDF
- 65. Moses RG, Kalra S, Brook D, Sockler J, Monyak J, Visvanathan J, Montanaro M, Fisher SA. A randomized controlled trial of the efficacy and safety of saxagliptin as add-on therapy in patients with type 2 diabetes and inadequate glycaemic control on metformin plus a sulphonylurea. Diabetes Obes Metab 2014;16:443-450. ArticlePubMed
- 66. Owens DR, Swallow R, Dugi KA, Woerle HJ. Efficacy and safety of linagliptin in persons with type 2 diabetes inadequately controlled by a combination of metformin and sulphonylurea: a 24-week randomized study. Diabet Med 2011;28:1352-1361. PubMed
- 67. Vilsboll T, Rosenstock J, Yki-Jarvinen H, Cefalu WT, Chen Y, Luo E, Musser B, Andryuk PJ, Ling Y, Kaufman KD, Amatruda JM, Engel SS, Katz L. Efficacy and safety of sitagliptin when added to insulin therapy in patients with type 2 diabetes. Diabetes Obes Metab 2010;12:167-177. ArticlePubMed
- 68. Kothny W, Foley J, Kozlovski P, Shao Q, Gallwitz B, Lukashevich V. Improved glycaemic control with vildagliptin added to insulin, with or without metformin, in patients with type 2 diabetes mellitus. Diabetes Obes Metab 2013;15:252-257. ArticlePubMed
- 69. Barnett AH, Charbonnel B, Donovan M, Fleming D, Chen R. Effect of saxagliptin as add-on therapy in patients with poorly controlled type 2 diabetes on insulin alone or insulin combined with metformin. Curr Med Res Opin 2012;28:513-523. ArticlePubMed
- 70. Duran-Garcia S, Lee J, Yki-Jarvinen H, Rosenstock J, Hehnke U, Thiemann S, Patel S, Woerle HJ. Efficacy and safety of linagliptin as add-on therapy to basal insulin and metformin in people with Type 2 diabetes. Diabet Med 2016;33:926-933. ArticlePubMed
- 71. Rosenstock J, Rendell MS, Gross JL, Fleck PR, Wilson CA, Mekki Q. Alogliptin added to insulin therapy in patients with type 2 diabetes reduces HbA(1C) without causing weight gain or increased hypoglycaemia. Diabetes Obes Metab 2009;11:1145-1152. ArticlePubMed
- 72. Cho YM. New clinical experience of gemigliptin, the new DPPIV inhibitor In: Proceedings of 9th International Diabetes Federation Western Pacific Region Congress & 4th Scientific Meeting of the Asian Association for the Study of Diabetes; 2012 Nov 25; Kyoto, JP.
- 73. Rhee EJ, Lee WY, Min KW, Shivane VK, Sosale AR, Jang HC, Chung CH, Nam-Goong IS, Kim JA, Kim SW. Gemigliptin Study 006 Group. Efficacy and safety of the dipeptidyl peptidase-4 inhibitor gemigliptin compared with sitagliptin added to ongoing metformin therapy in patients with type 2 diabetes inadequately controlled with metformin alone. Diabetes Obes Metab 2013;15:523-530. ArticlePubMed
- 74. Rhee EJ, Min KW, Jang HC, Chung CH, Nam-Goong IS, Bae HY, Baik SH, Shivane VK, Sosale AR, Dharmalingam M, Gandhi P, Gupta SK, Ahn JY, Bang KS, Lee MK. No. 111: Gemigliptin added to ongoing metformin therapy provides sustained glycemic control over 52 weeks and was well tolerated in patients with type 2 diabetes In: Oral presentation of 49th Annual Meeting of the European Association for the Study of Diabetes; 2013 Sep 23-27; Barcelona, ES.
- 75. Lee BW, Kim J, Park S, Jung C, Lee SH, Suh S, Lee W, Cho JH, Jang Y, Kim SH, Park CY. STABLE Study. No. 803: Effects of gemigliptin versus sitagliptin or glimepiride on glycaemic variability as initial combination therapy with metformin in drug-naïve patients with type 2 diabetes In: Poster presentation of 51st Annual Meeting of European Association for the Study of Diabetes; 2015 Sep 14-18; Stockholm, SZ.
