Risk Factors for the Progression of Intima-Media Thickness of Carotid Arteries: A 2-Year Follow-Up Study in Patients with Newly Diagnosed Type 2 Diabetes
Article information
Abstract
Background
Intima-media thickness (IMT) of the carotid arteries is known to have a positive correlation with the risk of cardiovascular disease. This study was designed to identify risk factors affecting the progression of carotid IMT in patients with type 2 diabetes mellitus (T2DM).
Methods
Patients with newly diagnosed T2DM with carotid IMT measurements were enrolled, and their clinical data and carotid IMT results at baseline and 2 years later were compared.
Results
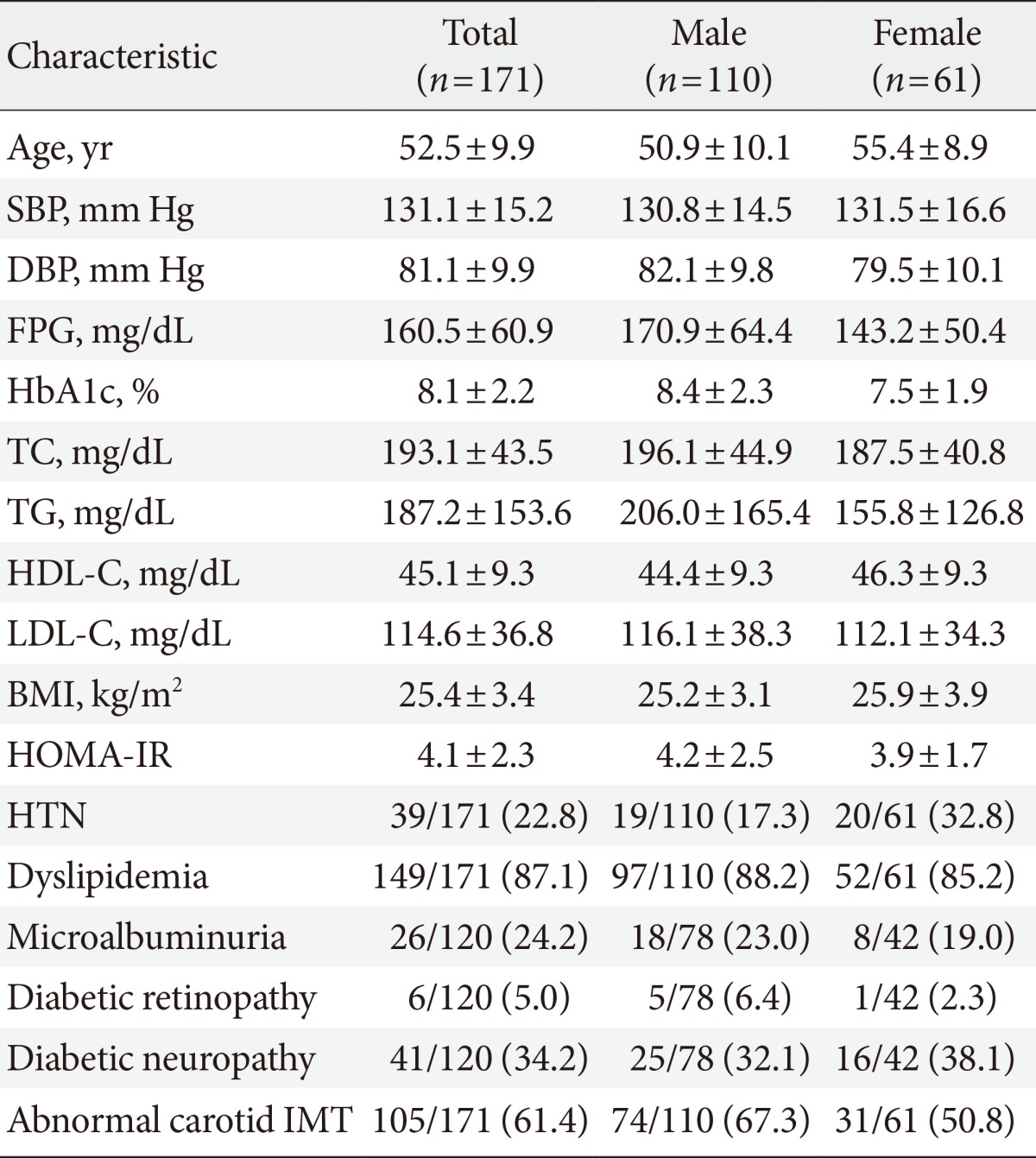
Of the 171 patients, 67.2% of males and 50.8% of females had abnormal baseline IMT of the left common carotid artery. At baseline, systolic blood pressure, body mass index and smoking in male participants, and fasting plasma glucose and glycated hemoglobin levels in females were significantly higher in patients with abnormal IMT than in those with normal IMT. Low density lipoprotein cholesterol (LDL-C) levels in males and high density lipoprotein cholesterol (HDL-C) levels in females at the 2-year follow-up were significantly different between the nonprogression and the progression groups. Reduction of the United Kingdom Prospective Diabetes Study (UKPDS) 10-year coronary heart disease (CHD) risk score after 2 years was generally higher in the nonprogression group than the progression group.
Conclusion
LDL-C levels in males and HDL-C levels in females at the 2-year follow-up were significantly different between participants with and without progression of carotid IMT. Furthermore, a reduction in the UKPDS 10-year CHD risk score appeared to delay the advancement of atherosclerosis. Therefore, the importance of establishing the therapeutic goal of lipid profiles should be emphasized to prevent the progression of carotid IMT in newly diagnosed T2DM patients.
INTRODUCTION
The incidence and complications of type 2 diabetes mellitus (T2DM) continue to increase rapidly [1]. Insulin resistance and hyperglycemia exacerbate macrovascular complications in diabetic patients when compared with nondiabetic patients. The disequilibrium between increases in toxic metabolites (e.g., advanced glycation end-products, protein kinase C activation, and oxidative stress) and decreases in protective endogenous factors (e.g., insulin, vascular endothelial growth factor, platelet-derived growth factors, and adiponectin) may play a role in the development of diabetic complications [2], which raises the likelihood of cardiovascular disease (CVD), such as myocardial infarction and stroke [3].
Early diagnosis and treatment of atherosclerosis are necessary to prevent serious diabetic complications. Many diagnostic tools, including myocardial scans, computed tomography, and magnetic resonance imaging, are used for early detection of atherosclerosis, but their applications are currently limited due to their high costs and low accuracy [4]. In contrast, the intima-media thickness (IMT) of the carotid arteries as measured by noninvasive method using B-mode ultrasound is known to be a useful marker of atherosclerosis and has been reported to be significantly correlated with the actual carotid IMT as determined by histologic examination [5,6]. Furthermore, previous studies identified a positive correlation between carotid IMT and risk of CVD [7-9].
The Insulin Resistance Atherosclerosis Study reported that carotid IMT did not significantly differ between diabetic and nondiabetic participants at the time of diagnosis, but significant differences in carotid IMT between these two groups were detectable after several years [10,11]. Diabetes mellitus (DM) duration was one of the factors that impacted this difference in carotid IMT [12], and the rate of increase in carotid IMT was significantly higher in diabetic patients than in nondiabetic participants [13].
However, few studies have investigated the risk factors that aggravate the progression of carotid atherosclerosis among patients with T2DM. This study was designed to determine the risk factors affecting the progression of carotid IMT in Korean patients newly diagnosed with T2DM.
METHODS
Study population
The study population comprised of patients with T2DM that were newly diagnosed between January 2007 and June 2008 at Kyung Hee University Hospital at Gangdong. All participants had initial carotid IMT measurements by ultrasonography at the time of their T2DM diagnosis. Patients with T2DM satisfied the criteria of the American Diabetic Association (ADA) [14]. Those with a previous history of CVD and trauma, surgical or radiation therapy at the cervical region, including the carotid artery, were excluded. All participants in this study provided informed consent before the research began.