- 76. Yoon S, Han B, Kim S, Han S, Jo YI, Jeong K, Oh KH, Park H, Park SH, Kang SW, Na KR, Jang Y, Kim SH, Cha D. No. 1199: Efficacy and safety of gemigliptin in type 2 diabetes patients with moderate to severe renal impairment In: Poster presentation of American Diabetes Association 76th Scientific sessions; 2016 Jun 10-14; New Orleans, LA.
- 77. Marfella R, Barbieri M, Grella R, Rizzo MR, Nicoletti GF, Paolisso G. Effects of vildagliptin twice daily vs. sitagliptin once daily on 24-hour acute glucose fluctuations. J Diabetes Complications 2010;24:79-83. ArticlePubMed
- 78. Rizzo MR, Barbieri M, Marfella R, Paolisso G. Reduction of oxidative stress and inflammation by blunting daily acute glucose fluctuations in patients with type 2 diabetes: role of dipeptidyl peptidase-IV inhibition. Diabetes Care 2012;35:2076-2082. PubMedPMC
REFERENCES
Fig. 1
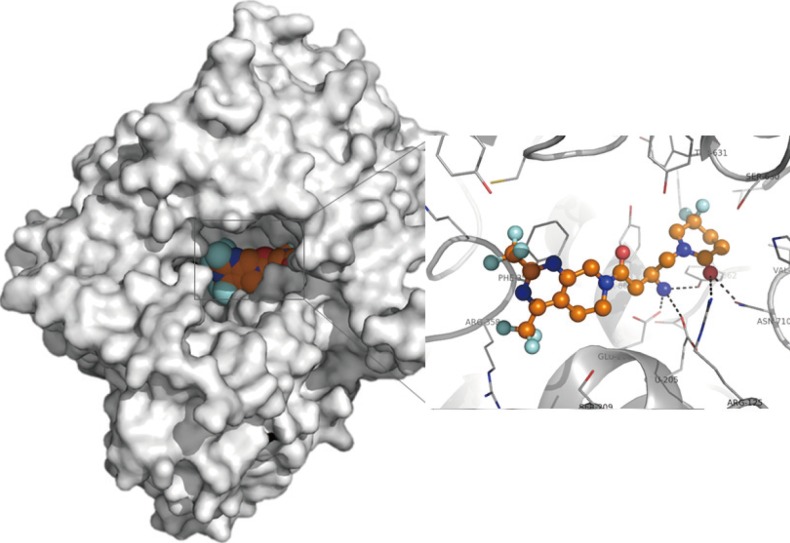
X-ray crystallographic structure of gemigliptin (orange) bound to the active site of dipeptidyl peptidase-4 enzyme.

Table 1
![dmj-40-339-i001.jpg]()
Key pharmacodynamics and pharmacokinetics properties of dipeptidyl peptidase-4 inhibitor
| Variable | Generic name (brand name) | ||||||
|---|---|---|---|---|---|---|---|
| Gemigliptin (Zemiglo) | Sitagliptin (Januvia) | Vildagliptin (Galvus) | Saxagliptin (Onglyza) | Linagliptin (Tragenta) | Alogliptin (Nesina) | Teneligliptin (Tenelia) | |
| Therapeutic dose, mg/day | 50 | 100 | 100 (50×2) | 5 | 5 | 25 | 20 |
| Dosing frequency | Once daily | Once daily | Twice daily | Once daily | Once daily | Once daily | Once daily |
| Chemistry [10,11] | |||||||
| Chemical structure | Pyrimidino piperidine derivative | Triazole pyrazine derivative | Cyanopyrrolidine | Cyanopyrrolidine | Xanthine derivative | Modified pyrimidinedione | L-prolylthiazolidine |
| Peptidomimetic | Peptidomimetic | Peptidomimetic | Peptidomimetic | Non-peptidomimetic | Non-peptidomimetic | Peptidomimetic | |
| Pharmacodynamics profiles [6,12,13,14] | |||||||
| Binding mode | S1, S2, S2 extensive subsites | S1, S2, S2 extensive subsites | S1, S2 subsites | S1, S2 subsites | S1, S2 & S1', S2' subsites | S1, S2 & S1', S2' subsites | S1, S2, S2 extensive subsites |
| Potency IC50, nmol/L | 6.3 | 19 | 62 | 50 | 1 | 24 | 1.8 |
| Selectivity (fold) | High | High | Moderate | Moderate | Moderate | High | High |
| DPP-8 | >27,000 | >2,600 | 270 | 390 | 40,000 | >14,000 | 700 |
| DPP-9 | >23,000 | >5,500 | 32 | 77 | >10,000 | >14,000 | 1,500 |
| FAP-α | >41,000 | >5,500 | 280 | >4,000 | 89 | >14,000 | >11,000 |
| Pharmacokinetics profiles [7,8,9,10,11,12] | |||||||
| Absorption | Rapid | Rapid | Rapid | Rapid | Rapid | Rapid | Rapid |
| No effect of food | No effect of food | No effect of food | No effect of food | No effect of food | No effect of food | No effect of food | |
| Cmax | 62.7 ng/mL | 959 nM | 397 ng/mL | 24 ng/mL (parent drug) | 9.6 nM | 110 ng/mL | 275 ng/mL |
| 47 ng/mL (metabolite) | |||||||
| Metabolism | Metabolized by ~10% to LC15-0636 | Modest metabolism (~16%) | Metabolized by ~55% to LAY151 | Metabolized to the active metabolite M2 | Minimally metabolism | Minimally metabolism | Metabolized by ~65.6% |