Carotid IMT measurements
High-resolution B-mode carotid ultrasonography was performed at baseline and after a 2-year follow-up period using identical scanning protocols and equipment (Envisor HD11XE; Koninklijke Philips Electronics, Eindhoven, the Netherlands). During the ultrasonographic examination, all participants were put in the supine position with their heads straight and their necks extended. Scanning was performed by insonating the vessel in a perpendicular orientation, optimizing the B-mode settings of gain, depth and focal zone placement, and compressing to enhance the image quality. The definition of IMT was the distance from the leading edge of the lumen-intima interface to the leading edge of the media-adventitia interface of the far wall [15]. A single longitudinal lateral image of the left common carotid artery (CCA) with the regular lumen-intima interface parallel to the adventitia was captured and transferred to a personal computer for automated measurements. IMT measurement was taken using Intimascope software (Media Cross Co., Ltd., Tokyo, Japan) that performs automated IMT measurements based on an algorithm that delineates the lumen-intima and media adventitia interfaces [16]. Two lines along the boundaries of the IMT that corresponded to the measurement points within the set segment were drawn manually. All of the measurements were made by a single certified sonographer for both years of the study. The present study used the mean IMT value obtained from Intimascope. The definition of a plaque was a focal structure that protruded by at least 0.5 mm into the arterial lumen or that occupied the vascular lumen with its thickness greater than 50% of the surrounding IMT value or greater than 1.5 mm as measured from the media-adventitia interface to the intima-lumen interface [15].
Analyses of clinical parameters
Systolic blood pressure (SBP) and diastolic blood pressure (DBP) were taken in the right arm and recorded as the mean of the two measurements obtained at least 5 minutes apart. Participants with hypertension included those who had either a SBP greater than 140 mm Hg or a DBP greater than 90 mm Hg. Any participant currently taking antihypertensive medications was also classified as hypertensive. Dyslipidemia was defined using the following criteria established by the National Cholesterol Education Program Adult Treatment Panel III: triglyceride (TG) ≥150 mg/dL, low density lipoprotein cholesterol (LDL-C) ≥100 mg/dL or high density lipoprotein cholesterol (HDL-C) ≤40 mg/dL in males and ≤50 mg/dL in females [17]. Those currently taking lipid-lowering medications were also classified as having dyslipidemia. All participants fasted for at least 8 hours before venous sampling. TG was measured by enzymatic colorimetric methods. HDL-C concentration in the supernatant was determined by an enzymatic colorimetric method after precipitation with dextran sulfate and magnesium chloride. The LDL-C concentration was directly assessed using the β-quantification procedure. Body mass index was calculated by dividing the weight in kilograms by the height in meters squared. The homeostasis model assessment-insulin resistance (HOMA-IR) was determined using the following equation: fasting glucose (mg/dL)×fasting insulin (µU/mL)/405 [18]. Cigarette smoking was ascertained by educational nurses during an interview. Diabetic retinopathy (DR) was defined when retinal photography revealed nonproliferative DR, proliferative DR (PDR), diabetic macular edema, or any combination thereof [19]. Diabetic nephropathy was determined to be when the urinary albumin to creatinine level was ≥30 mg/g [20]. Diabetic neuropathy was identified when a neuropathy consistent with diabetes was present or nerve conduction studies confirmed its presence [21]. Medical records were thoroughly reviewed to obtain information regarding the past medication use, including antihypertensive, antiplatelet, and lipid-lowering agents.
Calculated risks of the United Kingdom Prospective Diabetes Study risk engine
Risk for coronary heart disease (CHD) was estimated by substituting risk factors such as age, gender, race, current smoking status, glycated hemoglobin (HbA1c), SBP, total cholesterol, HDL-C, and presence or absence of atrial fibrillation into the United Kingdom Prospective Diabetes Study (UKPDS) risk engine calculator, which was provided by the Diabetes Trials Units: The Oxford Centre for Diabetes, Endocrinology and Metabolism (http://www.dtu.ox.ac.uk/riskengine) [22-24]. ΔCHD risk was calculated by subtracting the UKPDS 10-year CHD risk at the 2-year follow-up from that at baseline.
Data and statistical analyses
Baseline characteristics were classified according to gender and expressed as means and standard deviations. Abnormal carotid IMT was defined when the IMT was higher than the upper limit of the normal range of carotid IMT that was obtained from age- and gender-matched data from the Health Promotion Center, Kyung Hee University Hospital. In each age group, 30 participants with no cardiovascular risk factors, such as T2DM, hypertension, dyslipidemia, and smoking history, were randomly selected, and their carotid IMT values were measured to obtain the normal range (Supplementary Table 1). Participants with plaques were also classified as having an abnormal IMT. Since the Atherosclerosis Risk in Communities (ARIC) study reported that the mean carotid IMT increased by 0.007 to 0.01 mm annually [25], the mean carotid IMT should therefore increase by 0.014 to 0.02 mm during the 2-year follow-up period. Based on this assumption, progression of carotid IMT was defined when the left mean carotid IMT at the 2-year follow-up had increased by more than 0.01 mm compared with the baseline. Participants with nonprogression of carotid IMT included those whose left mean carotid IMT at the 2-year follow-up examination had decreased by more than 0.01 mm or had remained unchanged. The significance of differences between groups was assessed using Student t-test or chi-square tests using PASW version 18.0 (IBM Co., Armonk, NY, USA). A P<0.05 was considered statistically significant.