| Elimination half-life (t1/2), hr | 17.1 | 8–14 | 1.5–4.5 | 2–4 (parent drug) | 97–260 | 12–21 | 18.9–20 |
| 3–7 (metabolite) | |||||||
| Elimination route | Kidney and liver | Mainly kidney | Mainly kidney | Mainly kidney | Mainly liver | Mainly kidney | Kidney and liver |
| Renal (parent drug) | 63% (39%) | 87% (74%) | 85.4% (22.6%) | 74.9% (23.5%) | 5.5% (3.9%) | 75.6% (71.8%) | 45.4% (14.8%) |
| Feces (parent drug) | 27% (11%) | 13% (10%) | 14.8% (4.5%) | 22.1% (21.6%) | 81.5% (74.1%) | 12.9% (11.4%) | 46.5% (26.1%) |
| Dose reduction with RI (mild/moderate/severe) | No | Yes (100/50/25 mg) | Yes (50/50/50 mg) | Yes(5/2.5/2.5 mg) | No | Yes (25/12.5/6.25 mg) | No |
| Dose reduction with HI (mild/moderate) | No | No | No | No | No | No | No |
| Use with other drugs | - | - | - | Dose reduction with strong CYP3A4/5 inhibitors | - | - | - |
Table 2
![dmj-40-339-i002.jpg]()
Table 3
![dmj-40-339-i003.jpg]()
Efficacy of gemigliptin on glycemic control in patients with type 2 diabetes mellitus: summary of data from randomized controlled trials
| Parameter | Generic name (brand name) | ||||||
|---|---|---|---|---|---|---|---|
| Gemigliptin (Zemiglo) | Sitagliptin (Januvia) | Vildagliptin (Galvus) | Saxagliptin (Onglyza) | Linagliptin (Tragenta) | Alogliptin (Nesina) | Teneligliptin (Tenelia) | |
| Monotherapy, short-term [35,37,41,42,43,44,45,46,47,48,49,50,51] | |||||||
| Week | 12 | 18 | 24 | 12 | 18 | - | 12 |
| Baseline HbA1c | 8.24 | 8.04 | 8.56 | 7.9 | 8.1 | - | 7.8 |
| Change from baseline | –0.98 | –0.48 | –0.79 | –0.90 | –0.39 | - | –0.78 |
| Monotherapy, long-term | |||||||
| Week | 24 | 24 | 24 | 24 | 24 | 26 | 24 |
| Baseline HbA1c | 8.66 | 8.01 | 8.38 | 8.0 | 8.0 | 7.9 | 7.63 |
| Change from baseline | –1.24 | –0.61 | –0.72 | –0.5 | –0.44 | –0.6 | –0.90 |
| Initial combination therapy with metformin [37,52,53,54,55,56] | |||||||
| Week | 24 | 24 | 24 | 24 | 24 | 26 | - |
| Baseline HbA1c | 8.65 | 8.79 | 8.7 | 9.4 | 8.7 | 8.4 | - |
| Change from baseline | –2.06 | –1.90 | –1.8 | –2.5 | –1.6 | –1.6 | - |
| Combination therapy: add-on to metformin [38,57,58,59,60,61,62] | |||||||
| Week | 52 | 24 | 24 | 24 | 24 | 26 | 16 |
| Baseline HbA1c | 7.89 | 7.96 | 8.38 | 8.1 | 8.09 | 7.9 | 7.79 |
| Change from baseline | –1.06 | –0.67 | –0.88 | –0.7 | –0.49 | –0.6 | –0.87 |
| Combination therapy: add-on to metformin and sulfonylurea [39,63,64,65,66] | |||||||
| Week | 24 | 24 | 24 | 24 | 24 | - | - |
| Baseline HbA1c | 8.12 | 8.27 | 8.75 | 8.4 | 8.15 | - | - |
| Change from baseline | –0.88 | –0.59 | –1.01 | –0.74 | –0.72 | - | - |
| Combination therapy: add-on to insulin [40,67,68,69,70,71] | |||||||
| Week | 12a | 24 | 24 | 24 | 24 | 26 | - |
| Baseline HbA1c | 8.41 | 8.7 | 8.8 | 8.7 | 8.3 | 9.3 | - |
| Change from baseline | –0.95 | –0.6 | –0.8 | –0.7 | –0.6 | –0.7 | - |
Table 4
![dmj-40-339-i004.jpg]()
Lipid parameters in pivotal studies of gemigliptin
Table 5
![dmj-40-339-i005.jpg]()
Frequency of reported adverse events
Table 6
![dmj-40-339-i006.jpg]()
Reported hypoglycemia and change in body weight