RESULTS
Clinical characteristics of the study participants
This study enrolled 171 participants (male:female, 110:61), and their mean age was 52.5±9.9 years (males, 50.9±10.1; females, 55.4±8.9) (Table 1). SBP and DBP of both males and females were largely within the target BP recommended in the ADA consensus guidelines [14]. The mean fasting plasma glucose (FPG) and HbA1c were 160.5±60.0 mg/dL and 8.1%±2.2%, respectively; both of these mean values were above the therapeutic target level for T2DM. The mean LDL-C value was also higher than the treatment target for hyperlipidemia in patients with T2DM (114.6±36.8 mg/dL). BMI and incidence of smoking were significantly higher in male participants in the abnormal IMT group than those in the normal IMT group. The mean HOMA-IR was 4.1±2.3, which demonstrated insulin resistance in the study participants. Of the 171 participants, 24.2% had microalbuminuria, and 5.0% and 34.2% had accompanying DR and neuropathy, respectively. Abnormal baseline IMT of the left CCA was found in 61.4% of the participants, which included 67.2% of males (74/110) and 50.8% of females (31/61). Comparison of baseline and 2-year follow-up values revealed that the mean IMT of male participants was significantly lower at the 2-year follow-up compared to baseline (0.65±0.16 mm vs. 0.61±0.11 mm; P=0.008), whereas the mean IMT of female participants showed a nonsignificant decrease 2 years after diagnosis (0.65±0.18 mm vs. 0.62±0.17 mm; P=0.107) (Table 2). The number of males with newly observed plaques in the left CCA was lower at the 2-year follow-up than at baseline, while this number remained the same in females.
Comparison of participants with and without abnormal baseline carotid IMT
Comparison of the baseline clinical characteristics of males with normal and abnormal baseline mean IMT values revealed that SBP and BMI were significantly higher in those participants with an abnormal baseline IMT (Table 3). In addition, the percentage of males who smoked was significantly higher in the abnormal IMT group than in the normal IMT group. The same analysis among female participants revealed that FPG and HbA1c were significantly higher in those participants with an abnormal baseline IMT than they were in those with a normal baseline IMT. The UKPDS 10-year CHD risk at baseline did not differ significantly between participants with normal or abnormal IMT values regardless of gender. The number of patients with microalbuminuria, DR, or neuropathy did not differ significantly between participants with normal or abnormal baseline IMT regardless of gender.
Comparison between participants with and without progression of carotid IMT
There were 113 participants who underwent follow-up carotid IMT measurements (male:female, 73:40). We assigned these participants with a normal mean IMT at both baseline and the 2-year follow-up period to the nonprogression group, while those with a normal IMT at baseline but an abnormal IMT at 2 years were placed in the progression group (Table 4 for males and Table 5 for females). In both male and female participants, most clinical parameters had improved at the 2-year follow-up period compared with baseline levels. When baseline and follow-up levels of all parameters in the nonprogression group were compared with those in the progression group, the LDL-C level at the 2-year follow-up was significantly higher in males of the progression group than in the nonprogression group (79.79±24.6 mg/dL vs. 99.9±34.6 mg/dL; P=0.007) (Table 4, Fig. 1A). Interestingly, in females, the HDL-C level at the 2-year follow-up was significantly lower in the progression group than in the nonprogression group (53.0±14.9 mg/dL vs. 44.1±12.0 mg/dL; P=0.049) (Table 5, Fig. 1B). In females, the UKPDS 10-year CHD risk at baseline tended to be higher in the nonprogression group than the progression group, but the risk reduction after 2 years was significantly higher in the nonprogression group (-2.3±4.7 vs. +1.6±3.6; P=0.005) (Table 5). A similar trend was found in males (-4.9±11.2 vs. -0.7±8.1; P=0.008) (Table 5). The use of medications, such as antiplatelet agents, statins, and antihypertensive agents at the time of diagnosis did not significantly differ between the progression and nonprogression groups of both males and females.