Table 7
![dmj-40-339-i007.jpg]()
Article highlights
Figure & Data
References
Citations
Citations to this article as recorded by 

- A Multicenter, Randomized, Open-Label Study to Compare the Effects of Gemigliptin Add-on or Escalation of Metformin Dose on Glycemic Control and Safety in Patients with Inadequately Controlled Type 2 Diabetes Mellitus Treated with Metformin and SGLT-2 Inh
Hae Jin Kim, Jung Hyun Noh, Min Kyong Moon, Sung Hee Choi, Seung-Hyun Ko, Eun-Jung Rhee, Kyu Yeon Hur, In-Kyung Jeong, Mark Yorek
Journal of Diabetes Research.2024; 2024: 1. CrossRef - Gemigliptin, a potent selective dipeptidyl peptidase 4 inhibitor, protects endothelial progenitor cells by oxidative stress via caspase-3 dependent pathway
Mijung Lee, Amna Rashid Tariq, Manho Kim
Biochemistry and Biophysics Reports.2024; 38: 101673. CrossRef - Selective targeting of dipeptidyl‐peptidase 4 (DPP‐4) positive senescent chondrocyte ameliorates osteoarthritis progression
Du Hyun Ro, Gun Hee Cho, Ji Yoon Kim, Seong Ki Min, Ha Ru Yang, Hee Jung Park, Sun Young Wang, You Jung Kim, Myung Chul Lee, Hyun Cheol Bae, Hyuk‐Soo Han
Aging Cell.2024;[Epub] CrossRef - Opportunities and challenges of incretin-based hypoglycemic agents treating type 2 diabetes mellitus from the perspective of physiological disposition
Yaochen Xie, Qian Zhou, Qiaojun He, Xiaoyi Wang, Jincheng Wang
Acta Pharmaceutica Sinica B.2023; 13(6): 2383. CrossRef - Therapeutic Effects of Switching to Anagliptin from Other DPP-4 Inhibitors in T2DM Patients with Inadequate Glycemic Control: A Non-interventional, Single-Arm, Open-Label, Multicenter Observational Study
Sang-Yong Kim, Sungrae Kim
Diabetes Therapy.2023; 14(1): 109. CrossRef - Pharmacokinetic and pharmacodynamic interaction of DWP16001, a sodium–glucose cotransporter 2 inhibitor, with gemigliptin and metformin in healthy adults
Sae Im Jeong, Yun Kim, Jae Jin Nah, Wan Huh, In‐Jin Jang, Jun Gi Hwang, SeungHwan Lee
British Journal of Clinical Pharmacology.2023; 89(6): 1780. CrossRef - 1,2,3‐Triazole analogs with bulky and conformationally rigid substructures: Synthesis and in vitro evaluation as DPP‐4 inhibitors
Duy‐Viet Vo, Jongkook Lee, Haeil Park
Bulletin of the Korean Chemical Society.2023; 44(5): 425. CrossRef - DPP-4 inhibitors for treating T2DM - hype or hope? an analysis based on the current literature
Kunika Saini, Smriti Sharma, Yousuf Khan
Frontiers in Molecular Biosciences.2023;[Epub] CrossRef - The effect of gemigliptin treatment on immune parameters including regulatory T cells in patients with type 2 diabetes and moderate to very severe chronic renal impairment
Yanghyeon Kim, Nagyeom Lee, Sujung Heo, Ye Na Kim, Ho Sik Shin, Yeonsoon Jung, Hark Rim
Medicine.2023; 102(49): e36455. CrossRef - Increasing Age Associated with Higher Dipeptidyl Peptidase-4 Inhibition Rate Is a Predictive Factor for Efficacy of Dipeptidyl Peptidase-4 Inhibitors
Sangmo Hong, Chang Hee Jung, Song Han, Cheol-Young Park
Diabetes & Metabolism Journal.2022; 46(1): 63. CrossRef - Does DPP-IV Inhibition Offer New Avenues for Therapeutic Intervention in Malignant Disease?
Petr Busek, Jonathan S. Duke-Cohan, Aleksi Sedo
Cancers.2022; 14(9): 2072. CrossRef - Divergent Reaction of Activated Pyridines with α,α-Difluorinated gem-Diols: Regioselective Synthesis of gem-Difluorinated Dihydropyridines and Dihydropyridones
Koushik Patra, Mallu Kesava Reddy, Sumitava Mallik, Mahiuddin Baidya
Organic Letters.2022; 24(22): 4014. CrossRef - Gemigliptin exerts protective effects against doxorubicin-induced hepatotoxicity by inhibiting apoptosis via the regulation of fibroblast growth factor 21 expression
Kyeong-Min Lee, Yeo Jin Hwang, Gwon-Soo Jung
Biochemical and Biophysical Research Communications.2022; 626: 135. CrossRef - FDA-Approved Trifluoromethyl Group-Containing Drugs: A Review of 20 Years
Aathira Sujathan Nair, Ashutosh Kumar Singh, Astik Kumar, Sunil Kumar, Sunitha Sukumaran, Vishal Payyalot Koyiparambath, Leena K. Pappachen, T. M. Rangarajan, Hoon Kim, Bijo Mathew
Processes.2022; 10(10): 2054. CrossRef - Glucagon-like peptide-1 (GLP-1) receptor agonists and neuroinflammation: Implications for neurodegenerative disease treatment
Katherine O. Kopp, Elliot J. Glotfelty, Yazhou Li, Nigel H. Greig
Pharmacological Research.2022; 186: 106550. CrossRef - An evaluation of drug lag for new drugs approved by the Indian regulator relative to the United States, European Union, and Japanese regulatory agencies: A 15-year analysis (2004–2018)
Mahanjit Konwar, MiteshR Maurya, TusharB Nishandar, UrmilaM Thatte, NithyaJ Gogtay
Perspectives in Clinical Research.2021; 12(3): 159. CrossRef - Pharmacokinetic and Pharmacodynamic Comparison of Two Formulations of a Fixed-Dose Combination of Gemigliptin/Rosuvastatin 50/20 mg: A Randomized, Open-Label, Single-Dose, Two-Way Crossover Study
Eunsol Yang, Hyounggyoon Yoo, In-Jin Jang, Kyung-Sang Yu, SeungHwan Lee
Drug Design, Development and Therapy.2021; Volume 15: 651. CrossRef - Efficacy and Safety of the Novel Dipeptidyl Peptidase-4 Inhibitor Gemigliptin in the Management of Type 2 Diabetes: A Meta-Analysis
Deep Dutta, Anshita Agarwal, Indira Maisnam, Rajiv Singla, Deepak Khandelwal, Meha Sharma
Endocrinology and Metabolism.2021; 36(2): 374. CrossRef - Structure–Activity Relationship Analysis of Cocrystallized Gliptin-like Pyrrolidine, Trifluorophenyl, and Pyrimidine-2,4-Dione Dipeptidyl Peptidase-4 Inhibitors
Katarina Tomovic, Budimir S. Ilic, Andrija Smelcerovic
Journal of Medicinal Chemistry.2021; 64(14): 9639. CrossRef - A review upon medicinal perspective and designing rationale of DPP-4 inhibitors
Shubham Kumar, Anu Mittal, Amit Mittal
Bioorganic & Medicinal Chemistry.2021; 46: 116354. CrossRef - Effect of Dipeptidyl Peptidase-4 (DPP-4) Inhibition on Biomarkers of Kidney Injury and Vascular Calcification in Diabetic Kidney Disease: A Randomized Controlled Trial
Thananda Trakarnvanich, Bancha Satirapoj, Swangjit Suraamornkul, Thanit Chirananthavat, Anoma Sanpatchayapong, Torpong Claimon, Eusebio Chiefari
Journal of Diabetes Research.2021; 2021: 1. CrossRef - Antidiabetic effect of gemigliptin: a systematic review and meta-analysis of randomized controlled trials with Bayesian inference through a quality management system