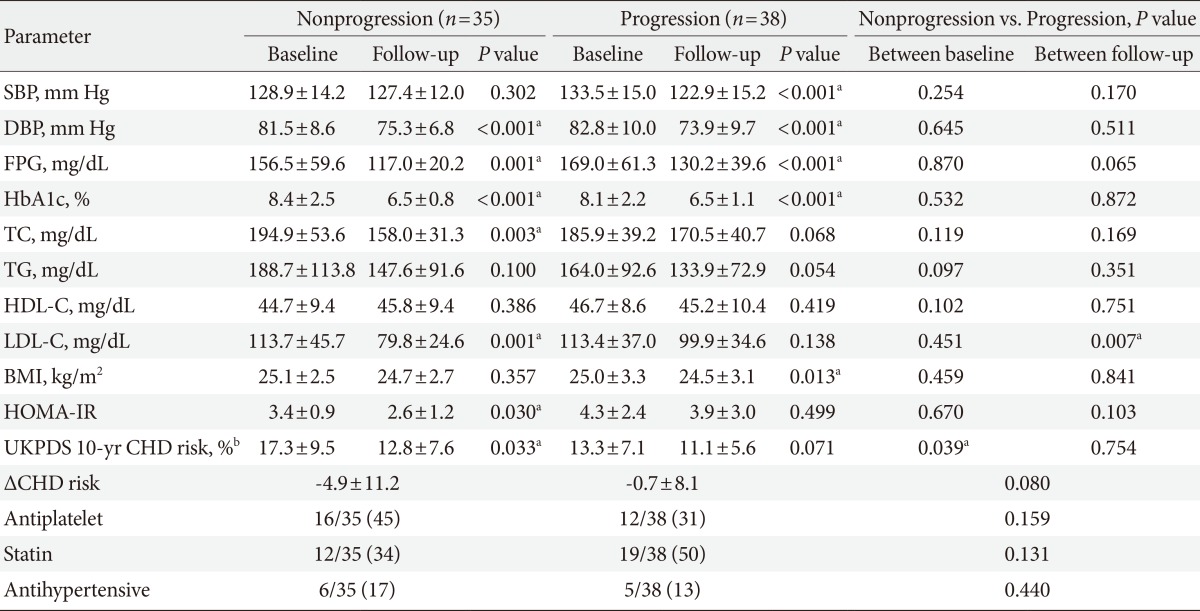
Comparison of clinical parameters between subjects according to the presence of progression of mean intima-media thickness at left common carotid artery in male
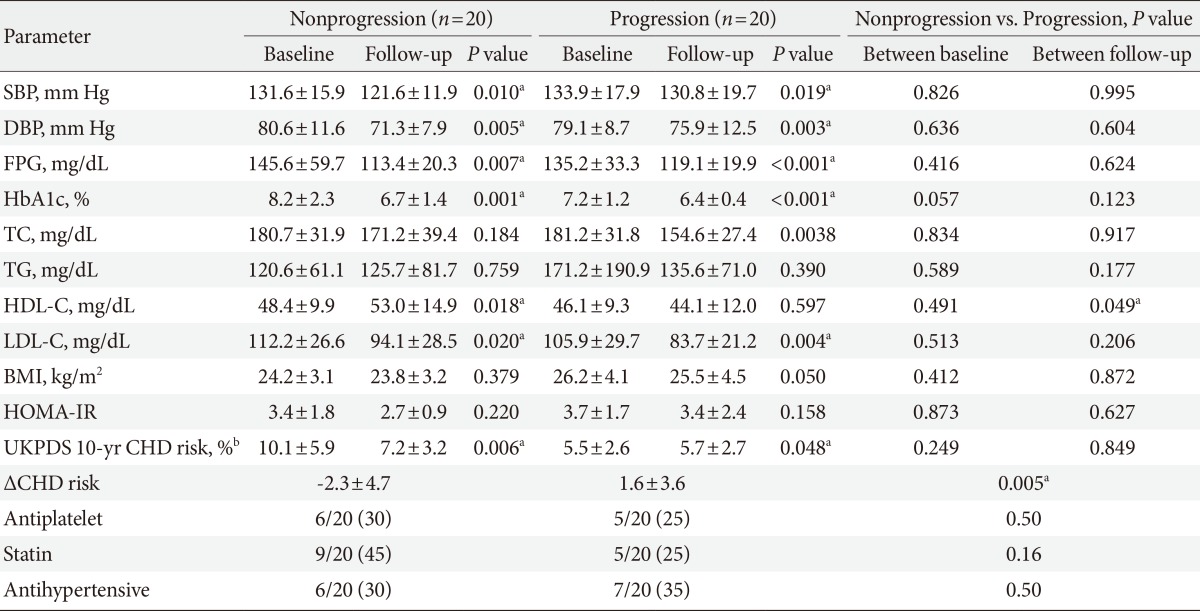
Comparison of clinical parameters between subjects according to the presence of progression of mean intima-media thickness at left common carotid artery in female
DISCUSSION
The present study demonstrated that LDL-C levels in males and HDL-C levels in females 2 years after the diagnosis of T2DM were significantly different between those participants with and without carotid IMT progression. Furthermore, although the UKPDS 10-year CHD risk score at baseline was generally higher in the nonprogression group than the progression group, the risk reduction (ΔCHD risk) of the nonprogression group 2 years after diagnosis was also higher than that of the progression group. These results suggested that active and comprehensive control of LDL-C, HDL-C, glucose levels, and blood pressure after the diagnosis of T2DM were more important than the baseline levels of these variables in preventing the progression of atherosclerosis.