Hojin Oh, Hai Duc Nguyen, In Mo Yoon, Byung-Ryong Ahn, Min-Sun Kim
Scientific Reports.2021;[Epub] CrossRef - Pleiotropic Benefits of DPP-4 Inhibitors Beyond Glycemic Control
Seon Mee Kang, Jeong Hyun Park
Clinical Medicine Insights: Endocrinology and Diabetes.2021; 14: 117955142110516. CrossRef - The vasodilatory effect of gemigliptin via activation of voltage-dependent K+ channels and SERCA pumps in aortic smooth muscle
Hee Seok Jung, Mi Seon Seo, Jin Ryeol An, Minji Kang, Ryeon Heo, Hongliang Li, Won-Kyo Jung, Il-Whan Choi, Eun-Hee Cho, Hongzoo Park, Young Min Bae, Won Sun Park
European Journal of Pharmacology.2020; 882: 173243. CrossRef - Development and Validation of a Rapid and Sensitive Method for the Simultaneous Estimation of Gemigliptin and Teneligliptin in Bulk and Dosage Forms by Using Liquid Chromatography-tandem Mass Spectrometry
Amrish Chandra, Ramji Rathod, Faraat Ali, Anuj Prakash, Robin Kumar, Gyanendra Nath Singh
Current Pharmaceutical Analysis.2020; 16(8): 1104. CrossRef - Incretin Mimetics as Rational Candidates for the Treatment of Traumatic Brain Injury
Elliot J. Glotfelty, Thomas E. Delgado, Luis B. Tovar-y-Romo, Yu Luo, Barry J. Hoffer, Lars Olson, Tobias E. Karlsson, Mark P. Mattson, Brandon K. Harvey, David Tweedie, Yazhou Li, Nigel H. Greig
ACS Pharmacology & Translational Science.2019; 2(2): 66. CrossRef - Mechanisms and pathways of anti‐inflammatory activity of DPP‐4 inhibitors in cardiovascular and renal protection
Katarina Tomovic, Jelena Lazarevic, Gordana Kocic, Marina Deljanin‐Ilic, Marko Anderluh, Andrija Smelcerovic
Medicinal Research Reviews.2019; 39(1): 404. CrossRef - Dipeptidyl Peptidase-4 Inhibitors versus Other Antidiabetic Drugs Added to Metformin Monotherapy in Diabetic Retinopathy Progression: A Real World-Based Cohort Study
Yoo-Ri Chung, Kyoung Hwa Ha, Hyeon Chang Kim, Sang Jun Park, Kihwang Lee, Dae Jung Kim
Diabetes & Metabolism Journal.2019; 43(5): 640. CrossRef - Effect of gemigliptin on cardiac ischemia/reperfusion and spontaneous hypertensive rat models
Dae-Hwan Nam, Jinsook Park, Sun-Hyun Park, Ki-Suk Kim, Eun Bok Baek
The Korean Journal of Physiology & Pharmacology.2019; 23(5): 329. CrossRef - Clinical Use of DPP-4 Inhibitors
Baptist Gallwitz
Frontiers in Endocrinology.2019;[Epub] CrossRef - Efficacy and Safety of Switching to Teneligliptin in Patients with Type 2 Diabetes Inadequately Controlled with Dipeptidyl Peptidase-4 Inhibitors: A 12-Week Interim Report
Hae Jin Kim, Young Sik Kim, Chang Beom Lee, Moon-Gi Choi, Hyuk-Jae Chang, Soo Kyoung Kim, Jae Myung Yu, Tae Ho Kim, Ji Hyun Lee, Kyu Jeung Ahn, Kyoungmin Kim, Kwan Woo Lee
Diabetes Therapy.2019; 10(4): 1271. CrossRef - Efficacy and Safety of Gemigliptin in Post-Transplant Patients With Type 2 Diabetes Mellitus
Jaehyun Bae, Youjin Kim, Yongin Cho, Minyoung Lee, Ji-Yeon Lee, Yong-ho Lee, Byung-Wan Lee, Bong-Soo Cha, Dong Jin Joo, Kyu Ha Huh, Myoung Soo Kim, Yu Seun Kim, Eun Seok Kang
Transplantation Proceedings.2019; 51(10): 3444. CrossRef - Protective effects of gemigliptin against type II collagen degradation in human chondrocytes
Momin Mohetaer, Guoqing Li, Yang Wang, Li Cao
Biomedicine & Pharmacotherapy.2018; 104: 590. CrossRef - Pharmacokinetic Interactions Between Gemigliptin and Metformin, and Potential Differences in the Pharmacokinetic Profile of Gemigliptin Between the Mexican and Korean Populations: A Randomized, Open-label Study in Healthy Mexican Volunteers
Ignacio Conde-Carmona, Sandra García-Medina, Juan M. Jiménez-Vargas, Alberto Martínez-Muñoz, Sung-Hack Lee
Clinical Therapeutics.2018; 40(10): 1729. CrossRef - Comparative efficacy and safety of gemigliptin versus linagliptin in type 2 diabetes patients with renal impairment: A 40‐week extension of the GUARD randomized study
Sang Youb Han, Sun Ae Yoon, Byoung Geun Han, Sung Gyun Kim, Young‐Il Jo, Kyung Hwan Jeong, Kook‐Hwan Oh, Hyeong Cheon Park, Sun‐Hee Park, Shin‐Wook Kang, Ki‐Ryang Na, Sun Woo Kang, Nam‐Ho Kim, Younghwan Jang, Bogyeong Kim, Seonghye Shin, Dae Ryong Cha
Diabetes, Obesity and Metabolism.2018; 20(2): 292. CrossRef - Comparative Cardiovascular Risks of Dipeptidyl Peptidase-4 Inhibitors: Analyses of Real-world Data in Korea
Kyoung Hwa Ha, Bongseong Kim, Hae Sol Shin, Jinhee Lee, Hansol Choi, Hyeon Chang Kim, Dae Jung Kim
Korean Circulation Journal.2018; 48(5): 395. CrossRef - Gemigliptin: Newer promising gliptin for type 2 diabetes mellitus
Manish Gutch, Abhay Joshi, Sukriti Kumar, Avinash Agarwal, RajendraKumar Pahan, SyedMohd Razi
Indian Journal of Endocrinology and Metabolism.2017; 21(6): 898. CrossRef - Protective Effects of Gemigliptin, a Dipeptidyl Peptidase-4 Inhibitor, against Cisplatin-Induced Nephrotoxicity in Mice
Seung Hee Choi, Jaechan Leem, In-Kyu Lee
Mediators of Inflammation.2017; 2017: 1. CrossRef - Effects of gemigliptin, a dipeptidyl peptidase‐4 inhibitor, on lipid metabolism and endotoxemia after a high‐fat meal in patients with type 2 diabetes
Chang Ho Ahn, Eun Ky Kim, Se Hee Min, Tae Jung Oh, Young Min Cho
Diabetes, Obesity and Metabolism.2017; 19(3): 457. CrossRef - Efficacy and safety of gemigliptin, a dipeptidyl peptidase‐4 inhibitor, in patients with type 2 diabetes mellitus inadequately controlled with combination treatment of metformin and sulphonylurea: a 24‐week, multicentre, randomized, double‐blind, placebo‐
Chang Ho Ahn, Kyung Ah Han, Jae Myung Yu, Joo Young Nam, Kyu Jeung Ahn, Tae Keun Oh, Hyoung Woo Lee, Dae Ho Lee, Jaetaek Kim, Choon Hee Chung, Tae Sun Park, Byung Joon Kim, Seok Won Park, Hyeong Kyu Park, Kwang Jae Lee, Sang‐Wook Kim, Jeong Hyun Park, Kwa
Diabetes, Obesity and Metabolism.2017; 19(5): 635. CrossRef
- Figure
- Related articles
-
- Glycemic Control and Adverse Clinical Outcomes in Patients with Chronic Kidney Disease and Type 2 Diabetes Mellitus: Results from KNOW-CKD
- Intensified Multifactorial Intervention in Patients with Type 2 Diabetes Mellitus
- Not Control but Conquest: Strategies for the Remission of Type 2 Diabetes Mellitus
- A Real-World Study of Long-Term Safety and Efficacy of Lobeglitazone in Korean Patients with Type 2 Diabetes Mellitus

 KDA
KDA PubReader
PubReader Cite
Cite