Carotid IMT measurement is a noninvasive procedure that enables clinicians to predict the future risk of CVD [7-9,26]. When compared with their nondiabetic counterparts, newly diagnosed diabetic patients display an increased carotid IMT at the time of diagnosis [27]. However, in clinical practice, it is common to encounter patients with well-controlled DM but also concurrent aggravation of carotid IMT. Thus, it is relatively difficult to predict possible candidates at higher risk for progression of carotid IMT. The present study was designed to identify risk factors that would enable clinicians to anticipate the progression of carotid IMT.
Participants with a past history of coronary artery disease and lower HDL-C levels have been reported to experience more rapid progression of carotid IMT than those without these risk factors [28]. Furthermore, the ARIC study revealed that diabetes, current smoking, HDL-C, pulse pressure, white blood cell count, and fibrinogen concentration were significantly associated with the progression of carotid IMT [25]. In a Japanese study of a male working population, the TC level appeared to be the only significant determinant of the progression of carotid IMT [29].
Our study showed that at the time of diagnosis of T2DM, the mean carotid IMT was 0.65±0.16 mm in males and 0.65±0.18 mm in females. Bae et al. [30] reported that the mean carotid IMT values of normal participants between 45 and 64 years of age were 0.64±0.12 and 0.70±0.11 mm for males and 0.54±0.09 and 0.71±0.12 mm in females. One meta-analysis by Brohall et al. [31] demonstrated that patients with T2DM had a 0.13-mm thicker carotid IMT than normal participants. In a Korean study, Seon et al. [32] reported the mean carotid IMT of Korean patients newly diagnosed with T2DM was 0.67±0.15 mm. Also, Park et al. [33] found that approximately 50% of newly diagnosed T2DM patients accompanied the carotid plaque, which implies that the prevalence of abnormal carotid IMT in newly diagnosed T2DM patients may exceed 50%. When a comparison was made between the 2-year follow-up data and baseline data between participants with and without carotid IMT progression, the LDL-C level at the 2-year follow-up was significantly higher in males in the progression group than in males from the nonprogression group, though most parameters had improved at follow-up compared with the baseline data. It should be noted that in the progression group, the mean LDL-C level at the 2-year follow-up had decreased below 100 mg/dL but was still significantly higher in the progression group than the non-progression group (99.9±34.6 mg/dL vs. 79.8±24.6 mg/dL; P=0.007) (Table 4).
This result was different from the previously reported data that demonstrated a significant association between HDL-C levels and IMT progression [25,28]. Insulin resistance itself does not play a principal role in escalating LDL-C [34]. Witztum [35] proposed an oxidation hypothesis of atherosclerosis in which LDL particle oxidation is stimulated by coexisting insulin resistance and consequently acts as a strong inflammatory stimulus of atherosclerosis in vessel walls. Unfortunately, in our study we did not measure apolipoprotein B levels, which reflect the number of LDL particles, but the significant difference observed in LDL-C levels at the 2-year follow-up between males with and without progression of carotid IMT implied a significant association between LDL-C levels and changes in carotid IMT. It is noteworthy that the mean LDL-C level of the progression group at the 2-year follow-up was in fact lower than the recommended therapeutic target for diabetic patient (99.9±34.6 mg/dL) (Table 4) [14], while in the nonprogression group, it was approximately 70 mg/dL (79.8±24.6 mg/dL) (Table 4). Though the follow-up LDL-C in the nonprogression group also failed to achieve the therapeutic target level (70 mg/dL) as recommended by the ADA [14], the percentage of participants who brought their LDL-C below 70 mg/dL was greater in the nonprogression group (37.1%, 13/35) than it was in the progression group (15.7%, 6/38). These findings indicated the importance of the extent that LDL-C was reduced at the follow-up period in comparison to the baseline level, and diabetic male patients with increased carotid IMT should also be regarded as high-risk patients; proper pharmacological treatment should be initiated to achieve the primary therapeutic goal of LDL-C <70 mg/dL.
In female participants in both the nonprogression and progression groups, most parameters had improved 2 years after the diagnosis compared to baseline. However, when baseline and 2-year follow-up levels were compared between groups, the mean HDL-C level at the 2-year follow-up was significantly lower in the progression group than in the nonprogression group (44.1±12.0 mg/dL vs. 53.0±14.9 mg/dL; P=0.049) (Table 5). In contrast to male patients, changes in carotid IMT appeared to be influenced by HDL-C. Therefore, in addition to pharmacologic treatment, the importance of lifestyle modifications, such as diet control and regular exercise, to raise HDL-C >50 mg/dL should be emphasized in female patients.
The UKPDS risk score is a tool designed to evaluate CVD risk in diabetic patients more accurately than the Framingham and Systematic Coronary Risk Evaluation methods [36]. Carotid IMT measurement with calculation of UKPDS engine score is known to augment the prediction ability of coronary artery stenosis [37]. Based on these findings, the additional analysis with the UKPDS engine score was intended to assess the risk of CVD in a multidimensional manner along with carotid IMT. According to a previous study, the UKPDS 10-year CHD risk in Korean patients with newly diagnosed T2DM was 14.92% [32], which was similar to our results (Table 3). We found no significant differences in CHD risk scores between those with normal and abnormal baseline IMT (Table 3), but scores at the 2-year follow-up significantly improved after 2 years in both the nonprogression and progression groups (Table 4 and 5, except for males in the progression group). The improvement in clinical parameters after 2 years of management likely contributed to this result. It should be noted that CHD risk scores in the progression groups did not improve (13.3%±7.1% vs. 11.1%±5.6%; P=0.071 in males) (Table 4) and actually increased (5.5%±2.6% vs. 5.7%±2.7%; P=0.048 in females) (Table 5), whereas CHD risk scores in the nonprogression group significantly improved after 2 years. Also, the mean risk reduction (ΔCHD risk of the UKPDS engine score) of the nonprogression group was higher than that of the progression group. These findings implied that those with advanced atherosclerosis may have a higher risk of CVD at the time of diagnosis with T2DM, but successful control of risk factors after diagnosis can markedly delay further atherosclerosis.
Our study had several limitations. First, it was a single center study, which limited the number of study participants. Second, the classification of normal and abnormal IMT was not based upon nationwide data, and other confounding factors were not statistically adjusted. Also, those who underwent carotid IMT measurement at the 2-year follow-up period and those excluded due to the follow-up loss showed significant differences with respect to their TG (P=0.048 for males) and BMI (P=0.026 for females), which could have affected the results of the analyses. Finally, we used the mean carotid IMT of the left CCA only. Different results could have been obtained if carotid IMT readings from other locations had been used.
In conclusion, levels of LDL-C in males and HDL-C in females at the 2-year follow-up after new diagnosis of T2DM rather than at the time of diagnosis with T2DM were significantly different between those with and without progression of carotid IMT. A reduction in the UKPDS 10-year CHD risk score was associated with delayed advancement of atherosclerosis. Therefore, the importance of establishing therapeutic lipid profile goals should be emphasized in newly diagnosed T2DM patients, as this could potentially halt the progression of carotid IMT. Additional large-scale studies of different population groups are needed to confirm factors contributing to the progression of carotid IMT.
Notes
No potential conflict of interest relevant to this article was reported.
References
Supplementary Material
Supplementary Table 1
The normal range of intima-media thickness at the left common carotid artery obtained from age- and gender-matched data from Health Promotion Center, Kyung Hee University Hospital